Advanced Photocatalytic Synthesis of 1-Methyl-3-Alkyl Quinoxalinones for Commercial Scale-Up
Introduction to Patent CN111892545A: A Breakthrough in Green Chemistry
The pharmaceutical industry is constantly seeking more sustainable and efficient pathways to synthesize complex heterocyclic scaffolds, and the recent disclosure in patent CN111892545A represents a significant leap forward in the production of 1-methyl-3-alkyl quinoxalinone compounds. This intellectual property details a novel, environmentally benign synthesis method that utilizes visible light photocatalysis to achieve direct C-H functionalization at the C-3 position of the quinoxalinone core. Unlike traditional thermal methods that often require harsh conditions and stoichiometric oxidants, this innovative approach leverages the intrinsic photosensitivity of the substrate itself in a hexafluoroisopropanol (HFIP) medium. By employing a specific wavelength of violet light (380-385 nm), the process facilitates a radical decarboxylative coupling with alkyl peroxides, resulting in high-purity products with exceptional yields. For procurement managers and R&D directors alike, this technology offers a compelling value proposition by drastically simplifying the synthetic route while adhering to strict green chemistry principles, positioning it as a vital asset for the reliable supply of high-purity pharmaceutical intermediates.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of quinoxalin-2(1H)-ones at the C-3 position has been plagued by significant inefficiencies and operational hazards that hinder large-scale manufacturing. Prior art methods, such as those utilizing tert-butyl hydroperoxide (TBHP) in conjunction with iodine catalysts and sulfite reducing agents, necessitate elevated temperatures and extended reaction times, often leading to energy-intensive processes with suboptimal yields capped at roughly 74%. Furthermore, these thermal radical pathways frequently suffer from poor substrate scope, typically restricting modifications to simple methylation rather than allowing for diverse alkyl chain introductions. Another reported method involving TBHP in acetic acid requires heating for up to 24 hours, which not only increases utility costs but also raises safety concerns regarding the stability of peroxides under prolonged thermal stress. These conventional techniques often generate substantial chemical waste due to the need for stoichiometric oxidants and metal catalysts, creating a heavy burden on downstream purification and waste treatment facilities, thereby inflating the overall cost of goods sold for the final active pharmaceutical ingredient.
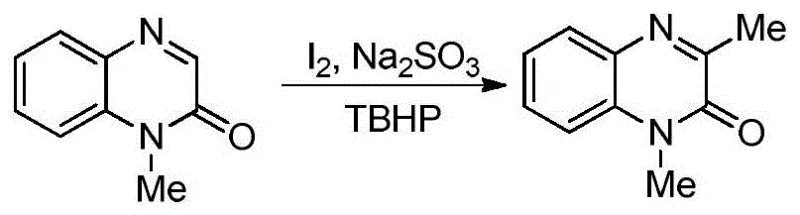
The Novel Approach
In stark contrast to these legacy technologies, the method described in CN111892545A introduces a paradigm shift by utilizing alkyl peroxides as versatile alkyl sources under mild, metal-free photocatalytic conditions. This new strategy operates at room temperature using low-energy 15W LED light sources, effectively eliminating the thermal degradation risks associated with peroxide handling and significantly reducing the carbon footprint of the manufacturing process. The reaction demonstrates remarkable versatility, successfully accommodating a wide range of alkyl groups including benzyl, methyl, and cyclohexyl moieties with yields reaching as high as 91%, far surpassing the limitations of previous methylation-only protocols. By removing the dependency on transition metal catalysts and external photosensitizers, the process streamlines the workflow, reduces raw material costs, and simplifies the impurity profile, making it an ideal candidate for cost reduction in API manufacturing where regulatory compliance regarding residual metals is stringent. This technological advancement ensures a more robust and scalable supply chain for complex quinoxalinone derivatives essential for oncology and antimicrobial drug development.
Mechanistic Insights into Visible-Light Induced Decarboxylative Coupling
For R&D teams evaluating the feasibility of this technology, understanding the underlying mechanistic pathway is crucial for troubleshooting and optimization. The reaction proceeds through a unique self-sensitized photocatalytic cycle where the 1-methylquinoxalinone substrate absorbs photons in the 380-385 nm range to reach an excited state, acting as its own photocatalyst without the need for additives like Ruthenium or Iridium complexes. Upon excitation, the substrate engages in a single-electron transfer (SET) with the alkyl peroxide reagent, triggering the homolytic cleavage of the weak O-O bond to generate an acyloxy radical which rapidly undergoes decarboxylation to form a reactive alkyl radical species. This carbon-centered radical then adds selectively to the C-3 position of the quinoxalinone ring, forming a nitrogen-centered radical intermediate that is subsequently oxidized by the radical cation of the substrate to restore aromaticity. The elegance of this mechanism lies in its atom economy and the regeneration of the ground-state catalyst, ensuring a continuous catalytic turnover that drives the reaction to completion with high selectivity and minimal byproduct formation.
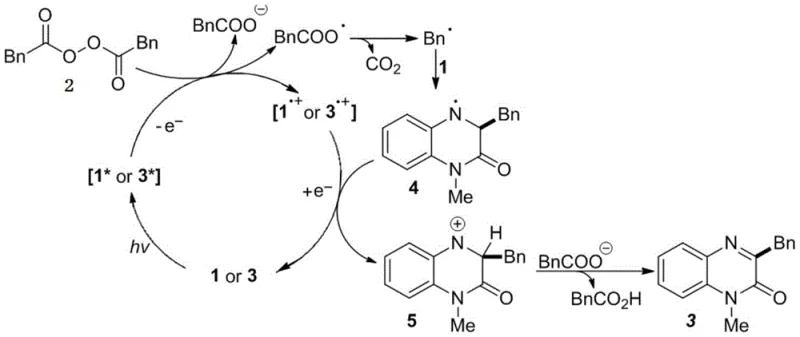
Furthermore, the choice of hexafluoroisopropanol (HFIP) as the solvent plays a pivotal role in stabilizing the charged intermediates and radical species involved in this transformation. HFIP is known for its strong hydrogen-bond donating ability and low nucleophilicity, which helps to stabilize the radical cation intermediates and facilitate the proton-coupled electron transfer steps necessary for the final aromatization. This specific solvent interaction prevents side reactions such as over-oxidation or polymerization of the radical species, which are common pitfalls in less optimized solvent systems like acetonitrile or dichloromethane where yields drop precipitously. The mechanistic robustness of this system allows for excellent functional group tolerance, meaning that sensitive moieties on the alkyl peroxide or the quinoxalinone ring remain intact during the reaction. This level of control over the reaction pathway is essential for producing high-purity pharmaceutical intermediates that meet the rigorous quality standards required for clinical trial materials and commercial drug substances.
How to Synthesize 1-Methyl-3-Alkyl Quinoxalinones Efficiently
Implementing this photocatalytic protocol in a laboratory or pilot plant setting requires precise control over light intensity and solvent quality to maximize the reaction efficiency. The standard procedure involves dissolving the 1-methylquinoxalinone starting material and the chosen alkyl peroxide in HFIP at a concentration between 0.1 mol/L and 0.5 mol/L to ensure optimal light penetration and mixing. The reaction mixture is then subjected to irradiation from a 15W LED source tuned specifically to the 380-385 nm wavelength band, as deviations outside this narrow window significantly reduce the quantum efficiency of the excitation process. Monitoring the reaction progress via thin-layer chromatography typically indicates completion within 2 hours, after which the solvent is removed under reduced pressure and the product is isolated via standard silica gel chromatography. Detailed standardized synthesis steps follow below to guide your technical team in replicating these results.
- Prepare the reaction mixture by dissolving 1-methylquinoxalinone and an alkyl peroxide compound (such as benzyl peroxide) in hexafluoroisopropanol (HFIP) solvent within a quartz reaction vessel.
- Irradiate the solution with a 15W LED light source emitting violet light in the 380-385 nm wavelength range while stirring at room temperature for approximately 2 hours.
- Upon completion, concentrate the reaction mixture using a rotary evaporator and purify the crude product via silica gel column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, adopting this photocatalytic methodology offers profound advantages that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. The elimination of expensive transition metal catalysts such as palladium, copper, or rhodium removes a major cost driver from the bill of materials, while simultaneously eradicating the need for complex and costly metal scavenging steps during downstream processing. This simplification of the purification train not only reduces the consumption of silica and solvents but also shortens the overall production cycle time, allowing for faster turnaround on custom synthesis orders and improved responsiveness to market demand fluctuations. Additionally, the use of commercially available and inexpensive alkyl peroxides as alkylating agents ensures a stable and diversified supply base, mitigating the risk of raw material shortages that often plague specialized reagent markets. The ambient temperature operation further contributes to substantial cost savings by lowering energy consumption for heating and cooling, aligning perfectly with corporate sustainability goals and reducing the environmental compliance burden associated with hazardous waste disposal.
- Cost Reduction in Manufacturing: The absence of precious metal catalysts and external photosensitizers dramatically lowers the raw material costs, while the simplified workup procedure reduces labor and consumable expenses associated with purification. By avoiding high-temperature heating and long reaction times, the process significantly cuts down on utility costs, making the production of these valuable intermediates much more economically viable compared to thermal radical methods. The high yields achieved (often exceeding 85%) mean less starting material is wasted, directly improving the mass balance and overall process efficiency which translates to a lower cost per kilogram of the final product.
- Enhanced Supply Chain Reliability: Utilizing commodity chemicals like alkyl peroxides and standard LED lighting equipment ensures that the supply chain is not dependent on single-source suppliers for exotic reagents or specialized hardware. The robustness of the reaction conditions allows for flexible manufacturing scheduling, as the process is less sensitive to minor variations in temperature or pressure compared to high-pressure hydrogenation or cryogenic reactions. This reliability ensures consistent delivery timelines for clients, reducing the risk of production delays that can impact the launch of downstream drug products and strengthening the partnership between the supplier and the pharmaceutical innovator.
- Scalability and Environmental Compliance: The metal-free nature of this synthesis inherently produces a cleaner impurity profile, facilitating easier regulatory approval and reducing the environmental impact of effluent streams. Scaling this photochemical process is straightforward using modern flow chemistry reactors or large-scale batch photoreactors, allowing for seamless transition from gram-scale R&D to multi-ton commercial production without re-optimizing the core chemistry. The use of HFIP, while a specialty solvent, can be efficiently recovered and recycled, further minimizing waste generation and supporting a circular economy approach to chemical manufacturing that meets increasingly strict global environmental regulations.
Frequently Asked Questions (FAQ)
To address common technical inquiries regarding the implementation and scope of this patented technology, we have compiled the following answers based on the experimental data and mechanistic studies provided in the patent literature. These insights are designed to help technical decision-makers evaluate the fit of this synthesis route for their specific project needs and understand the operational parameters required for success. Understanding these nuances is key to leveraging the full potential of this green chemistry innovation for your supply chain.
Q: Why is hexafluoroisopropanol (HFIP) critical for this photocatalytic reaction?
A: HFIP acts as a unique solvent that stabilizes radical intermediates and enhances the photosensitivity of the quinoxalinone substrate, enabling the reaction to proceed with high efficiency (up to 86% yield) compared to common solvents like acetonitrile or DMF which result in negligible conversion.
Q: Does this method require expensive transition metal catalysts?
A: No, this patented process is entirely metal-free. The 1-methylquinoxalinone substrate itself acts as the photosensitizer under 380-385 nm irradiation, eliminating the need for costly external photocatalysts or transition metals like palladium or copper.
Q: What is the substrate scope for the alkyl group introduction?
A: The method demonstrates excellent functional group compatibility, successfully introducing various C1-C10 alkyl groups including methyl, benzyl, and cyclohexyl groups with high selectivity and yields ranging from 85% to 91%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Methyl-3-Alkyl Quinoxalinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this photocatalytic technology and possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring such innovations to the global market. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of maintaining stringent purity specifications required for GMP-grade pharmaceutical intermediates. We are committed to delivering high-quality 1-methyl-3-alkyl quinoxalinone derivatives that adhere to the highest standards of safety and efficacy, ensuring that your drug development pipeline remains uninterrupted by supply bottlenecks or quality issues. Our team of expert chemists is ready to assist you in optimizing this route for your specific analogues, guaranteeing a seamless transition from benchtop discovery to industrial manufacturing.
We invite you to contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and project timelines. By partnering with us, you gain access to specific COA data and route feasibility assessments that will empower you to make informed decisions about your sourcing strategy. Let us help you leverage this cutting-edge synthesis method to accelerate your time-to-market and achieve your commercial objectives with confidence and reliability.
