Advanced Scalable Production of 4-Hydroxy-N,N,2-Trimethylbenzimidazole-6-Carboxamide for Global Pharma Supply Chains
Introduction to Next-Generation Tegoprazan Intermediate Manufacturing
The pharmaceutical industry's relentless pursuit of potent proton pump inhibitors has placed significant demand on the supply chain for high-quality intermediates, specifically 4-hydroxy-N,N,2-trimethylbenzimidazole-6-carboxamide, a critical precursor for tegoprazan. Patent CN115536593A introduces a transformative scalable production method that addresses the longstanding bottlenecks of low yield, complex purification, and hazardous reagent usage associated with legacy synthesis pathways. By fundamentally redesigning the synthetic trajectory to start from inexpensive 3-nitro-4-aminobenzoic acid, this innovation bypasses the need for繁琐的 orthogonal protection strategies that have historically plagued the manufacturing of this benzimidazole derivative. The disclosed technology achieves a chemical purity of ≥99% through a series of robust, controllable reactions that prioritize atom economy and operational safety, marking a significant leap forward in process chemistry. For global procurement leaders, this represents a pivotal opportunity to secure a more resilient and cost-effective supply source for this high-value pharmaceutical building block.
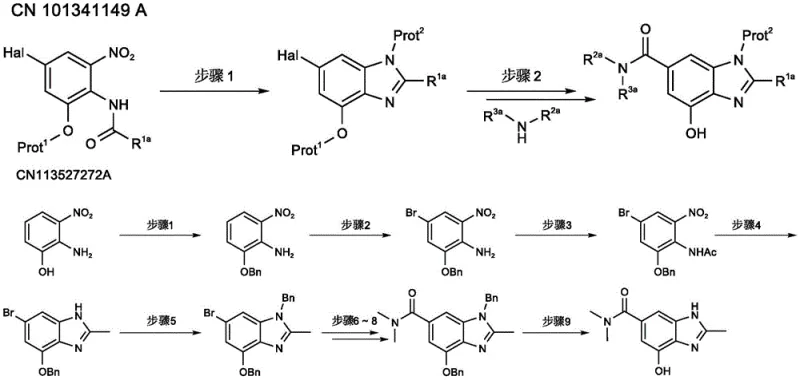
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 4-hydroxy-N,N,2-trimethylbenzimidazole-6-carboxamide has been hindered by a reliance on expensive starting materials such as 2-amino-3-nitro-5-bromophenol, which immediately inflates the raw material cost basis. As illustrated in the prior art pathways, the conventional route necessitates a cumbersome sequence of orthogonal protection steps, requiring sequential O-benzyl and N-acetyl protection to manage the reactivity of the phenolic and amino groups during the construction of the heterocyclic core. Furthermore, the traditional introduction of the carboxyl functionality involves a hazardous transformation of a bromo group to a cyano group using transition metal catalysis, followed by hydrolysis, which introduces significant safety risks and waste disposal challenges due to cyanide handling. The post-treatment difficulties are exacerbated by the use of iron powder and acetic acid systems for nitro reduction, which generate substantial amounts of solid waste and complicate the isolation of the intermediate product. Additionally, the final deprotection steps often require rigorous purification protocols, including column chromatography, to remove stubborn impurities, thereby drastically reducing the overall throughput and scalability of the process.
The Novel Approach
In stark contrast, the novel methodology detailed in the patent leverages a strategic retrosynthetic disconnection that initiates with the readily available and cost-efficient 3-nitro-4-aminobenzoic acid, effectively eliminating the need for initial phenolic protection. This innovative route streamlines the formation of the benzimidazole skeleton by combining the reduction of the nitro group and the cyclization reaction into a single, efficient operation mediated by reduced iron powder in glacial acetic acid, significantly shortening the reaction timeline. Instead of employing dangerous cyanation chemistry, the new process utilizes a sophisticated Miyaura borylation followed by an oxidative rearrangement to install the crucial hydroxyl group, a transformation that offers superior selectivity and milder reaction conditions. The elimination of the N-acetyl protection step further simplifies the workflow, allowing for a direct progression from the brominated amide to the cyclized benzimidazole core without intermediate isolation losses. This holistic redesign not only enhances the overall yield but also ensures that the final product can be isolated through simple crystallization and filtration, making it ideally suited for continuous manufacturing environments.
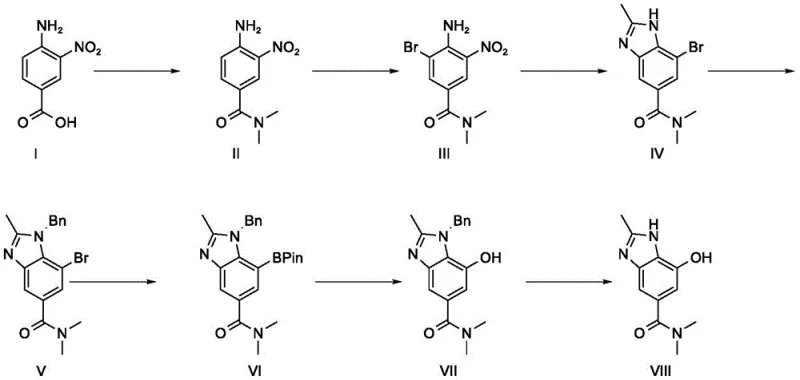
Mechanistic Insights into Reductive Cyclization and Oxidative Rearrangement
The core of this technological breakthrough lies in the precise control of the reductive cyclization mechanism, where the nitro group of the brominated amide intermediate is selectively reduced to an amine in situ, which immediately attacks the adjacent amide carbonyl to close the benzimidazole ring. By maintaining the reaction temperature strictly between 100-110°C in glacial acetic acid, the process ensures complete conversion while minimizing the formation of over-reduced byproducts or polymeric tars that often contaminate iron-mediated reductions. The subsequent Miyaura borylation step utilizes a palladium catalyst system, specifically tris(dibenzylideneacetone)dipalladium, to effectuate the substitution of the bromine atom with a pinacol boronate ester, a transformation that is critical for the downstream installation of the hydroxyl moiety. This organometallic step is meticulously optimized to prevent protodeboronation, ensuring high fidelity in the transfer of the boron group which serves as a masked oxygen equivalent. The final oxidative rearrangement employs hydrogen peroxide under basic conditions to convert the C-B bond into a C-O bond via a 1,2-migration mechanism, a strategy that is far more atom-economical than traditional nucleophilic aromatic substitution routes. This sequence demonstrates a profound understanding of functional group tolerance, allowing the benzimidazole nitrogen to remain intact throughout the harsh oxidative conditions required for phenol formation.
Impurity control is rigorously managed through temperature regulation and reagent selection, particularly during the bromination stage where maintaining the reaction mixture at 0-10°C is paramount to preventing polybromination and oxidative degradation of the electron-rich aromatic ring. The choice of 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (EDC·HCl) as the condensing agent in the initial amidation step is a strategic decision to avoid the formation of insoluble urea byproducts associated with DCC, which would otherwise necessitate difficult filtration or chromatographic separation. Comparative data indicates that deviating from the optimal temperature range during bromination can lead to a precipitous drop in yield from 88% to 67%, highlighting the sensitivity of the electrophilic aromatic substitution to thermal energy. Furthermore, the final hydrogenolysis step utilizes trifluoroacetic acid as an additive to facilitate the cleavage of the benzyl protecting group, preventing the formation of intractable salts that could trap the product in the aqueous phase during workup. These mechanistic refinements collectively ensure that the impurity profile remains well within the stringent specifications required for GMP-grade pharmaceutical intermediates, reducing the burden on downstream quality control laboratories.
How to Synthesize 4-Hydroxy-N,N,2-Trimethylbenzimidazole-6-Carboxamide Efficiently
The execution of this synthesis requires strict adherence to the defined operational parameters to maximize yield and purity, beginning with the activation of the carboxylic acid using EDC·HCl in dichloromethane at room temperature for 4 to 6 hours. Following the isolation of the amide, the bromination must be conducted with careful thermal management, quenching excess bromine with sodium thiosulfate to ensure a safe and clean reaction mixture before proceeding to the reductive cyclization. The cyclization step demands the use of reduced iron powder at elevated temperatures, followed by a hot filtration to remove iron sludge, a critical unit operation that prevents contamination of the final product with heavy metals.
- Perform dehydration condensation of 3-nitro-4-aminobenzoic acid with dimethylamine hydrochloride using EDC·HCl to form the amide intermediate.
- Execute controlled bromination under acidic conditions at 0-10°C followed by iron-mediated reductive cyclization to construct the benzimidazole core.
- Complete the synthesis via N-benzylation, Miyaura borylation, oxidative rearrangement to install the hydroxyl group, and final catalytic hydrogenolysis.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers substantial advantages that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers seeking reliable partners. By shifting the starting material to a commodity chemical, the volatility associated with sourcing specialized nitro-bromophenols is eliminated, stabilizing the cost structure and ensuring long-term availability of raw materials. The removal of column chromatography from the purification train is a game-changer for scale-up, as it drastically reduces solvent consumption, waste generation, and processing time, translating into significant operational expenditure savings. Moreover, the avoidance of cyanide reagents mitigates regulatory compliance burdens and insurance costs related to hazardous material handling, making the facility more sustainable and easier to permit in diverse global jurisdictions. The robust nature of the crystallization-based purifications ensures that batch-to-batch consistency is maintained even at multi-ton scales, reducing the risk of costly batch failures or rejections.
- Cost Reduction in Manufacturing: The strategic substitution of expensive protected starting materials with 3-nitro-4-aminobenzoic acid fundamentally lowers the material cost basis, while the elimination of silica gel chromatography removes a major cost driver associated with solvent recovery and stationary phase disposal. The use of EDC·HCl instead of DCC prevents the formation of dicyclohexylurea, a byproduct that is notoriously difficult to remove and often leads to product loss during purification, thereby improving the effective yield per kilogram of input. Additionally, the one-pot reduction and cyclization strategy consolidates two distinct unit operations into a single reactor run, saving on labor, energy, and equipment occupancy time. These cumulative efficiencies result in a drastically simplified cost model that allows for competitive pricing without compromising on margin.
- Enhanced Supply Chain Reliability: Sourcing stability is significantly improved as the new route relies on bulk chemicals that are produced by multiple vendors globally, reducing the risk of supply disruption due to single-source dependency. The simplified workflow, characterized by fewer isolation steps and standard filtration techniques, reduces the overall lead time required to manufacture a batch, enabling faster response to fluctuating market demand. The high atom economy of the borylation-oxidation sequence minimizes the volume of chemical waste generated, aligning with modern green chemistry principles and ensuring uninterrupted production even under tightening environmental regulations. This reliability makes the supplier a more attractive partner for long-term contracts and just-in-time delivery models essential for modern pharmaceutical supply chains.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing heterogeneous catalysts like palladium on carbon that can be easily filtered and recycled, minimizing heavy metal contamination in the final API. The replacement of hazardous cyanation chemistry with a benign oxidative rearrangement significantly lowers the environmental footprint and safety risk profile of the manufacturing site. Operational safety is further enhanced by the use of stable reagents like iron powder and hydrogen peroxide, which are easier to handle and store compared to pyrophoric or highly toxic alternatives. This alignment with environmental, social, and governance (ESG) goals ensures that the production facility remains compliant with international standards, securing its position as a sustainable source for critical healthcare ingredients.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and quality assurance of this advanced synthetic route, providing clarity for potential partners evaluating the technology. These answers are derived directly from the experimental data and comparative examples provided in the patent documentation, ensuring accuracy and relevance to industrial application. Understanding these nuances is critical for R&D teams assessing the feasibility of technology transfer and for quality assurance personnel defining specification limits.
Q: How does this new route improve purity compared to conventional methods?
A: The novel process eliminates the need for silica gel column chromatography by selecting condensing agents like EDC·HCl that generate water-soluble byproducts, ensuring chemical purity ≥99% through simple crystallization and filtration.
Q: What are the safety advantages regarding reagent handling in this synthesis?
A: This method avoids the use of hazardous cyanide reagents required in traditional routes for carboxyl introduction, replacing them with a safer Miyaura borylation and oxidation sequence that significantly reduces operational risk.
Q: Is the process suitable for large-scale commercial manufacturing?
A: Yes, the route utilizes cheap and readily available starting materials like 3-nitro-4-aminobenzoic acid and employs robust unit operations such as filtration and crystallization, making it highly adaptable for ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Hydroxy-N,N,2-Trimethylbenzimidazole-6-Carboxamide Supplier
At NINGBO INNO PHARMCHEM, we leverage our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring this cutting-edge synthesis to life for our global clientele. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 4-hydroxy-N,N,2-trimethylbenzimidazole-6-carboxamide meets the exacting standards required for proton pump inhibitor development. We understand that the transition from bench-scale innovation to industrial reality requires a partner who can navigate the complexities of process safety, waste management, and regulatory compliance with equal proficiency. Our team is dedicated to optimizing this novel route further, continuously seeking opportunities to enhance yield and reduce environmental impact through green chemistry initiatives.
We invite you to engage with our technical procurement team to discuss how this optimized manufacturing process can drive value for your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this superior synthetic pathway. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your supply chain requirements, ensuring a seamless integration of this high-quality intermediate into your production pipeline.
