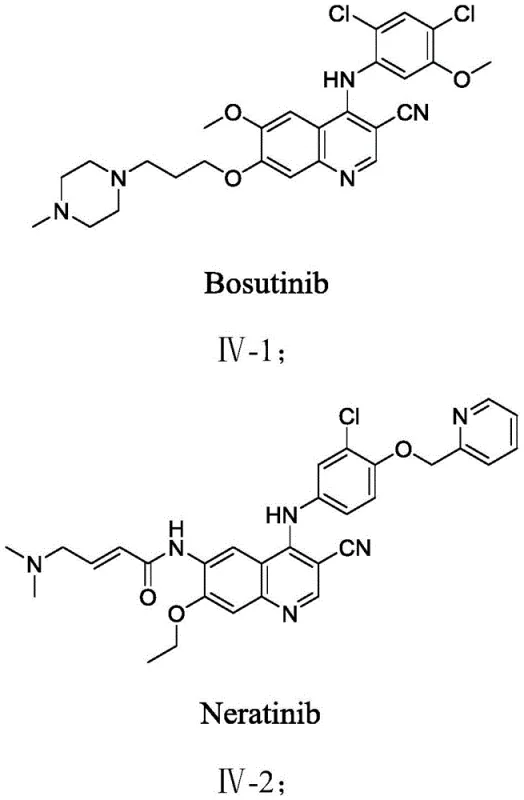
Advanced Ruthenium-Catalyzed Synthesis of 3-Cyanoquinoline Derivatives for Oncology Drug Manufacturing
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical heterocyclic scaffolds, particularly those serving as core structures for kinase inhibitors. Patent CN113620875B discloses a groundbreaking methodology for the preparation of 3-cyanoquinoline derivatives, a class of compounds pivotal in the development of anti-tumor agents such as Bosutinib, Neratinib, and Pyrotinib. This innovation addresses long-standing challenges in organic synthesis by providing a versatile route that accommodates diverse substituent groups at positions R1 through R6, thereby expanding the chemical space available for medicinal chemists. The disclosed method utilizes a Ruthenium-catalyzed system that operates efficiently under air conditions, eliminating the stringent requirement for inert atmospheres often associated with transition metal catalysis. By leveraging easily accessible raw materials and avoiding toxic nitrile sources, this technology represents a significant leap forward in green chemistry principles applied to complex pharmaceutical intermediate manufacturing.
The strategic importance of 3-cyanoquinoline derivatives cannot be overstated, given their role as dual inhibitors for protein kinases like SRC/ABL and tyrosine kinases like HER2/EGFR. Traditional synthetic approaches to these scaffolds often involve multi-step sequences with poor atom economy or the use of hazardous reagents. In contrast, the methodology described in CN113620875B streamlines the construction of the quinoline core through a direct annulation strategy. This not only enhances the overall efficiency of the synthesis but also aligns with the increasing regulatory pressure to minimize environmental impact in chemical production. For R&D directors and process chemists, this patent offers a tangible solution to improve the purity profile and impurity spectrum of key intermediates, ensuring a more reliable supply chain for downstream API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-cyanoquinoline derivatives has been plagued by several intrinsic limitations that hinder efficient large-scale production. Conventional routes frequently rely on the use of toxic cyanide salts or volatile cyanating agents, which pose severe safety risks to personnel and require specialized containment infrastructure to prevent environmental contamination. Furthermore, many traditional methods necessitate harsh reaction conditions, including high temperatures and strong acidic or basic media, which can lead to the decomposition of sensitive functional groups and the formation of complex impurity profiles. The reliance on stoichiometric amounts of oxidants or pre-functionalized starting materials often results in low atom economy and generates substantial quantities of chemical waste. Additionally, the need for strictly anhydrous and oxygen-free environments increases operational complexity and cost, making these processes less attractive for commercial manufacturing where robustness and reproducibility are paramount.
The Novel Approach
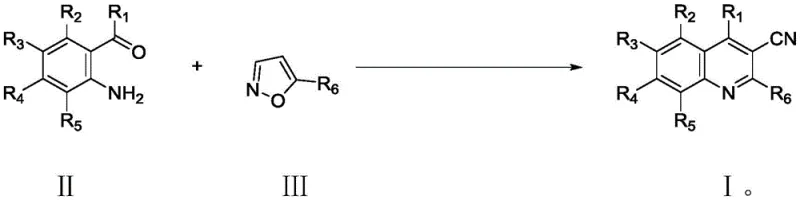
The novel approach detailed in patent CN113620875B fundamentally shifts the paradigm by utilizing isoxazole derivatives as safe and effective nitrile sources. This strategy circumvents the hazards associated with free cyanide, replacing them with stable heterocyclic precursors that undergo controlled ring-opening under catalytic conditions. The reaction proceeds under mild thermal conditions, typically between 60°C and 120°C, and remarkably tolerates the presence of oxygen, allowing operations to be conducted under simple air atmospheres rather than expensive inert gas setups. The use of a Ruthenium catalyst in conjunction with silver additives facilitates a cascade sequence involving N-O bond cleavage, aldol condensation, and subsequent cyclization. This tandem process not only shortens the synthetic route but also improves the overall yield and selectivity. The simplicity of the post-treatment workup, involving basic filtration and concentration, further underscores the practical advantages of this method for industrial applications.
Mechanistic Insights into Ruthenium-Catalyzed Cyclization
The mechanistic pathway of this transformation is a sophisticated example of transition metal-catalyzed C-H functionalization and heterocycle assembly. The process initiates with the coordination of the Ruthenium catalyst to the isoxazole ring, promoting the cleavage of the weak N-O bond. This critical step generates a reactive 3-oxopropionitrile intermediate or its equivalent enol form in situ. Simultaneously, the 2-aminobenzaldehyde derivative acts as the electrophilic partner. The generated nitrile-containing species then undergoes an aldol-type condensation with the aldehyde moiety of the aminobenzaldehyde, forming an alpha,beta-unsaturated carbonyl intermediate. This conjugated system is primed for the final cyclization event, where the ortho-amino group attacks the activated unsaturated system, followed by dehydration and aromatization to yield the stable 3-cyanoquinoline scaffold. The presence of the silver additive is believed to assist in the halide abstraction from the Ruthenium precursor, generating the active cationic catalytic species necessary for driving the cycle forward efficiently.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or high-energy pathways. The concerted nature of the condensation and cyclization steps minimizes the formation of side products derived from random radical coupling or polymerization. The mild acidity provided by additives like trifluoroacetic acid helps to protonate intermediates selectively, guiding the reaction towards the desired quinoline product while suppressing alternative condensation pathways. Furthermore, the high chemoselectivity of the Ruthenium system allows for the tolerance of various functional groups such as halogens, esters, and ethers on the benzene ring without requiring extensive protecting group strategies. This inherent selectivity translates directly to a cleaner crude reaction mixture, reducing the burden on downstream purification units and ensuring that the final intermediate meets the stringent purity specifications required for pharmaceutical grade materials.
How to Synthesize 3-Cyanoquinoline Derivatives Efficiently
Implementing this synthesis protocol requires careful attention to reagent quality and reaction parameters to maximize yield and reproducibility. The procedure involves combining the 2-aminobenzaldehyde substrate with the isoxazole derivative in a suitable polar solvent such as trifluoroethanol. The catalytic system, comprising a Ruthenium complex and a silver salt, is introduced along with a Brønsted acid promoter. The reaction mixture is then heated to the optimal temperature range, allowing the cascade transformation to proceed to completion. Monitoring the reaction progress via TLC or HPLC is recommended to determine the precise endpoint, although the patent indicates that 6 to 14 hours is generally sufficient for high conversion. Upon completion, the removal of the catalyst and metal residues is achieved through simple filtration over diatomaceous earth, followed by solvent evaporation. The resulting crude solid can be purified using standard column chromatography or recrystallization techniques to afford the target 3-cyanoquinoline derivative in high purity.
- Mix 2-aminobenzaldehyde derivative, isoxazole derivative, Ruthenium catalyst, Silver additive, acid, and solvent in a reaction vessel.
- Heat the mixture to 60-120°C under air atmosphere for 6-14 hours to facilitate N-O cleavage and cyclization.
- Filter the reaction mixture through diatomaceous earth, concentrate the filtrate, and purify via column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits that extend beyond mere chemical efficiency. The elimination of toxic cyanide sources significantly reduces the regulatory burden and safety compliance costs associated with handling hazardous materials. This shift not only lowers the insurance and infrastructure overheads but also mitigates the risk of production stoppages due to safety incidents. Moreover, the ability to run the reaction under air conditions removes the dependency on nitrogen or argon supply chains, simplifying the utility requirements of the manufacturing facility. The use of commercially available and inexpensive starting materials ensures a stable supply base, reducing vulnerability to raw material price volatility. These factors collectively contribute to a more resilient and cost-effective supply chain for critical oncology intermediates.
- Cost Reduction in Manufacturing: The streamlined nature of this one-pot synthesis eliminates multiple isolation and purification steps typically required in traditional multi-step routes. By avoiding the use of expensive and hazardous cyanating reagents, the raw material costs are substantially optimized. Furthermore, the simplified workup procedure reduces solvent consumption and waste disposal fees, leading to significant operational expenditure savings. The high catalytic efficiency means that lower loading of the precious metal catalyst may be achievable upon further optimization, further driving down the cost of goods sold for the final intermediate.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, specifically the tolerance to air and moisture, ensures consistent batch-to-batch performance even in varying manufacturing environments. This reliability minimizes the risk of batch failures and reworks, which are major disruptors in supply chain continuity. The broad substrate scope demonstrated in the patent examples implies that the same platform technology can be used to produce a wide array of substituted quinolines, allowing for flexible production scheduling and rapid response to changing market demands for different kinase inhibitor precursors.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of explosive or highly toxic reagents make this process inherently safer for scale-up from kilogram to tonne levels. The green chemistry attributes, such as high atom economy and reduced waste generation, align perfectly with modern environmental, social, and governance (ESG) goals. This compliance facilitates smoother regulatory approvals and enhances the corporate sustainability profile, which is increasingly important for partnerships with major multinational pharmaceutical companies seeking eco-friendly suppliers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing pipelines. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process development decisions.
Q: What are the key advantages of using isoxazoles as nitrile sources?
A: Using isoxazoles avoids the need for toxic cyanide compounds, significantly improving safety profiles and simplifying waste treatment protocols in large-scale manufacturing.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the reaction operates under air conditions with mild temperatures (60-120°C) and uses commercially available raw materials, making it highly suitable for commercial scale-up.
Q: What is the typical purity profile of the resulting intermediates?
A: The method achieves high selectivity and conversion rates. Post-treatment involving filtration and column chromatography ensures high-purity pharmaceutical intermediates suitable for downstream API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Cyanoquinoline Derivatives Supplier
As the global demand for targeted cancer therapies continues to rise, the need for high-quality, reliably sourced intermediates has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced synthetic methodologies like the one described in CN113620875B to deliver superior value to our partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 3-cyanoquinoline derivative meets the exacting standards of the pharmaceutical industry.
We invite you to collaborate with us to optimize your supply chain and accelerate your drug development timelines. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific project needs. Our technical procurement team is ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals. Contact us today to discuss how we can become your trusted partner in the synthesis of complex pharmaceutical intermediates.
