Advanced Synthesis of 7-Benzyl-4,7-Diazaspiro[2.5]Octane for Scalable Pharmaceutical Manufacturing
Advanced Synthesis of 7-Benzyl-4,7-Diazaspiro[2.5]Octane for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry constantly seeks robust and scalable synthetic routes for complex heterocyclic scaffolds, particularly spiro-compounds which are increasingly prevalent in modern drug discovery pipelines. Patent CN111943893A discloses a groundbreaking synthesis method for 4,7-diazaspiro[2.5]octane derivatives, specifically targeting the efficient production of 7-benzyl-4,7-diazaspiro[2.5]octane. This technology represents a significant paradigm shift from traditional methodologies by completely eliminating the need for hazardous reduction reactions involving boron trifluoride adducts. For R&D directors and process chemists, this innovation offers a cleaner, safer, and more controllable pathway to access these valuable nitrogen-containing building blocks. The strategic design of this route ensures high purity profiles while mitigating the safety risks associated with handling energetic and corrosive reagents on an industrial scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthesis strategies, such as those disclosed in patents CN105111155A and CN108863958A, typically rely on a critical reduction step to convert carbon-oxygen double bonds within an amide intermediate into the desired amine functionality. This transformation necessitates the use of boron trifluoride diethyl etherate complexed with sodium borohydride, a combination that presents severe operational challenges. Boron trifluoride derivatives are notoriously corrosive to standard reactor materials, highly toxic upon inhalation, and possess significant flammability and explosivity hazards. Furthermore, the quenching of these reduction reactions generates substantial volumes of boron-containing waste, complicating downstream purification and environmental compliance. These factors collectively impose a heavy burden on manufacturing costs and limit the feasible batch sizes for safe production.
The Novel Approach
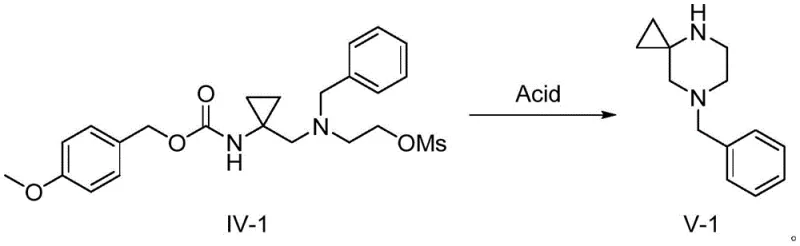
In stark contrast, the methodology presented in CN111943893A circumvents these pitfalls through a clever redesign of the synthetic logic, achieving the target spiro-cycle via direct cyclization rather than reduction. The process initiates with a substituted carbamate derivative and proceeds through a sequence of substitution, protection, esterification, and finally, acid-mediated cyclization. By transforming the terminal hydroxyl group into a superior leaving group (mesylate) and leveraging the inherent nucleophilicity of the deprotected amine, the ring closure occurs spontaneously under mild acidic conditions. This approach not only enhances the safety profile by removing explosive reagents but also streamlines the workflow, offering a reliable pharmaceutical intermediate supplier with a distinct competitive advantage in process efficiency and waste minimization.
![Specific synthetic route for 7-benzyl-4,7-diazaspiro[2.5]octane showing substitution, protection, esterification and cyclization](/insights/img/7-benzyl-4-7-diazaspiro-octane-synthesis-pharma-supplier-20260304025032-02.webp)
Mechanistic Insights into Nucleophilic Substitution and Acid-Catalyzed Cyclization
The success of this synthetic route hinges on the precise manipulation of leaving groups and nucleophiles to drive the formation of the strained spiro-system. The initial step involves the nucleophilic attack of ethanolamine on the mesylate precursor (Compound I). The mesylate group is an exceptional leaving group, facilitating a smooth SN2-type displacement even under relatively mild basic conditions using potassium carbonate in acetonitrile. This step installs the necessary ethylamine side chain while retaining the carbamate protection on the cyclopropyl amine, ensuring chemoselectivity. The subsequent benzylation of the secondary amine prevents unwanted intermolecular polymerization during the activation of the terminal alcohol, demonstrating a high level of control over the reaction trajectory and impurity profile.
The culmination of the synthesis is the elegant one-pot deprotection and cyclization sequence. Upon exposure to trifluoroacetic acid, the p-methoxybenzyloxycarbonyl (PMB) protecting group is cleaved, liberating the free primary amine. Simultaneously, the activated mesylate at the end of the side chain becomes susceptible to intramolecular nucleophilic attack. The liberated amine attacks the methylene carbon bearing the mesylate group, displacing the sulfonate and closing the six-membered piperazine ring fused to the cyclopropane. This mechanism avoids the formation of over-reduced byproducts often seen in hydride reductions, resulting in a cleaner crude reaction mixture and simplifying the isolation of the high-purity 7-benzyl-4,7-diazaspiro[2.5]octane product.
How to Synthesize 7-Benzyl-4,7-Diazaspiro[2.5]Octane Efficiently
The execution of this synthesis requires careful attention to stoichiometry and temperature control, particularly during the activation and cyclization phases. The protocol utilizes common organic solvents like acetonitrile and dichloromethane, which are easily recovered and recycled, further enhancing the green chemistry metrics of the process. Operators should monitor reaction progress via TLC to ensure complete consumption of starting materials before proceeding to workup, as residual mesylates can lead to alkylation impurities. The following guide outlines the standardized operational procedure derived from the patent examples, ensuring reproducibility and optimal yield for pilot and commercial batches.
- Perform nucleophilic substitution of the mesylate starting material with ethanolamine in acetonitrile using potassium carbonate.
- Protect the secondary amine with a benzyl group using benzyl bromide and base to prevent side reactions.
- Convert the terminal hydroxyl group into a mesylate leaving group using methanesulfonyl chloride and triethylamine.
- Execute final cyclization via acidic deprotection and intramolecular nucleophilic substitution to form the spiro ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible benefits that extend beyond mere chemical elegance. The elimination of specialized and hazardous reducing agents significantly reduces the complexity of raw material sourcing and storage requirements. Facilities no longer need to maintain strict protocols for handling pyrophoric or highly corrosive substances, thereby lowering insurance premiums and safety infrastructure costs. Moreover, the use of commodity chemicals like ethanolamine and benzyl bromide ensures a stable and resilient supply chain, mitigating the risk of production stoppages due to reagent shortages. This stability is crucial for maintaining continuous manufacturing operations and meeting tight delivery schedules for downstream API production.
- Cost Reduction in Manufacturing: The removal of the boron trifluoride/sodium borohydride reduction step leads to substantial cost savings by eliminating the need for expensive quenching procedures and specialized waste treatment for boron residues. The simplified workup procedures reduce solvent consumption and labor hours, directly impacting the cost of goods sold. Additionally, the higher selectivity of the cyclization step minimizes the formation of difficult-to-remove impurities, reducing the load on purification units and increasing overall throughput. These efficiencies compound to deliver a more economically viable manufacturing process without compromising on quality standards.
- Enhanced Supply Chain Reliability: By relying on stable, shelf-stable reagents that are widely available in the global chemical market, this process decouples production from the volatility of specialty reagent supply chains. The robustness of the reaction conditions, which tolerate ambient temperatures and standard atmospheric pressure, allows for flexible scheduling and easier technology transfer between manufacturing sites. This flexibility ensures that supply chain disruptions at one location can be rapidly mitigated by shifting production to alternative facilities, guaranteeing consistent availability of this critical pharmaceutical intermediate for clients.
- Scalability and Environmental Compliance: The absence of explosive reagents and the use of mild reaction conditions make this process inherently safer for scale-up from kilogram to multi-ton quantities. The waste stream is significantly cleaner, lacking heavy metals or toxic boron species, which simplifies effluent treatment and ensures compliance with increasingly stringent environmental regulations. This environmental compatibility not only reduces disposal costs but also aligns with the sustainability goals of major pharmaceutical companies, making the supplier a more attractive partner for long-term contracts focused on green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their existing supply chains. The answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making.
Q: Why is this new synthesis method safer than conventional routes?
A: Conventional methods rely on boron trifluoride diethyl etherate and sodium borohydride for reduction, which are corrosive, toxic, and flammable. This new route eliminates the reduction step entirely, utilizing mild substitution and cyclization reactions instead.
Q: What are the key reagents used in this process?
A: The process utilizes readily available and stable reagents including ethanolamine, benzyl bromide, methanesulfonyl chloride, and trifluoroacetic acid, avoiding hazardous reducing agents.
Q: Is this process suitable for large-scale production?
A: Yes, the reaction conditions are mild (room temperature to 0-10°C) and avoid explosive reagents, making it highly suitable for commercial scale-up and improving supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Benzyl-4,7-Diazaspiro[2.5]Octane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality spiro-compounds play in the development of next-generation therapeutics. Our technical team has thoroughly analyzed the route disclosed in CN111943893A and validated its potential for robust commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 7-benzyl-4,7-diazaspiro[2.5]octane meets the highest industry standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific drug development programs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this optimized route can improve your project economics. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us support your journey from discovery to commercial launch with our superior manufacturing capabilities.
