Advanced Catalytic Synthesis of Carbon-Nitrogen Axis Chiral Sulfonamides for Commercial Scale-up
The landscape of asymmetric synthesis is continuously evolving, driven by the demand for high-purity chiral building blocks essential for modern drug discovery. Patent CN114436912A introduces a groundbreaking methodology for constructing carbon-nitrogen axis chiral sulfonamide compounds, a structural motif prevalent in bioactive molecules, organocatalysts, and ligands. This technology leverages the unique properties of cinchona alkaloid derivatives to facilitate the enantioselective alkylation of benzenesulfonamides with Morita-Baylis-Hillman (MBH) carbonates or allenyl esters. Unlike traditional approaches that often struggle with low selectivity or harsh conditions, this innovation offers a pathway to optical purity with enantiomeric excess values reaching up to 99% and yields as high as 99%. For R&D directors and process chemists, this represents a significant leap forward in accessing complex chiral architectures efficiently.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of axial chiral biaryl and amide compounds has relied heavily on aryl-aryl coupling, chirality transfer, or kinetic resolution strategies. These conventional pathways frequently suffer from substantial drawbacks that hinder their industrial applicability. Traditional resolution methods, for instance, are inherently limited by a maximum theoretical yield of 50%, necessitating the recycling of unwanted enantiomers which complicates the process flow. Furthermore, existing asymmetric catalytic methods for forming carbon-nitrogen axis chiral amides are relatively underdeveloped compared to their carbon-carbon counterparts. Many prior art techniques require stoichiometric amounts of expensive chiral auxiliaries or additional synthetic steps to prepare specialized catalysts, leading to inflated production costs and increased waste generation. Additionally, these older methods often exhibit narrow substrate scope and poor enantioselectivity, making them unsuitable for the diverse library synthesis required in modern pharmaceutical development.
The Novel Approach
In stark contrast, the methodology disclosed in CN114436912A utilizes commercially available benzenesulfonamides and easily prepared MBH carbonates or allenyl esters as starting materials. By employing a cinchona alkaloid derivative catalyst, specifically beta-ICD, the reaction proceeds under remarkably mild conditions, typically between -30°C and -50°C in organic solvents like mesitylene. This novel approach eliminates the need for stoichiometric chiral reagents, relying instead on a catalytic amount (as low as 1 mol%) of the organocatalyst to induce high levels of stereocontrol. The process is robust enough to be conducted under air conditions, removing the stringent requirement for inert gas setups that often complicate scale-up operations. This shift from resolution or transition-metal catalysis to organocatalysis not only simplifies the synthetic route but also aligns with green chemistry principles by reducing heavy metal contamination risks.
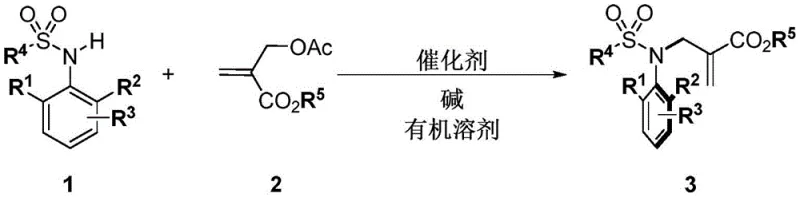
Mechanistic Insights into Cinchona Alkaloid-Catalyzed Alkylation
The core of this technological advancement lies in the precise activation of the electrophile by the chiral organocatalyst. The cinchona alkaloid derivative, acting as a Lewis base or through hydrogen bonding interactions, activates the MBH carbonate or allenyl ester, generating a reactive zwitterionic or ion-pair intermediate. This activated species then undergoes nucleophilic attack by the sulfonamide nitrogen. The bulky quinuclidine and quinoline moieties of the catalyst create a well-defined chiral environment that shields one face of the reacting center, thereby directing the formation of the carbon-nitrogen bond with high facial selectivity. This mechanism ensures that the resulting sulfonamide possesses a stable chiral axis, preventing racemization under the reaction conditions. The use of bases like cesium carbonate further facilitates the deprotonation of the sulfonamide, enhancing its nucleophilicity without compromising the integrity of the chiral catalyst system.
From an impurity control perspective, the high enantioselectivity inherent in this catalytic cycle is a critical advantage for pharmaceutical manufacturing. High ee values (>90%, often >99%) mean that the formation of the undesired enantiomer is minimized at the source, rather than having to be removed through difficult downstream purification processes like chiral HPLC or repeated recrystallizations. This reduces the overall impurity profile of the final API intermediate, simplifying regulatory filings and quality control assessments. Furthermore, the reaction tolerates a wide range of substituents on the benzene ring, including halogens, esters, and alkyl groups, demonstrating broad functional group compatibility. This versatility allows medicinal chemists to explore diverse chemical spaces without needing to redesign the synthetic strategy for each new analog, thereby accelerating lead optimization campaigns.
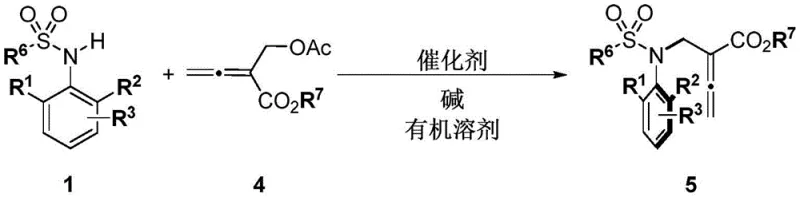
How to Synthesize Carbon-Nitrogen Axis Chiral Sulfonamides Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and stereoselectivity. The process begins with the preparation of the reaction mixture under controlled temperature conditions, followed by the slow addition of the electrophile to manage exotherms and maintain catalyst efficiency. The standardized protocol involves using mesitylene as the solvent and cesium carbonate as the base, with reaction times varying from 12 to 72 hours depending on the specific substrate reactivity. Detailed standard operating procedures for scaling this reaction from gram to kilogram quantities are essential for ensuring reproducibility and safety. For a comprehensive guide on the specific molar ratios, workup procedures, and purification techniques validated in the patent examples, please refer to the standardized synthesis steps provided below.
- Prepare the reaction mixture by combining benzenesulfonamide, cesium carbonate, and beta-ICD catalyst in mesitylene solvent under nitrogen atmosphere.
- Cool the system to -30°C to -50°C and add the MBH carbonate or allenyl ester electrophile slowly to initiate the alkylation.
- Stir the reaction for 12 to 72 hours, monitor by TLC, and purify the crude product via column chromatography to obtain high-purity chiral sulfonamides.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology offers tangible strategic benefits beyond mere chemical elegance. The shift towards organocatalysis using cinchona alkaloids addresses several critical pain points associated with traditional chiral synthesis, particularly regarding cost structure and supply chain resilience. By eliminating the reliance on precious transition metals like palladium or rhodium, manufacturers can avoid the volatility associated with noble metal pricing and the complex regulatory hurdles regarding heavy metal residues in final drug products. This transition translates directly into a more predictable cost model and reduced compliance burdens during the quality assurance phase.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of cheap and easily obtainable raw materials. Benzenesulfonamides and MBH carbonates are commodity chemicals that can be sourced from multiple suppliers, mitigating the risk of single-source dependency. Furthermore, the catalyst loading is exceptionally low, often as little as 1 mol%, which drastically reduces the cost per kilogram of the product compared to methods requiring stoichiometric chiral reagents. The mild reaction conditions also imply lower energy consumption for heating or cooling, contributing to overall operational expenditure savings. The simplified purification process, driven by high selectivity, reduces solvent usage and waste disposal costs, creating a leaner and more cost-effective manufacturing workflow.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the robustness of the reaction conditions. The ability to perform the synthesis under air conditions removes the need for specialized inert atmosphere equipment, allowing for greater flexibility in manufacturing site selection and batch scheduling. The starting materials are stable and commercially available in bulk quantities, ensuring that production timelines are not held hostage by the long lead times often associated with custom-synthesized chiral catalysts or sensitive organometallic reagents. This reliability is crucial for maintaining consistent supply to downstream API manufacturers, especially in the face of global logistical disruptions.
- Scalability and Environmental Compliance: Scaling complex chiral syntheses is notoriously difficult, but this method demonstrates excellent potential for commercial scale-up. The use of non-toxic organic solvents and the absence of heavy metals simplify the environmental, health, and safety (EHS) profile of the process. Waste streams are easier to treat, and the final product does not require extensive metal scavenging steps, which can be a bottleneck in large-scale production. The high atom economy and reduced waste generation align with increasingly stringent environmental regulations, positioning this technology as a sustainable choice for long-term manufacturing partnerships and green chemistry initiatives.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its integration into their supply chains. The following questions address common inquiries regarding the practical application, scope, and benefits of the patented method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a reliable basis for decision-making.
Q: What are the advantages of using cinchona alkaloid catalysts for sulfonamide synthesis?
A: Cinchona alkaloid derivatives, such as beta-ICD, enable highly enantioselective formation of carbon-nitrogen axial chirality under mild conditions without requiring stoichiometric chiral reagents or expensive transition metals.
Q: Can this synthesis method be performed under air conditions?
A: Yes, the patented process is robust and can be carried out under air conditions, which significantly simplifies operational requirements compared to traditional methods that often demand strict inert atmospheres.
Q: What is the typical enantiomeric excess achieved with this method?
A: The method demonstrates exceptional stereocontrol, achieving enantiomeric excess (ee) values of up to 99%, ensuring high optical purity suitable for pharmaceutical applications without extensive recrystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbon-Nitrogen Axis Chiral Sulfonamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organocatalytic methods like those described in CN114436912A for the production of high-value pharmaceutical intermediates. Our team of expert process chemists is dedicated to translating such innovative academic and patent technologies into robust, commercial-scale manufacturing processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from the laboratory bench to full-scale industrial output. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of chiral sulfonamide delivered meets the highest international standards for enantiomeric excess and chemical purity.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your next drug development program. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to request specific COA data for our chiral building blocks and to discuss route feasibility assessments for your target molecules. Let us help you secure a reliable supply of high-purity intermediates while optimizing your overall production costs and accelerating your time to market.
