Advanced Industrial Synthesis Of 4-Bromopyridazine For Global Pharmaceutical Supply Chains
Advanced Industrial Synthesis Of 4-Bromopyridazine For Global Pharmaceutical Supply Chains
The pharmaceutical and agrochemical industries continuously demand high-purity heterocyclic building blocks to construct complex active pharmaceutical ingredients (APIs) with enhanced biological activity. Patent CN102924386A introduces a robust and economically viable industrial preparation method for 4-bromopyridazine, a critical intermediate known for its utility in developing antihypertensive, cardiotonic, and antiviral agents. This technology addresses the historical challenges associated with synthesizing pyridazine derivatives, which often suffer from difficult reaction conditions and prohibitive costs. By leveraging a streamlined four-step sequence starting from the inexpensive and abundant 3,6-dichloropyridazine, this method achieves high yields while maintaining operational simplicity. The structural integrity of the final product, as depicted below, is crucial for downstream coupling reactions in drug discovery pipelines.
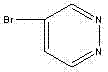
Furthermore, the patent highlights significant improvements in process safety and scalability, particularly in the final bromination step where traditional methods often pose thermal risks. For R&D directors and procurement specialists, understanding this pathway offers a strategic advantage in securing reliable supply chains for nitrogen-containing heterocycles. The ability to produce 4-bromopyridazine with purity levels exceeding 98% ensures that subsequent synthetic steps in API manufacturing proceed with minimal impurity profiles, thereby reducing the burden on downstream purification processes. This technological breakthrough represents a substantial shift towards more efficient and cost-effective manufacturing of specialized chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of halogenated pyridazines has been plagued by inefficient multi-step routes that rely on expensive starting materials or harsh reaction conditions that are difficult to control on a large scale. Conventional methods often involve low-yielding substitution reactions that generate significant amounts of hazardous waste, complicating environmental compliance and increasing disposal costs for manufacturing facilities. Additionally, the lack of regioselectivity in traditional halogenation processes can lead to complex mixtures of isomers, necessitating energy-intensive chromatographic separations that drastically reduce overall throughput. These inefficiencies create bottlenecks in the supply chain, leading to volatile pricing and inconsistent availability of key intermediates like 4-bromopyridazine for global pharmaceutical clients. The reliance on unstable reagents or conditions requiring extreme temperatures further exacerbates safety risks, making scale-up a formidable challenge for process chemists aiming to meet commercial demand.
The Novel Approach
In contrast, the methodology outlined in CN102924386A presents a transformative approach by utilizing 3,6-dichloropyridazine, a commercially abundant and cost-effective feedstock, to access the target molecule through a logical and high-yielding sequence. This novel route eliminates the need for exotic reagents and instead employs standard industrial unit operations such as chlorination, hydrolysis, catalytic hydrogenation, and bromination. A key innovation lies in the final transformation, where the conversion of 4-hydroxypyridazine to 4-bromopyridazine is optimized to manage the exothermic nature of phosphorus oxybromide reactions safely. As illustrated in the reaction scheme below, the direct bromination using POBr3 allows for precise control over reaction parameters, ensuring high conversion rates without compromising safety.
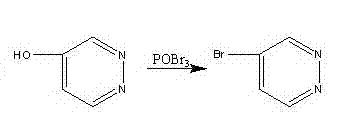
By streamlining the synthesis into just four distinct steps, the process minimizes material handling and reduces the cumulative loss of yield typically seen in longer synthetic pathways. This efficiency translates directly into lower production costs and a reduced carbon footprint, aligning with modern green chemistry principles. The method's robustness is evidenced by successful scaling from gram-level laboratory experiments to multi-kilogram pilot runs, demonstrating its readiness for immediate industrial adoption. For supply chain managers, this reliability means a consistent source of high-quality intermediates, mitigating the risks associated with supplier disruptions and market fluctuations.
Mechanistic Insights into Catalytic Hydrogenation and Bromination
The core of this synthesis strategy relies on two pivotal chemical transformations: the catalytic hydrodechlorination and the subsequent nucleophilic substitution bromination. In the third step, the removal of chlorine atoms from the pyridazine ring is achieved through catalytic hydrogenation using palladium on carbon (Pd/C) under controlled pressure. This step is critical for establishing the correct oxidation state and functional group arrangement required for the final product. The mechanism involves the adsorption of hydrogen onto the palladium surface, followed by the oxidative addition of the carbon-chlorine bond and subsequent reductive elimination to form the carbon-hydrogen bond. Maintaining the reaction temperature between 20°C and 80°C and hydrogen pressure at 1.0-2.0 MPa ensures complete conversion while preventing over-reduction of the heterocyclic ring system.
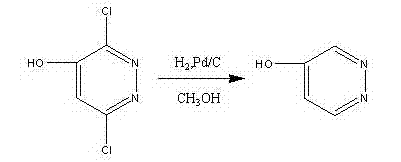
Following the formation of 4-hydroxypyridazine, the final step employs phosphorus oxybromide (POBr3) to effect the conversion of the hydroxyl group to a bromide. This reaction proceeds via the activation of the hydroxyl oxygen by the phosphorus species, creating a good leaving group that is subsequently displaced by a bromide ion. The patent emphasizes the importance of the workup procedure, where the reaction mixture is concentrated and carefully quenched with ice water to neutralize excess POBr3. This controlled quenching prevents violent exotherms and ensures the safe isolation of the product. The resulting crude material is then purified through extraction and recrystallization, yielding 4-bromopyridazine with exceptional purity suitable for sensitive pharmaceutical applications.
How to Synthesize 4-Bromopyridazine Efficiently
The synthesis of 4-bromopyridazine via this patented route requires precise adherence to reaction conditions to maximize yield and safety. The process begins with the chlorination of the starting material, followed by hydrolysis to introduce the hydroxyl functionality. The subsequent catalytic reduction and bromination steps demand careful monitoring of temperature and pressure to ensure reproducibility. Operators must be trained in handling reactive halogenating agents and high-pressure hydrogenation equipment to maintain operational excellence. For a comprehensive guide on the specific stoichiometry, solvent choices, and purification techniques detailed in the patent, please refer to the standardized synthesis protocol provided below.
- Chlorinate 3,6-dichloropyridazine at 80-160°C with chlorine gas to form 3,4,6-trichloropyridazine.
- Hydrolyze the trichloro-intermediate with NaOH solution at 100°C to yield 3,6-dichloro-4-hydroxypyridazine.
- Perform catalytic hydrogenation using Pd/C catalyst under 1.0-2.0 MPa pressure to remove chlorine atoms and form 4-hydroxypyridazine.
- React 4-hydroxypyridazine with phosphorus oxybromide (POBr3) at 60-120°C, followed by careful quenching and purification to obtain 4-bromopyridazine.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this manufacturing process offers profound benefits for procurement strategies and supply chain stability in the fine chemicals sector. By anchoring the synthesis on 3,6-dichloropyridazine, a commodity chemical with a stable global supply, manufacturers can decouple their production costs from the volatility associated with rare or specialized precursors. This foundational stability allows for more accurate long-term forecasting and budget planning, essential for maintaining healthy margins in competitive markets. Furthermore, the simplification of the post-treatment procedures, particularly in the bromination stage, reduces the requirement for specialized containment infrastructure and extensive waste treatment protocols. These operational efficiencies collectively contribute to a leaner manufacturing model that is both economically resilient and environmentally sustainable.
- Cost Reduction in Manufacturing: The utilization of inexpensive raw materials combined with a high-yielding four-step sequence significantly lowers the cost of goods sold (COGS) for 4-bromopyridazine. The elimination of complex purification steps and the high atom economy of the chlorination and hydrolysis stages further drive down production expenses. By avoiding the use of precious metal catalysts in non-critical steps and optimizing the loading of palladium in the hydrogenation step, the process minimizes capital tied up in catalyst recovery systems. These factors culminate in a highly competitive pricing structure that provides substantial cost savings compared to legacy synthetic routes.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals ensures that production schedules are not disrupted by raw material shortages. The robustness of the reaction conditions, which tolerate slight variations in temperature and pressure without significant yield loss, adds a layer of operational flexibility that is crucial for meeting tight delivery deadlines. Additionally, the scalability demonstrated in the patent examples confirms that the process can be ramped up quickly to accommodate surges in demand from downstream API manufacturers. This reliability fosters stronger partnerships between chemical suppliers and pharmaceutical companies, ensuring a continuous flow of critical intermediates.
- Scalability and Environmental Compliance: The process design inherently supports large-scale production, with safety measures integrated directly into the workflow, such as the controlled quenching of reactive byproducts. This proactive approach to safety reduces the risk of plant shutdowns due to incidents, thereby guaranteeing consistent output. Moreover, the simplified waste stream, characterized by fewer organic solvents and manageable inorganic salts, facilitates easier compliance with increasingly stringent environmental regulations. The ability to operate within standard industrial parameters makes this technology an ideal candidate for green manufacturing initiatives and sustainable supply chain certifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the industrial production of 4-bromopyridazine. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on the feasibility and advantages of this specific synthetic route. Understanding these details is essential for technical teams evaluating potential suppliers or considering technology transfer opportunities. The answers reflect a commitment to transparency and technical accuracy, ensuring that stakeholders have the information needed to make informed decisions.
Q: What are the primary advantages of the 4-step synthesis method described in CN102924386A?
A: The method utilizes cheap and readily available 3,6-dichloropyridazine as a starting material, features a short reaction sequence with high overall yields, and employs a simplified post-treatment process for the bromination step that enhances safety during scale-up.
Q: How does the patent address safety concerns during the bromination step?
A: The process optimizes the post-treatment of the bromination reaction by carefully controlling the quenching of excess phosphorus oxybromide with ice water and adjusting pH with ammonia, significantly reducing the risk of exothermic runaway reactions common in large-scale halogenation.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the patent explicitly demonstrates scalability through examples ranging from gram-scale to kilogram-scale (up to 10kg input), confirming that the reaction conditions, such as temperature control and catalyst loading, are robust enough for industrial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Bromopyridazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates like 4-bromopyridazine play in the development of life-saving medications and advanced agrochemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and consistency. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest international standards. Our commitment to process optimization allows us to deliver products that not only meet but exceed the expectations of discerning R&D and procurement professionals globally.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can enhance your supply chain efficiency and reduce your overall manufacturing costs. Let us be your partner in innovation and reliability.
