Advanced Asymmetric Synthesis of 3,3-Disubstituted-2-Oxindoles for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing complex heterocyclic scaffolds that serve as the backbone for potent bioactive molecules. Patent CN102659494A introduces a groundbreaking asymmetric synthesis method for 3,3-disubstituted-2-oxindole compounds, a privileged structure found in numerous natural products and therapeutic agents. This innovation addresses the critical need for efficient access to chiral oxindoles, which exhibit diverse biological activities ranging from antioxidant and anticancer properties to neuroprotective effects. The core technology leverages a chiral organic base catalyst to facilitate the reaction between 3-monosubstituted-2-oxindoles and 1,4-naphthoquinones under mild aerobic conditions. 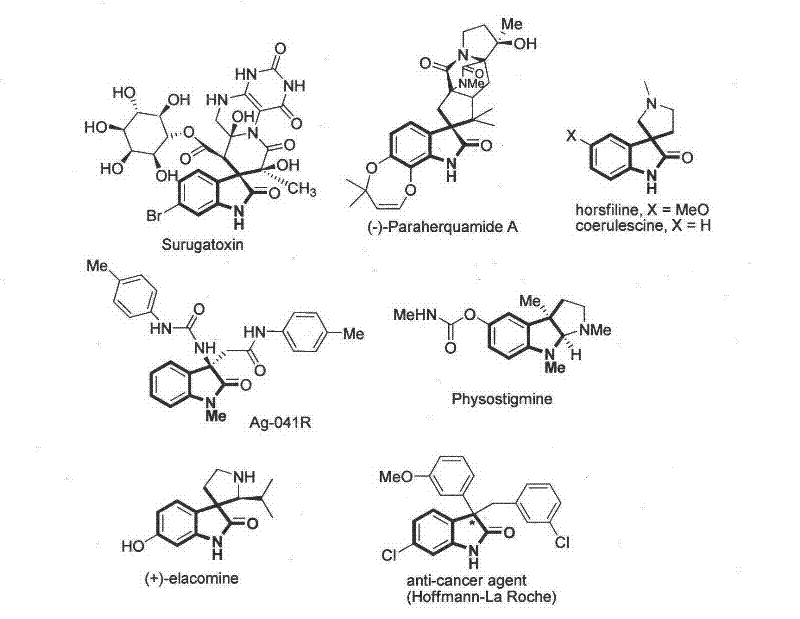 As illustrated in the structural diversity of known bioactive molecules like Surugatoxin and (-)-Paraherquamide A, the ability to precisely construct the quaternary carbon center at the C3 position is paramount for drug discovery. This patent provides a reliable pharmaceutical intermediate supplier with a versatile platform to access these high-value targets with exceptional stereochemical control.
As illustrated in the structural diversity of known bioactive molecules like Surugatoxin and (-)-Paraherquamide A, the ability to precisely construct the quaternary carbon center at the C3 position is paramount for drug discovery. This patent provides a reliable pharmaceutical intermediate supplier with a versatile platform to access these high-value targets with exceptional stereochemical control.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 3,3-disubstituted-2-oxindole core has been plagued by significant synthetic challenges, particularly regarding the introduction of diverse substituents at the quaternary C3 center. Prior art methods, such as asymmetric conjugate additions, Aldol reactions, and Morita-Baylis-Hillman reactions, have predominantly focused on forming Csp3-Csp3 bonds, effectively limiting the structural diversity to alkyl group introductions. 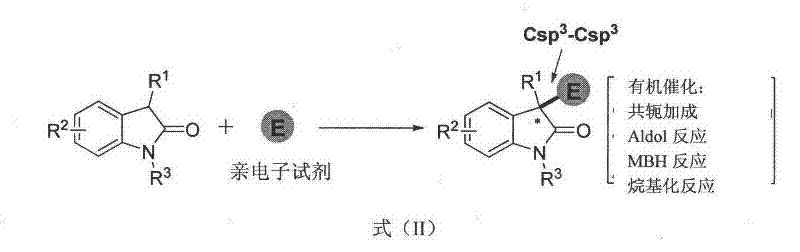 These conventional approaches often rely on harsh reaction conditions, expensive chiral phase-transfer catalysts, or sensitive organometallic reagents that complicate scale-up and increase production costs. Furthermore, the inability to efficiently introduce aryl or olefinic groups at the C3 position restricts the chemical space available for medicinal chemists, hindering the optimization of drug candidates. The reliance on transition metals also introduces the risk of heavy metal contamination, necessitating costly purification steps to meet stringent regulatory standards for active pharmaceutical ingredients.
These conventional approaches often rely on harsh reaction conditions, expensive chiral phase-transfer catalysts, or sensitive organometallic reagents that complicate scale-up and increase production costs. Furthermore, the inability to efficiently introduce aryl or olefinic groups at the C3 position restricts the chemical space available for medicinal chemists, hindering the optimization of drug candidates. The reliance on transition metals also introduces the risk of heavy metal contamination, necessitating costly purification steps to meet stringent regulatory standards for active pharmaceutical ingredients.
The Novel Approach
The present invention overcomes these historical bottlenecks by providing a novel methodology for introducing aryl or olefin groups at the C3 position, thereby constructing a Csp3-Csp2 bond with high efficiency. 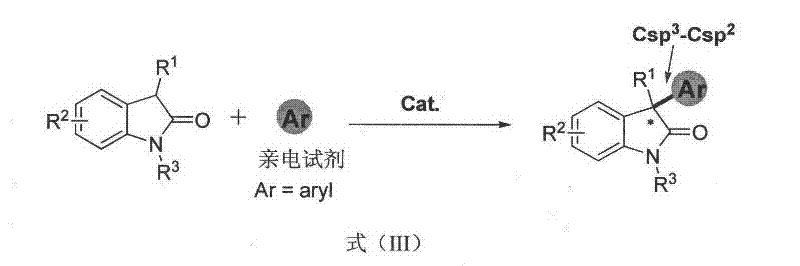 This strategic shift allows for the direct functionalization of the oxindole substrate with aromatic systems, vastly expanding the library of accessible derivatives for high-throughput screening. By utilizing a specialized chiral organic catalyst in conjunction with air oxidation, the process achieves high conversion rates without the need for stoichiometric oxidants or inert atmospheres. This approach not only simplifies the operational procedure but also aligns with green chemistry principles by using oxygen from the air as the terminal oxidant. The result is a streamlined synthesis that delivers products with very high enantiomeric excess (ee) values, providing a key skeleton structure for the synthesis of many natural products and medicines that was previously difficult to access.
This strategic shift allows for the direct functionalization of the oxindole substrate with aromatic systems, vastly expanding the library of accessible derivatives for high-throughput screening. By utilizing a specialized chiral organic catalyst in conjunction with air oxidation, the process achieves high conversion rates without the need for stoichiometric oxidants or inert atmospheres. This approach not only simplifies the operational procedure but also aligns with green chemistry principles by using oxygen from the air as the terminal oxidant. The result is a streamlined synthesis that delivers products with very high enantiomeric excess (ee) values, providing a key skeleton structure for the synthesis of many natural products and medicines that was previously difficult to access.
Mechanistic Insights into Organocatalytic Asymmetric Oxidative Coupling
The core of this technological breakthrough lies in the sophisticated interplay between the chiral organic catalyst and the substrates under aerobic conditions. The reaction mechanism involves the activation of the 3-substituted-2-oxindole by the chiral base, generating a nucleophilic species that undergoes asymmetric conjugate addition to the 1,4-naphthoquinone. 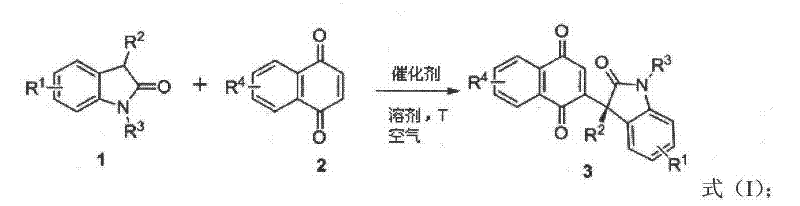 Following the initial addition, the intermediate is oxidized by molecular oxygen from the air to restore aromaticity and form the final 3,3-disubstituted product. The catalyst library, encompassing structures from Formula 1 to Formula 14, includes various chiral amines, thioureas, and cinchona alkaloid derivatives tailored to maximize stereo-induction.
Following the initial addition, the intermediate is oxidized by molecular oxygen from the air to restore aromaticity and form the final 3,3-disubstituted product. The catalyst library, encompassing structures from Formula 1 to Formula 14, includes various chiral amines, thioureas, and cinchona alkaloid derivatives tailored to maximize stereo-induction. 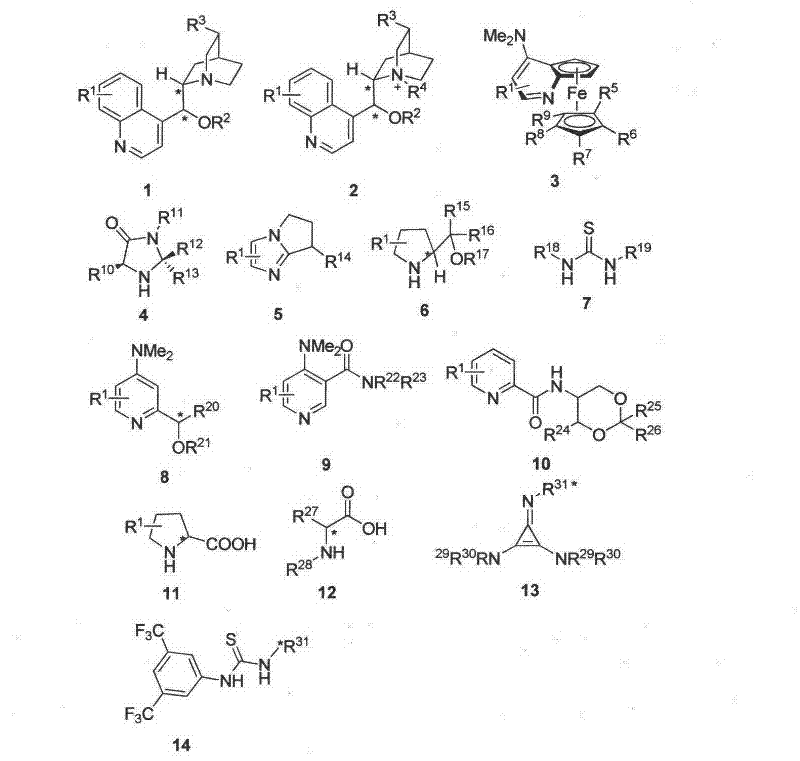 The precise tuning of the catalyst's steric and electronic properties ensures that the nucleophilic attack occurs exclusively from one face of the planar quinone, resulting in the observed high enantioselectivity. This mechanistic pathway avoids the formation of racemic byproducts and minimizes side reactions, ensuring a clean impurity profile that is essential for downstream pharmaceutical processing.
The precise tuning of the catalyst's steric and electronic properties ensures that the nucleophilic attack occurs exclusively from one face of the planar quinone, resulting in the observed high enantioselectivity. This mechanistic pathway avoids the formation of racemic byproducts and minimizes side reactions, ensuring a clean impurity profile that is essential for downstream pharmaceutical processing.
Impurity control is inherently built into this catalytic system due to the mild reaction conditions and the specificity of the organocatalyst. Unlike metal-catalyzed processes that may suffer from non-selective radical pathways or metal-mediated decomposition, this organic system operates at temperatures ranging from room temperature to -50°C, with a preferred temperature of -20°C to optimize selectivity. The use of common solvents such as chloroform, dichloromethane, or toluene further enhances the reproducibility of the reaction. The tolerance of the catalyst to air and moisture means that rigorous exclusion of environmental factors is not required, reducing the complexity of the manufacturing setup. Consequently, the crude reaction mixture typically contains fewer side products, simplifying the purification process and improving the overall yield of the high-purity pharmaceutical intermediate.
How to Synthesize 3,3-Disubstituted-2-Oxindole Efficiently
The practical implementation of this synthesis route is straightforward and amenable to standard laboratory and pilot plant equipment. The general procedure involves preparing two separate solutions: one containing the 1,4-naphthoquinone substrate dissolved in a solvent like dichloromethane, and another containing the oxindole substrate and the chiral catalyst. The quinone solution is cooled to 0°C, while the catalyst mixture is maintained at -20°C to ensure optimal kinetic control. The quinone is then added dropwise to the catalyst mixture, allowing the reaction to proceed under an air atmosphere for approximately 12 hours. 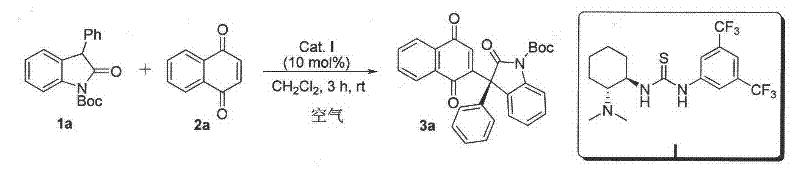 Monitoring via TLC confirms the consumption of starting materials, after which the reaction is quenched with water. The workup involves standard extraction with ethyl acetate, washing with saturated brine to remove residual salts, and drying over anhydrous magnesium sulfate. The detailed standardized synthesis steps for specific derivatives are outlined below.
Monitoring via TLC confirms the consumption of starting materials, after which the reaction is quenched with water. The workup involves standard extraction with ethyl acetate, washing with saturated brine to remove residual salts, and drying over anhydrous magnesium sulfate. The detailed standardized synthesis steps for specific derivatives are outlined below.
- Dissolve the 1,4-naphthoquinone substrate in dichloromethane and cool to 0°C, while separately dissolving the 3-substituted-2-oxindole and chiral catalyst in solvent at -20°C.
- Add the quinone solution dropwise to the oxindole mixture and stir under air atmosphere at low temperature for 12 hours to facilitate asymmetric conjugate addition and oxidation.
- Quench the reaction with water, extract with ethyl acetate, wash with brine, dry over magnesium sulfate, and purify via flash column chromatography to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, this patented technology offers substantial strategic advantages by fundamentally altering the cost and risk profile of oxindole production. The elimination of expensive transition metal catalysts and the reliance on air as a free, abundant oxidant drastically reduces the raw material costs associated with the synthesis. Furthermore, the simplicity of the post-reaction workup, which avoids complex metal scavenging procedures, translates into significant operational savings and shorter batch cycle times. The use of stable, commercially available chiral organic catalysts ensures a secure supply chain, mitigating the risks associated with sourcing proprietary or scarce ligands often required in metal-catalyzed asymmetric synthesis.
- Cost Reduction in Manufacturing: The process utilizes inexpensive and readily available industrial commodities as starting materials, which are stable and do not require special storage conditions, leading to lower inventory holding costs. By avoiding the use of precious metals and stoichiometric oxidants, the direct material costs are significantly minimized, while the simplified purification process reduces solvent consumption and waste disposal expenses. This economic efficiency makes the commercial scale-up of complex pharmaceutical intermediates much more viable compared to traditional methods.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which are insensitive to water and utilize air, ensures consistent production outcomes regardless of minor environmental fluctuations. The catalysts employed are common commercial reagents with established supply chains, preventing bottlenecks that often occur with specialized custom-synthesized ligands. This reliability guarantees a steady flow of high-quality intermediates, reducing lead time for high-purity pharmaceutical intermediates and ensuring continuity for downstream drug manufacturing.
- Scalability and Environmental Compliance: The mild reaction temperatures and lack of exothermic hazards make this process inherently safer and easier to scale from gram to ton quantities without extensive re-engineering. The absence of heavy metals simplifies regulatory compliance and environmental reporting, as there is no need for rigorous testing and removal of metal residues. This green chemistry approach aligns with modern sustainability goals, reducing the environmental footprint of chemical manufacturing while maintaining high productivity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the process capabilities and limitations. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing development pipelines.
Q: What are the key advantages of this organocatalytic method over traditional metal-catalyzed routes?
A: This method eliminates the need for expensive and toxic transition metal catalysts, utilizing stable chiral organic small molecules instead. It operates under mild conditions using air as the oxidant, significantly reducing environmental impact and purification costs associated with heavy metal removal.
Q: What level of enantioselectivity can be achieved with this process?
A: The patent data demonstrates excellent stereocontrol, with specific examples achieving enantiomeric excess (ee) values as high as 97%. This high optical purity is critical for pharmaceutical applications where specific enantiomers dictate biological activity.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It uses readily available industrial commodities as raw materials, requires no special storage conditions, and features a simple post-treatment workflow involving standard extraction and chromatography, making it highly practical for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3-Disubstituted-2-Oxindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this organocatalytic technology in accelerating the development of next-generation therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the high enantiomeric excess values promised by this patent. We are committed to delivering high-purity pharmaceutical intermediates that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced synthesis method for your specific drug development projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data for our oxindole portfolio and discuss route feasibility assessments for your target molecules. Together, we can drive innovation and efficiency in the production of these vital pharmaceutical building blocks.
