Advanced Synthesis of Polysubstituted Tetrahydrofuran and Tetrahydropyran Dienoids for Commercial Scale-up
Introduction to Novel Dienoid Synthesis Technology
The landscape of organic synthesis is continually evolving to meet the rigorous demands of the pharmaceutical and fine chemical industries. A significant breakthrough in this domain is documented in Chinese patent CN112142694A, which discloses a highly efficient method for preparing polysubstituted tetrahydrofuran and tetrahydropyran dienoid compounds. These specific chemical structures, represented generally by Formula I, are not merely academic curiosities but serve as indispensable building blocks for the construction of complex bioactive molecules. Their utility extends into the synthesis of benzofuran and benzopyran derivatives, which are privileged scaffolds found in numerous drug candidates targeting HIV, tumors, and fungal infections. The ability to access these densely functionalized heterocycles through a streamlined process represents a major advancement for R&D teams seeking reliable pathways to high-value intermediates.
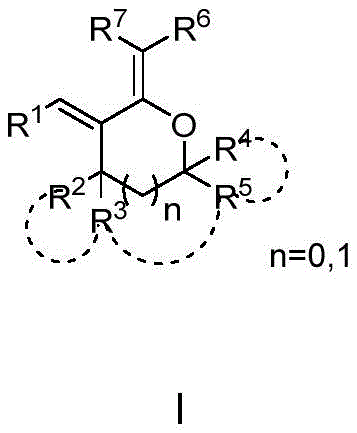
Furthermore, the versatility of these dienoids allows them to function as monomers for polymer synthesis and potentially as active pharmaceutical ingredients themselves. The patent highlights a strategic approach that bypasses the limitations of traditional methodologies, offering a route that is both operationally simple and economically viable. For procurement managers and supply chain directors, understanding the underlying chemistry of such innovations is crucial for securing long-term sources of high-purity pharmaceutical intermediates. This report delves deep into the mechanistic nuances and commercial implications of this technology, providing a comprehensive analysis for stakeholders aiming to optimize their supply chains for complex organic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted tetrahydrofuran and tetrahydropyran dienes has been fraught with challenges that hinder large-scale adoption. Prior art, including references such as J. Chem. Soc., Chem. Commun. (1986) and various Organic Letters publications, often describes routes that suffer from low atom economy, harsh reaction conditions, or the requirement for exotic reagents that are difficult to source commercially. Traditional methods may involve multi-step sequences with poor overall yields, leading to significant waste generation and inflated production costs. Additionally, the use of sensitive reagents often necessitates stringent exclusion of moisture and oxygen, complicating the engineering requirements for reactor setups. These factors collectively contribute to extended lead times and reduced reliability in the supply of these critical intermediates, creating bottlenecks for downstream drug development projects.
The Novel Approach
In stark contrast, the methodology outlined in patent CN112142694A introduces a robust two-step strategy that dramatically simplifies the production workflow. The novel approach utilizes readily available alpha-alkenyl lactones and common nucleophiles such as nitriles, nitroalkanes, or esters. By employing a base-mediated 1,2-addition followed by a catalytic dehydration, the process achieves high conversion rates under relatively mild thermal conditions ranging from -78°C to 60°C. This reduction in operational severity translates directly into enhanced safety profiles and lower energy consumption. The simplicity of the reaction design means that standard glass-lined or stainless-steel reactors can be utilized without the need for specialized high-pressure or cryogenic equipment beyond standard cooling capabilities. This accessibility makes the technology particularly attractive for cost reduction in pharmaceutical intermediate manufacturing, allowing producers to scale up with confidence.
Mechanistic Insights into Base-Mediated Addition and Acid-Catalyzed Dehydration
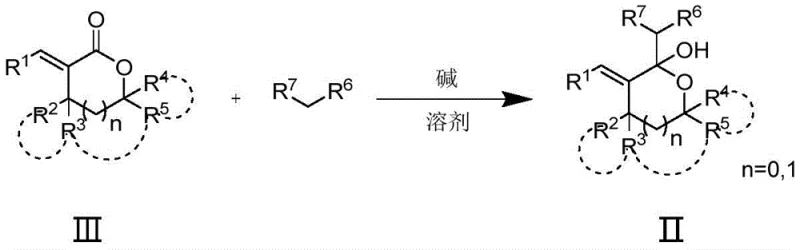
The core of this synthetic innovation lies in its elegant two-stage mechanism, which ensures high regioselectivity and minimal byproduct formation. The first stage involves the nucleophilic attack on the alpha-alkenyl lactone (Formula III). In the presence of a base such as sodium ethoxide, potassium tert-butoxide, or even Grignard reagents, the nucleophile adds to the carbonyl or conjugated system to generate a hemiacetal intermediate (Formula II). This step is critical as it establishes the carbon framework and stereochemistry of the final product. The reaction is typically conducted in ether solvents like tetrahydrofuran or 1,4-dioxane, which stabilize the ionic intermediates effectively. The control of temperature during this phase, often between 0°C and 30°C, is vital to prevent side reactions and ensure the formation of the desired hemiacetal with high purity.
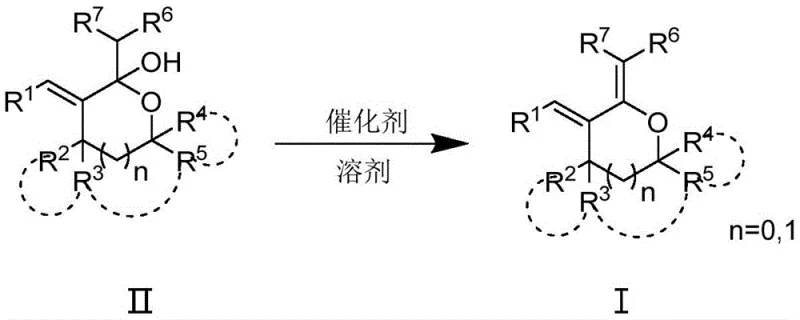
Following the isolation of the hemiacetal, the second stage employs a Lewis acid or Bronsted acid catalyst to drive dehydration. Catalysts such as iron(III) chloride, copper(II) triflate, or zinc acetate facilitate the elimination of water to form the exocyclic double bond characteristic of the dienoid system (Formula I). The preference for iron-based catalysts is particularly noteworthy from a commercial perspective, as iron salts are abundant, non-toxic, and inexpensive compared to palladium or rhodium alternatives. This dehydration step proceeds efficiently at temperatures between 20°C and 50°C, further underscoring the energy efficiency of the process. The mechanistic pathway avoids the formation of stable impurities that are difficult to separate, thereby streamlining the purification process and enhancing the overall yield of the target high-purity OLED material or pharmaceutical precursor.
How to Synthesize Polysubstituted Tetrahydrofuran Dienoids Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and reaction monitoring to maximize yield and purity. The process begins with the precise weighing of the nucleophile and solvent, followed by the controlled addition of the base to generate the reactive species. The alpha-alkenyl lactone is then introduced dropwise to manage the exotherm and ensure complete conversion. After the first reaction phase, standard aqueous workup and extraction techniques are employed to isolate the hemiacetal intermediate. This intermediate is then subjected to the dehydration conditions using the selected catalyst. Detailed standard operating procedures regarding specific molar ratios, solvent volumes, and quenching protocols are essential for reproducibility. For a comprehensive guide on the exact experimental parameters and safety precautions, please refer to the standardized synthesis steps provided below.
- Perform a 1,2-addition reaction between an alpha-alkenyl lactone and a nucleophile (such as malononitrile) in the presence of a base and ether solvent at temperatures ranging from -78°C to 60°C to form a hemiacetal intermediate.
- Isolate the hemiacetal intermediate through standard workup procedures such as aqueous quenching and extraction, ensuring high purity before the subsequent dehydration step.
- Subject the hemiacetal to dehydration using a Lewis acid or Bronsted acid catalyst (e.g., FeCl3 or Cu(OTf)2) in a suitable solvent at 20°C to 50°C to yield the final polysubstituted tetrahydrofuran dienoid compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented technology offers substantial benefits that address key pain points in the chemical supply chain. The shift towards using earth-abundant metal catalysts like iron and copper eliminates the dependency on volatile precious metal markets, stabilizing raw material costs. Furthermore, the operational simplicity of the process reduces the burden on manufacturing facilities, allowing for faster turnaround times and increased throughput. This efficiency is critical for maintaining supply continuity in the face of fluctuating market demands. By adopting this methodology, companies can achieve significant cost savings in fine chemical intermediates production without compromising on the quality or purity specifications required for downstream applications.
- Cost Reduction in Manufacturing: The utilization of inexpensive iron-based catalysts instead of precious metals drastically lowers the direct material costs associated with the synthesis. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to lower utility bills. The high yields reported in the patent examples minimize waste disposal costs and maximize the output per batch, leading to a more favorable cost of goods sold (COGS) profile for the final intermediate.
- Enhanced Supply Chain Reliability: The starting materials, including alpha-alkenyl lactones and common nitriles, are commercially available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions means that production is less susceptible to minor variations in environmental factors, ensuring consistent batch-to-batch quality. This reliability is paramount for pharmaceutical clients who require strict adherence to supply agreements and regulatory standards.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively with simple equipment setups that can be easily expanded from pilot to commercial scale. The use of less hazardous reagents and the generation of fewer toxic byproducts align with modern environmental regulations and green chemistry initiatives. This compliance reduces the regulatory burden and facilitates smoother permitting processes for manufacturing sites, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled answers to common inquiries regarding the synthesis and application of these compounds. These responses are derived directly from the technical disclosures within the patent documentation and reflect the current state of the art in heterocyclic chemistry. Understanding these details is essential for assessing the feasibility of integrating this route into existing production pipelines. The following section addresses specific concerns regarding catalyst selection, substrate scope, and potential industrial applications.
Q: What are the primary applications of these polysubstituted tetrahydrofuran dienoids?
A: These compounds serve as critical synthons for preparing polysubstituted benzofurans and benzopyrans via Diels-Alder reactions. They are widely utilized in the development of pharmaceutical agents with anti-HIV, anti-tumor, and antifungal activities, as well as in materials science for polymer synthesis.
Q: Why is the iron-based catalytic system preferred for this synthesis?
A: Iron-based catalysts, such as FeCl3 or Fe(acac)3, offer superior catalytic activity while being significantly more cost-effective and environmentally benign compared to precious metal catalysts. This choice aligns with green chemistry principles and reduces overall production costs.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the process is highly scalable due to its mild reaction conditions (0°C to 50°C), use of commercially available raw materials, and simple purification methods like extractive distillation. The robustness of the two-step sequence ensures consistent quality and supply continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Tetrahydrofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in CN112142694A. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a steady supply of high-quality intermediates. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that validate every batch against the highest industry standards. We understand that in the competitive landscape of pharmaceutical development, time and quality are the most valuable currencies, and we are dedicated to delivering both.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your path to market while optimizing your production costs.
