Revolutionizing Aryl Ester Production with Robust Palladium-Catalyzed Direct C-O Coupling Technology
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct carbon-heteroatom bonds, particularly for the synthesis of complex ester motifs found in numerous bioactive molecules. Patent CN110683926B introduces a significant technological advancement in this domain by disclosing a novel preparation method for carboxylic acid aryl ester compounds. This innovation leverages a sophisticated dual-catalyst system comprising an organopalladium complex and a silver salt to facilitate direct selective C-O coupling between carboxylic acids and halogenated aromatic hydrocarbons. Unlike traditional esterification methods that often require harsh conditions or multiple activation steps, this patented approach operates under relatively mild inert gas atmospheres while achieving exceptional yields. The technology addresses critical bottlenecks in synthetic organic chemistry by expanding the scope of compatible substrates to include a wide variety of steric and electronic environments. For R&D directors and process chemists, this represents a pivotal shift towards more atom-economical and streamlined synthesis strategies for high-value intermediates.
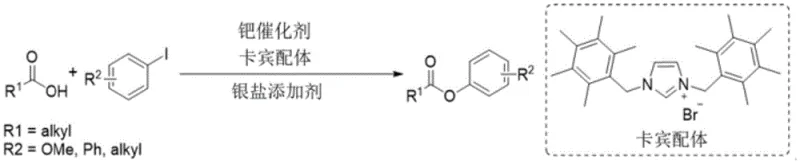
The limitations of conventional methods for forming aryl esters have long hindered the rapid development of new drug candidates and agrochemical agents. Historically, methods such as those reported by Professor Itomi's group relied on electron-rich dibenzyl azacarbene ligands which severely restricted the substrate scope to specific methoxy or alkyl-substituted iodobenzenes and bulky aliphatic carboxylic acids. Furthermore, recent photocatalytic approaches, while innovative, often struggle with compatibility issues regarding electron-donating coupling reagents, limiting their utility in diverse synthetic libraries. These existing technologies frequently suffer from unsatisfactory reaction yields and narrow applicability, forcing chemists to resort to multi-step sequences that increase waste and cost. The novel approach described in the patent overcomes these deficiencies by utilizing a robust organopalladium complex formed from palladium compounds and rigid nitrogen-containing ligands such as o-phenanthroline or bipyridine derivatives. This catalytic system effectively promotes the direct bonding of the acyl oxygen bond of the carboxylic acid to the halogen atom of the aryl halide, bypassing the need for pre-functionalization.
Mechanistic Insights into Pd-Catalyzed Direct C-O Coupling
The core of this technological breakthrough lies in the precise engineering of the catalytic cycle, which relies on the synergistic interaction between the palladium center and the specialized ligand environment. The organopalladium complex is generated in situ from palladium precursors such as palladium acetate or palladium trifluoroacetate combined with electron-rich ligands like 3,4,7,8-tetramethyl-1,10-phenanthroline. These rigid nitrogen ligands play a critical role in stabilizing high-valent palladium intermediates that are otherwise prone to decomposition, thereby ensuring the longevity and turnover number of the catalyst. The presence of a silver salt acts as a crucial halogen scavenger, driving the equilibrium forward by removing halide ions generated during the oxidative addition step. This mechanism allows for the activation of the C-X bond in the aryl halide followed by coordination with the carboxylate anion, ultimately leading to reductive elimination to form the desired ester product. The careful optimization of the molar ratio between the palladium compound and the nitrogen ligand, typically maintained at 1:2, ensures maximum catalytic activity and minimizes the formation of inactive metal aggregates.
Impurity control is another vital aspect of this mechanism, particularly for pharmaceutical applications where strict purity specifications are mandatory. The use of specific solvents such as trifluorotoluene or toluene provides an inert medium that dissolves all reactants effectively while preventing side reactions that could lead to complex impurity profiles. The reaction temperature, optimized between 100°C and 150°C, is high enough to overcome the activation energy barrier for the C-O bond formation but controlled enough to prevent thermal degradation of sensitive functional groups on the substrate. By maintaining an inert gas atmosphere using argon or nitrogen, the system prevents the oxidation of the low-valent palladium species which could otherwise lead to catalyst deactivation and the formation of palladium black. This rigorous control over reaction parameters results in a clean reaction profile with yields reaching as high as 95%, significantly reducing the burden on downstream purification processes.
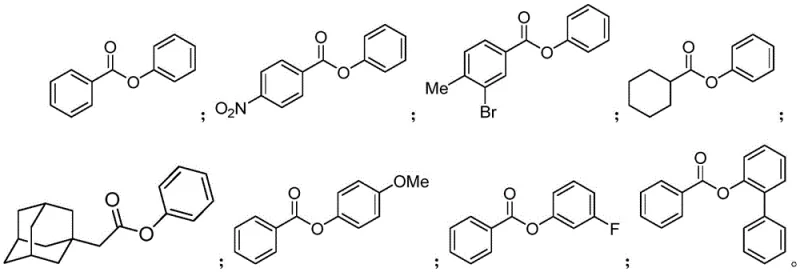
How to Synthesize Carboxylic Acid Aryl Esters Efficiently
The synthesis protocol outlined in the patent provides a standardized framework for producing high-purity aryl esters suitable for immediate application in drug discovery pipelines. The process begins with the precise weighing and mixing of the carboxylic acid substrate, the aryl halide coupling partner, the palladium catalyst system, and the silver salt additive in a dry reaction vessel. It is imperative to ensure that the reaction environment is thoroughly purged with inert gas to exclude moisture and oxygen, which are detrimental to the catalytic cycle. The reaction mixture is then heated to the specified temperature range for a duration sufficient to achieve full conversion, typically monitored by TLC or HPLC analysis. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining carboxylic acid, aryl halide, palladium catalyst precursor, nitrogen-containing ligand, and silver salt in a suitable solvent.
- Ensure the system is under an inert gas atmosphere such as argon or nitrogen to prevent oxidation of sensitive intermediates.
- Heat the reaction mixture to temperatures between 100-150°C for 8 to 20 hours to achieve high conversion yields up to 95%.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers substantial strategic benefits by simplifying the sourcing of raw materials and reducing overall manufacturing complexity. The ability to utilize a broad range of commercially available carboxylic acids and aryl halides means that supply chain managers are not locked into proprietary or scarce starting materials, thereby enhancing supply continuity. The high efficiency of the reaction translates directly into reduced raw material consumption per unit of product, which is a key driver for cost optimization in large-scale manufacturing. Furthermore, the robustness of the catalyst system reduces the risk of batch failures, ensuring consistent delivery schedules to downstream customers who rely on just-in-time inventory models. This reliability is paramount for maintaining the integrity of global pharmaceutical supply chains where delays can have cascading effects on clinical trial timelines.
- Cost Reduction in Manufacturing: The elimination of multiple synthetic steps required in traditional esterification protocols leads to significant operational savings and reduced waste generation. By achieving direct coupling, the process avoids the costs associated with activating agents, protecting group manipulations, and extensive purification procedures typically needed to remove byproducts. The high yield of up to 95% ensures that the maximum amount of valuable starting material is converted into the desired product, minimizing the financial loss associated with unreacted feedstock. Additionally, the use of earth-abundant solvents and recyclable catalyst components further contributes to a lower total cost of ownership for the manufacturing process.
- Enhanced Supply Chain Reliability: The versatility of the substrate scope allows manufacturers to pivot quickly between different ester variants without needing to retool entire production lines or source exotic reagents. This flexibility is crucial for responding to fluctuating market demands and ensuring that critical intermediates are always available for API synthesis. The reliance on stable and well-characterized chemical inputs reduces the risk of supply disruptions caused by the volatility of niche chemical markets. Consequently, procurement teams can negotiate better terms with suppliers due to the standardized nature of the required inputs and the predictability of the consumption rates.
- Scalability and Environmental Compliance: The reaction conditions are amenable to scale-up from laboratory benchtop to multi-ton commercial production without compromising safety or efficiency. The use of inert atmospheres and controlled heating profiles aligns with modern safety standards, reducing the risk of exothermic runaways common in less controlled esterification methods. Moreover, the high atom economy of the direct coupling reaction aligns with green chemistry principles, resulting in lower E-factors and reduced environmental impact. This compliance with environmental regulations facilitates smoother regulatory approvals and enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Pd-catalyzed C-O coupling technology in industrial settings. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for their specific production needs. The information covers catalyst loading, substrate compatibility, and operational parameters critical for successful execution.
Q: What represents the primary advantage of this Pd-catalyzed method over traditional esterification?
A: This method enables direct C-O coupling between carboxylic acids and aryl halides without requiring pre-activation of the acid, offering broader substrate compatibility and higher yields compared to conventional methods.
Q: Which ligands are critical for stabilizing the palladium catalyst in this process?
A: Rigid nitrogen-containing ligands, specifically o-phenanthroline derivatives like 4,7-dimethoxy-1,10-phenanthroline, are essential for stabilizing high-valent palladium intermediates.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the use of robust reaction conditions, readily available starting materials, and high yields supports effective commercial scale-up for complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carboxylic Acid Aryl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN110683926B for accelerating the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries are seamlessly translated into reliable commercial supplies. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of carboxylic acid aryl ester meets the exacting standards required by global pharmaceutical regulators. Our commitment to quality and consistency makes us a trusted partner for companies seeking to secure their supply chains for critical intermediates.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this more efficient manufacturing process. We encourage potential partners to contact us for specific COA data and route feasibility assessments to validate the suitability of this technology for your target molecules. Let us collaborate to drive down costs and enhance the efficiency of your chemical supply chain today.
