Scalable Synthesis of Febuxostat Intermediate: Technical & Commercial Insights
Scalable Synthesis of Febuxostat Intermediate: Technical & Commercial Insights
The pharmaceutical landscape for gout management has been significantly transformed by xanthine oxidase inhibitors, with Febuxostat standing out as a potent therapeutic agent. Central to the supply chain of this critical medication is the efficient production of its key precursor, 2-(3-cyano-4-isobutoxyphenyl)-4-methyl-5-thiazolecarboxylic acid. Patent CN101497589B discloses a robust and industrially viable synthetic methodology that addresses historical bottlenecks in manufacturing this complex heterocyclic compound. By leveraging a strategic sequence of iodination, alkylation, and cyanation reactions starting from commodity chemicals, this technology offers a pathway to high-purity intermediates essential for API production. For R&D directors and procurement specialists, understanding the nuances of this patented route is vital for securing a reliable supply of high-purity pharmaceutical intermediates while optimizing production costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing this thiazole carboxylic acid derivative have historically suffered from significant economic and operational drawbacks that hinder large-scale adoption. Traditional routes often relied on expensive and difficult-to-source starting materials, such as 4-hydroxyphenyl thioformamide, which drastically inflated raw material costs and introduced supply chain volatility. Furthermore, earlier processes described in Japanese patents frequently necessitated rigorous purification steps, specifically column chromatography, to isolate key intermediates like 4-isobutoxy-1,3-benzenedinitrile. In an industrial setting, column chromatography is notoriously inefficient, consuming vast quantities of solvents and silica gel while limiting batch sizes, thereby rendering the process economically unfeasible for metric-ton production. These legacy methods also involved longer synthetic sequences with lower overall yields, compounding the cost burden and generating excessive chemical waste that complicated environmental compliance.
The Novel Approach
The innovative strategy outlined in the patent data circumvents these obstacles by employing 4-hydroxybenzonitrile as a cost-effective and readily available feedstock. This novel approach streamlines the synthesis into a logical progression of iodination, etherification, and cyanation to construct the core aromatic scaffold before introducing the thiazole ring. A critical advantage of this method is the elimination of column chromatography; intermediates are purified through crystallization and filtration, operations that are inherently scalable and solvent-efficient. By utilizing cuprous cyanide for the cyanation step and sodium hydrosulfide for thioamidation under controlled conditions, the process achieves high conversion rates without requiring exotic catalysts. This shift from laboratory-scale purification techniques to unit operations compatible with standard chemical reactors represents a paradigm shift in cost reduction in pharmaceutical intermediates manufacturing, ensuring consistent quality and supply continuity.
Mechanistic Insights into the Multi-Step Synthesis Route
The chemical architecture of this synthesis relies on precise regioselective functionalization of the benzene ring to ensure the correct positioning of the cyano and isobutoxy groups. The process initiates with the electrophilic iodination of 4-hydroxybenzonitrile, where the hydroxyl group directs the iodine atom to the ortho-position, forming 3-iodo-4-hydroxybenzonitrile. Following this, an SN2 nucleophilic substitution occurs where the phenolic oxygen attacks isobutyl bromide in the presence of a base like potassium carbonate, effectively installing the isobutoxy moiety. The subsequent Rosenmund-von Braun type cyanation replaces the iodine atom with a nitrile group using cuprous cyanide at elevated temperatures, yielding the pivotal 4-isobutoxy-1,3-benzenedinitrile intermediate. This specific dinitrile structure serves as the foundation for the heterocyclic ring formation, demonstrating the importance of maintaining high purity at this stage to prevent downstream impurities.
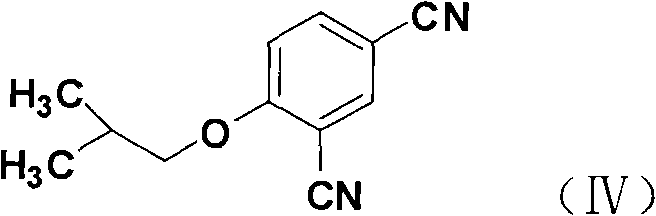
Following the construction of the substituted benzonitrile core, the mechanism proceeds through a selective thioamidation where one nitrile group is converted into a thioamide functionality using sodium hydrosulfide and magnesium chloride. This transformation is critical as it provides the necessary sulfur and nitrogen atoms for the subsequent cyclization. The final ring closure involves the reaction of this thioamide with ethyl 2-chloroacetoacetate, forming the thiazole ring via condensation and cyclization. The resulting ethyl ester is then hydrolyzed under basic conditions to yield the free carboxylic acid. Throughout this cascade, impurity control is managed by the specificity of the reagents; for instance, the use of anhydrous magnesium chloride during thioamidation helps moderate the reactivity of sodium hydrosulfide, preventing over-reaction or degradation of the sensitive nitrile groups, thereby ensuring the final product meets stringent purity specifications.
How to Synthesize 2-(3-cyano-4-isobutoxyphenyl)-4-methyl-5-thiazolecarboxylic acid Efficiently
Executing this synthesis requires careful attention to reaction parameters such as temperature control and stoichiometry to maximize yield and minimize byproduct formation. The patent details specific molar ratios and solvent systems, such as using DMF for the alkylation and cyanation steps to ensure solubility of the inorganic salts and organic substrates. Operators must monitor the exothermic nature of the iodination and maintain pH levels during the workup phases to ensure complete precipitation of the products. While the general workflow is established, precise optimization of stirring rates and addition speeds is often necessary when transitioning from gram-scale experiments to kiloliter reactors. Detailed standardized operating procedures for each unit operation are essential to replicate the high yields reported in the patent examples consistently.
- Perform iodination of 4-hydroxybenzonitrile using iodine and potassium iodide in aqueous ammonia to form 3-iodo-4-hydroxybenzonitrile.
- Execute alkylation with isobutyl bromide and subsequent cyanation with cuprous cyanide to generate 4-isobutoxy-1,3-benzenedinitrile.
- Conduct thioamidation using sodium hydrosulfide, followed by cyclization with ethyl 2-chloroacetoacetate and final hydrolysis to yield the target acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond simple chemistry, directly impacting the bottom line and operational resilience. The primary advantage lies in the sourcing strategy; by utilizing 4-hydroxybenzonitrile and isobutyl bromide, manufacturers can tap into established global supply chains for commodity chemicals rather than relying on bespoke, low-volume specialty reagents. This shift significantly mitigates the risk of raw material shortages and price spikes that often plague the pharmaceutical supply chain. Furthermore, the simplification of the purification process removes the bottleneck of chromatographic separation, which is a major driver of both capital expenditure and operating costs in fine chemical manufacturing. This streamlined approach allows for faster batch turnover times and reduced solvent inventory requirements.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive purification media and the reduction in solvent usage associated with column chromatography. By switching to crystallization-based purification, the process drastically lowers the cost of goods sold (COGS) through reduced waste disposal fees and lower energy consumption for solvent recovery. Additionally, the higher overall yield achieved through this optimized route means that less raw material is required to produce the same amount of final product, further enhancing margin potential. The avoidance of precious metal catalysts or complex reagents also contributes to a leaner cost structure, making the final intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of robust, non-proprietary starting materials that are produced by multiple vendors worldwide. This diversification of the supply base reduces dependency on single-source suppliers and minimizes the risk of production stoppages due to upstream disruptions. The simplicity of the reaction conditions, which do not require extreme pressures or cryogenic temperatures, also means that the process can be manufactured in a wider range of facilities, increasing the available capacity pool. Consequently, lead times for high-purity pharmaceutical intermediates can be stabilized, ensuring that downstream API manufacturers receive their materials on schedule without unexpected delays.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this route is designed for industrial viability, adhering to green chemistry principles by minimizing waste generation. The absence of silica gel waste from chromatography columns significantly reduces the solid waste burden, simplifying disposal and lowering environmental compliance costs. The process is inherently scalable because it relies on standard unit operations like filtration, distillation, and crystallization, which can be easily enlarged from pilot plants to full commercial production scales. This scalability ensures that as demand for the final gout medication grows, the supply of the intermediate can be ramped up seamlessly without requiring fundamental changes to the manufacturing infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of this critical pharmaceutical intermediate. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry stakeholders. Understanding these details helps partners make informed decisions regarding technology transfer and procurement strategies. We encourage further discussion with our technical team to explore how these capabilities align with your specific project requirements.
Q: Why is this synthesis route preferred over prior art methods?
A: This route utilizes readily available starting materials like 4-hydroxybenzonitrile and avoids expensive reagents such as 4-hydroxyphenyl thioformamide. Crucially, it eliminates the need for column chromatography purification for intermediates, making it highly suitable for industrial scale-up.
Q: What are the key purity specifications achievable?
A: The patent specifies that the final product can achieve an HPLC purity of ≥99.9% through recrystallization steps, ensuring high quality suitable for pharmaceutical applications without extensive downstream processing.
Q: Is the process safe for large-scale manufacturing?
A: Yes, the process operates under relatively mild conditions (e.g., 50-80°C for alkylation) and uses standard reagents. The avoidance of hazardous purification techniques and the use of stable intermediates enhance overall operational safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(3-cyano-4-isobutoxyphenyl)-4-methyl-5-thiazolecarboxylic acid Supplier
At NINGBO INNO PHARMCHEM, we combine deep technical expertise with robust manufacturing capabilities to deliver high-quality pharmaceutical intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the clinical trial phase or full commercial launch. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-(3-cyano-4-isobutoxyphenyl)-4-methyl-5-thiazolecarboxylic acid meets the highest industry standards. Our commitment to quality and consistency makes us a trusted partner for multinational pharmaceutical companies seeking a reliable source for complex heterocyclic building blocks.
We invite you to engage with our technical procurement team to discuss your specific needs and explore how our optimized synthesis routes can benefit your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages of partnering with us. We are ready to provide specific COA data and route feasibility assessments to support your vendor qualification process. Contact us today to secure a sustainable and cost-effective supply of this critical intermediate for your gout medication projects.
