Advanced Organocatalytic Synthesis of Sulfonyl Substituted Styrene Axial Chiral Compounds
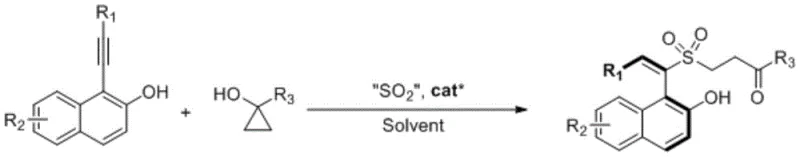
The landscape of asymmetric synthesis is continually evolving, driven by the demand for efficient, scalable, and environmentally benign methodologies for constructing complex chiral architectures. A significant breakthrough in this domain is documented in Chinese Patent CN113429323A, which discloses a novel preparation method for sulfonyl substituted styrene type axial chiral compounds. This technology represents a paradigm shift from traditional transition metal-catalyzed processes to a sophisticated metal-free organocatalytic system. By leveraging a three-component reaction involving 1-substituted cyclopropanols, solid sulfur dioxide surrogates, and 1-alkynyl-2-naphthols, this invention successfully constructs axially chiral styrene frameworks with exceptional stereocontrol. The significance of this development cannot be overstated for the fine chemical industry, as axial chiral compounds serve as critical scaffolds for chiral ligands, organocatalysts, and functional materials. The ability to access these structures under mild conditions without the burden of heavy metal contamination addresses a persistent pain point for pharmaceutical manufacturers seeking reliable sulfonyl substituted styrene suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of styrene-based axial chiral compounds has been fraught with significant technical hurdles that impede large-scale adoption. Conventional strategies often rely heavily on transition metal catalysis, which introduces a myriad of complications including high catalyst costs, sensitivity to air and moisture, and the stringent requirement for residual metal removal to meet pharmaceutical standards. Furthermore, existing methods for introducing sulfonyl groups frequently necessitate the pre-synthesis and isolation of sulfinate salts, which are often unstable and require careful storage and handling. These multi-step sequences not only increase the overall production time but also accumulate waste, negatively impacting the atom economy and environmental footprint of the process. Additionally, many traditional routes suffer from limited substrate scope and struggle to maintain high enantiomeric purity due to the relatively low rotational energy barrier of the styrene axis, leading to potential racemization during synthesis or purification.
The Novel Approach
In stark contrast, the methodology outlined in patent CN113429323A offers a streamlined, one-pot solution that elegantly circumvents these legacy issues. The core innovation lies in the in-situ generation of reactive intermediates: the ring-opening of cyclopropanol coupled with the release of sulfur dioxide from stable solid surrogates generates sulfinic acid anions directly within the reaction matrix. This eliminates the need for isolating sensitive sulfinate precursors. Coupled with a highly active chiral small molecule catalyst, this system activates the 1-alkynyl-2-naphthol to form a dienoquinone intermediate, which then undergoes a highly stereoselective nucleophilic attack. The result is a robust protocol that operates at room temperature, tolerates a wide variety of functional groups, and delivers products with yields up to 87 percent and ee values reaching 98 percent. This approach not only simplifies the operational complexity but also drastically reduces the cost reduction in chiral intermediate manufacturing by minimizing unit operations and reagent costs.
Mechanistic Insights into Organocatalytic Asymmetric Sulfonylation
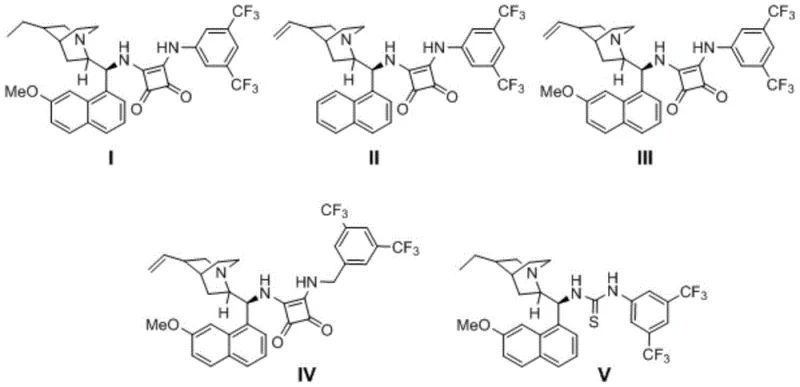
The success of this transformation is fundamentally rooted in the precise design of the chiral organocatalyst, which acts as a dual-activation platform. The catalysts employed, typically derivatives of cinchona alkaloids functionalized with thiourea or squaramide moieties, utilize a network of hydrogen-bonding interactions to organize the transition state. Specifically, the catalyst simultaneously activates the electrophilic dienoquinone intermediate generated from the naphthol alkyne and stabilizes the nucleophilic sulfinic acid anion. This bifunctional activation lowers the activation energy of the C-S bond formation while imposing a rigid chiral environment that dictates the facial selectivity of the attack. The steric bulk of the catalyst framework effectively shields one face of the planar intermediate, forcing the nucleophile to approach from the preferred trajectory, thereby locking the axial chirality with high fidelity. This mechanism avoids the use of toxic heavy metals, aligning with green chemistry principles while delivering performance metrics that rival or exceed those of precious metal systems.
Beyond the primary catalytic cycle, the impurity control mechanism inherent in this design is equally impressive. The mild reaction conditions, typically maintained between 15°C and 40°C, prevent thermal degradation pathways that often plague high-temperature syntheses. Moreover, the use of solid sulfur dioxide surrogates such as DABCO(SO2)2 ensures a controlled, steady release of SO2, preventing local concentration spikes that could lead to over-sulfonylation or side reactions. The compatibility of the system with common organic solvents like trifluorotoluene and dichloromethane further facilitates easy workup procedures. By avoiding transition metals, the process inherently sidesteps the formation of metal-complexed impurities, which are notoriously difficult to remove and can act as catalysts for product decomposition during storage. This results in a cleaner crude profile, reducing the burden on downstream purification and ensuring that the final high-purity OLED material or pharmaceutical intermediate meets rigorous quality specifications with minimal effort.
How to Synthesize Sulfonyl Substituted Styrene Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural nuances to maximize yield and optical purity. The process begins with the careful selection of the chiral catalyst, where squaramide derivatives have shown particular efficacy for certain substrate classes. The reaction is heterogeneous in nature initially but becomes homogeneous as the reagents dissolve and react. It is crucial to maintain an inert atmosphere, typically using nitrogen or argon, to prevent oxidation of the sensitive intermediates. The reaction time is a critical parameter, generally requiring 70 to 100 hours to reach full conversion, reflecting the trade-off between the mildness of the conditions and the reaction kinetics. Detailed standard operating procedures regarding stoichiometry, solvent choice, and purification protocols are essential for reproducibility.
- Charge a dry reaction tube with 1-alkynyl-2-naphthol, 1-substituted cyclopropanol, a sulfur dioxide substitute (e.g., DABCO(SO2)2), and a chiral small molecule catalyst (thiourea or squaramide derivative) under an inert atmosphere.
- Add an organic solvent such as trifluorotoluene or dichloromethane and stir the mixture at room temperature (15-40°C) for approximately 70 to 100 hours to allow the nucleophilic substitution to proceed.
- Upon completion, purify the crude reaction mixture directly via column chromatography using a mixed mobile phase of petroleum ether, ethyl acetate, and dichloromethane to isolate the target axial chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this organocatalytic methodology offers tangible strategic benefits that extend beyond mere technical feasibility. The elimination of transition metal catalysts removes a significant cost center associated with both the purchase of precious metals like palladium or rhodium and the subsequent validation of their removal. This shift translates directly into substantial cost savings in the overall manufacturing budget. Furthermore, the reliance on abundant and inexpensive sulfur dioxide surrogates, rather than specialized sulfinate salts, stabilizes the raw material supply chain against market volatility. The simplicity of the reaction setup, which requires no specialized high-pressure equipment or cryogenic cooling, allows for deployment in standard glass-lined reactors, enhancing the flexibility of existing manufacturing assets.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the removal of expensive catalytic systems and the simplification of the synthetic sequence. By avoiding the pre-preparation of sulfinate salts and eliminating the need for heavy metal scavengers, the process significantly reduces the number of processing steps and the consumption of auxiliary materials. This streamlined workflow leads to lower labor costs and reduced waste disposal fees, contributing to a leaner and more competitive cost structure. Additionally, the high atom economy of the three-component coupling ensures that a greater proportion of raw materials end up in the final product, maximizing resource efficiency and minimizing the environmental levy associated with chemical waste.
- Enhanced Supply Chain Reliability: From a logistics perspective, the use of stable, solid sulfur dioxide surrogates like DABCO(SO2)2 greatly enhances supply chain resilience. Unlike gaseous sulfur dioxide, which requires specialized containment and transport infrastructure, these solid adducts can be shipped and stored using standard chemical handling protocols. This reduces the risk of supply disruptions due to regulatory changes in hazardous material transport. Moreover, the broad substrate scope of the reaction means that a single manufacturing line can be adapted to produce a diverse library of chiral intermediates by simply swapping the alkyne or cyclopropanol starting materials. This versatility allows suppliers to respond rapidly to changing customer demands without the need for extensive retooling or new equipment investments.
- Scalability and Environmental Compliance: Scaling this process from gram to ton quantities is facilitated by the mild thermal profile and the absence of exothermic hazards often associated with metal-catalyzed oxidations. The reaction can be safely conducted at ambient temperatures, reducing energy consumption for heating or cooling and lowering the carbon footprint of the manufacturing operation. The metal-free nature of the synthesis also simplifies regulatory compliance, particularly for pharmaceutical applications where strict limits on elemental impurities are enforced. By generating a cleaner product profile with fewer toxic byproducts, the process eases the burden on wastewater treatment facilities and aligns with increasingly stringent global environmental regulations, ensuring long-term operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is crucial for R&D teams evaluating the feasibility of adopting this method for their specific pipeline projects.
Q: What are the advantages of using sulfur dioxide surrogates in this synthesis?
A: Using solid sulfur dioxide surrogates like DABCO(SO2)2 avoids the handling hazards of gaseous SO2 and eliminates the need for pre-preparing unstable sulfinate salts, streamlining the workflow and improving safety.
Q: How does this metal-free method compare to transition metal catalysis?
A: This organocatalytic approach eliminates the need for expensive transition metals, thereby removing the costly and complex heavy metal removal steps required for pharmaceutical grade intermediates while maintaining high enantioselectivity.
Q: What levels of enantiomeric excess (ee) can be achieved?
A: The patented method demonstrates excellent stereocontrol, achieving enantiomeric excess (ee) values of up to 98% across a broad range of substrates, ensuring high optical purity for downstream applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfonyl Substituted Styrene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the organocatalytic synthesis described in CN113429323A for the production of high-value chiral intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of metal-free synthesis, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards. We understand that consistency and quality are paramount in the pharmaceutical supply chain, and our dedicated technical team is committed to delivering products that exceed expectations.
We invite you to collaborate with us to leverage this advanced technology for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate your time-to-market with our reliable sulfonyl substituted styrene supplier capabilities.
