Revolutionizing Beta-Functionalization Of Aliphatic Aldehydes Via Calixarene-Assisted Palladium Catalysis For Commercial Scale-Up
The landscape of organic synthesis for pharmaceutical intermediates is constantly evolving, driven by the need for more efficient and direct functionalization strategies. A significant breakthrough in this domain is documented in patent CN113731506A, which discloses a sophisticated method for assisting palladium-catalyzed C-H arylation reactions of aliphatic aldehydes using a specialized calix[4]arene amide compound. This technology addresses a long-standing challenge in synthetic chemistry: the direct beta-functionalization of saturated fatty aldehydes without the need for cumbersome pre-activation steps. By leveraging the unique supramolecular properties of calixarene ligands, this process enables the construction of complex beta-aryl aldehyde scaffolds, which are critical building blocks for numerous bioactive molecules and drug candidates. The implications for industrial manufacturing are profound, as it simplifies synthetic routes that traditionally required multiple protection and deprotection sequences.
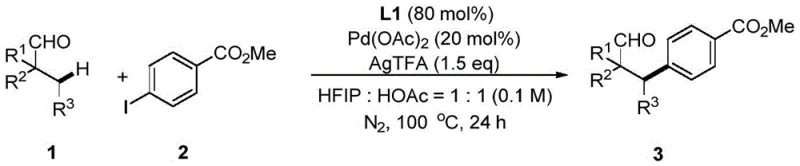
For research and development teams focused on process chemistry, the ability to directly modify the beta-position of aliphatic aldehydes represents a paradigm shift from conventional methodologies. Historically, accessing these structural motifs often relied on the addition of nucleophiles to alpha,beta-unsaturated aldehydes, a pathway that necessitates the pre-functionalization of saturated precursors and limits substrate scope. In contrast, the novel approach detailed in the patent utilizes a transition metal palladium catalyst assisted by a specifically designed calix[4]arene amide ligand, designated as L1. This system operates under relatively mild thermal conditions, typically around 100°C, in a mixed solvent system of hexafluoroisopropanol and acetic acid. The reaction proceeds with high site selectivity, effectively bypassing the thermodynamic and kinetic barriers associated with unactivated C-H bonds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for introducing aryl groups at the beta-position of aliphatic aldehydes are fraught with inefficiencies that hinder scalable manufacturing. Conventional strategies often depend on the use of alpha,beta-unsaturated aldehydes as starting materials, which themselves require synthesis from saturated precursors through oxidation or elimination reactions. This multi-step sequence not only increases the overall material cost but also introduces additional waste streams and purification burdens. Furthermore, the reactivity of unsaturated systems can lead to side reactions such as polymerization or over-reduction, complicating the impurity profile of the final active pharmaceutical ingredient. The reliance on pre-functionalized substrates restricts the chemical diversity accessible to medicinal chemists, forcing them to design syntheses around available building blocks rather than optimal molecular architectures. These limitations underscore the urgent need for a direct C-H activation strategy that can tolerate diverse functional groups while maintaining operational simplicity.
The Novel Approach
The methodology presented in CN113731506A overcomes these historical bottlenecks by employing a supramolecular catalytic system that activates inert C-H bonds directly. The core innovation lies in the use of a calix[4]arene amide compound which acts as a highly effective ligand for the palladium catalyst. This ligand architecture creates a specific microenvironment around the metal center, facilitating the coordination of the aldehyde substrate through imine formation. The reaction conditions are robust, utilizing readily available aryl iodides and simple aliphatic aldehydes such as trimethylacetaldehyde. The process eliminates the need for directing groups on the substrate, thereby reducing the step count and improving the overall atom economy of the synthesis. By streamlining the pathway to beta-aryl aldehydes, this technology offers a viable route for the cost reduction in pharmaceutical intermediates manufacturing, allowing for faster iteration in drug discovery and more efficient scale-up for commercial production.
Mechanistic Insights into Calixarene-Assisted Pd-Catalyzed Cyclization
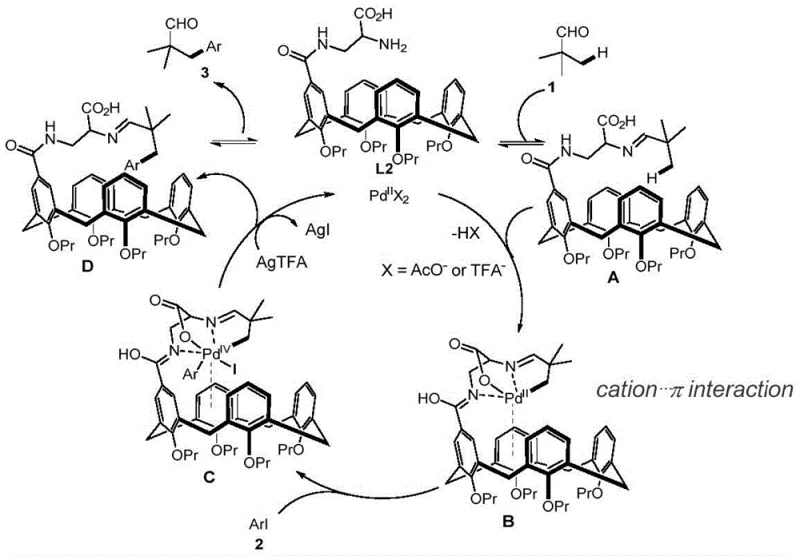
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors evaluating its feasibility for complex molecule synthesis. The proposed mechanism involves a synergistic interaction between the palladium catalyst and the calixarene ligand L1. Initially, the aliphatic aldehyde condenses with the amine moiety of the ligand to form an imine intermediate. This transient species then coordinates with the palladium center, positioning the beta-C-H bond in close proximity for activation. A key feature of this mechanism is the cation-pi interaction between the cationic palladium species and the electron-rich aromatic cavity of the calixarene. This non-covalent interaction stabilizes the transition state and lowers the activation energy required for the cyclometalation step, leading to the formation of a five-membered palladacycle intermediate. Such precise control over the coordination geometry ensures high regioselectivity, preventing unwanted arylation at other positions on the aldehyde chain.
Following the C-H activation, the catalytic cycle proceeds through oxidative addition of the aryl iodide to the palladium center, forming a high-valent palladium complex. Subsequent reductive elimination releases the arylated imine product and regenerates the active palladium species. The imine intermediate then hydrolyzes under the reaction conditions to release the free beta-aryl aldehyde and restore the calixarene ligand for another turnover. This mechanistic pathway explains the observed tolerance for various functional groups on both the aldehyde and the aryl iodide components. The ability of the calixarene ligand to modulate the electronic properties of the palladium center is instrumental in suppressing side reactions such as homocoupling of the aryl iodide or decomposition of the aldehyde. This level of mechanistic sophistication provides a solid foundation for developing high-purity pharmaceutical intermediates with consistent quality attributes.
How to Synthesize Beta-Aryl Aliphatic Aldehydes Efficiently
Implementing this advanced synthetic methodology requires careful attention to reaction parameters to maximize yield and purity. The protocol outlined in the patent data provides a clear roadmap for executing the transformation on a laboratory scale, which can be adapted for larger batches with appropriate engineering controls. The procedure involves combining the aliphatic aldehyde and aryl iodide substrates with the calixarene ligand L1, a palladium source such as palladium acetate, and a silver salt additive in a solvent mixture of hexafluoroisopropanol and acetic acid. The reaction is conducted under an inert nitrogen atmosphere to prevent oxidation of sensitive intermediates. Detailed standardized synthesis steps see the guide below.
- Combine aliphatic aldehyde, aryl iodide, calixarene ligand L1, silver salt additive, and palladium catalyst in a mixed solvent of hexafluoroisopropanol and acetic acid.
- Stir the reaction mixture vigorously under nitrogen protection at 100°C for approximately 24 hours to ensure complete conversion.
- Filter the reaction mixture through diatomaceous earth to remove insolubles, concentrate the filtrate, and purify the crude product via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this calixarene-assisted technology offers significant strategic benefits beyond mere technical novelty. The simplification of the synthetic route directly translates to enhanced supply chain reliability by reducing the number of discrete processing steps and intermediate isolations required. Fewer steps mean fewer opportunities for yield loss, equipment bottlenecks, and quality deviations, resulting in a more robust and predictable manufacturing timeline. Additionally, the use of stable and commercially available starting materials like simple aliphatic aldehydes and aryl iodides mitigates the risk of raw material shortages that often plague complex custom synthesis projects. This stability in the supply base allows for better long-term planning and inventory management, ensuring continuous availability of critical intermediates for downstream drug production.
- Cost Reduction in Manufacturing: The elimination of pre-functionalization steps and protecting group manipulations leads to substantial cost savings in raw materials and processing time. By avoiding the use of expensive and specialized reagents required for traditional alpha,beta-unsaturated aldehyde synthesis, the overall cost of goods sold is significantly optimized. Furthermore, the simplified workup procedure, which involves basic filtration and chromatography, reduces the consumption of solvents and consumables associated with complex purification protocols. The efficiency of the catalytic system also implies that lower loadings of expensive palladium catalysts may be achievable with further optimization, contributing to a leaner cost structure for high-value chemical production.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals as starting materials ensures a diversified and resilient supply base that is less susceptible to market volatility. Unlike proprietary building blocks that may be sourced from a single vendor, the substrates for this reaction are widely produced by multiple global suppliers, reducing the risk of supply disruption. The robustness of the reaction conditions, which tolerate standard laboratory glassware and heating mantles, facilitates technology transfer between different manufacturing sites without the need for specialized equipment. This flexibility empowers supply chain managers to establish redundant production capabilities, safeguarding against unforeseen logistical challenges and ensuring uninterrupted delivery to customers.
- Scalability and Environmental Compliance: The process demonstrates excellent potential for commercial scale-up due to its homogeneous nature and manageable exotherm profile. The use of a mixed solvent system that can be recovered and recycled aligns with modern green chemistry principles, minimizing the environmental footprint of the manufacturing operation. The absence of hazardous reagents and the generation of minimal inorganic waste simplify the disposal process and reduce compliance costs associated with environmental regulations. As regulatory scrutiny on pharmaceutical manufacturing intensifies, adopting cleaner and more efficient technologies like this positions companies favorably for audits and inspections, fostering trust with regulatory bodies and end-users alike.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and disclosure sections of the source intellectual property, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the fit of this technology within their existing portfolios and manufacturing frameworks.
Q: What is the primary advantage of using calixarene ligands in this C-H activation process?
A: The calix[4]arene amide ligand facilitates site-selective activation of the beta-C-H bond through cation-pi interactions with the palladium center, eliminating the need for pre-functionalization of the aldehyde substrate.
Q: Does this method require harsh pre-activation steps for the aldehyde substrate?
A: No, unlike conventional methods that require alpha,beta-unsaturated precursors or protecting groups, this protocol allows for direct arylation of saturated aliphatic aldehydes in a single step.
Q: What are the optimal reaction conditions described in the patent data?
A: The optimal conditions involve using palladium acetate as the catalyst, silver trifluoroacetate as the additive, and a 1:1 volume ratio of hexafluoroisopropanol to acetic acid at 100°C under nitrogen.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Aryl Aliphatic Aldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methods like the calixarene-assisted C-H arylation described in CN113731506A. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into reliable industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that monitor every batch for impurities and residual metals. We understand that consistency is key for pharmaceutical clients, and our state-of-the-art facilities are equipped to handle the specific solvent systems and temperature controls required for this sophisticated chemistry.
We invite you to collaborate with us to leverage this technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this route can optimize your budget without compromising quality. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your target compounds. Let us help you accelerate your development timeline and secure a competitive advantage in the global market through superior chemical manufacturing solutions.
