Advanced Electrochemical Synthesis of 3-Alkenyl-1,6-Diacid for Commercial Scale-Up
Introduction to Patent CN101691664A Technology
The chemical industry is currently undergoing a paradigm shift towards greener, more sustainable manufacturing processes, and patent CN101691664A represents a significant breakthrough in this domain by introducing a novel electrochemical method for synthesizing 3-alkenyl-1,6-diacids. This technology fundamentally alters the traditional approach to producing valuable dicarboxylic acid intermediates by utilizing carbon dioxide as a direct carboxylation agent in a high-pressure electrochemical cell, effectively turning a greenhouse gas into a valuable chemical feedstock. Unlike conventional thermal catalytic methods that often require expensive transition metal ligands and harsh oxidative conditions, this invention leverages electrons as clean reagents to drive the coupling of 1,3-dienes with CO2, resulting in a process that is inherently safer and more environmentally benign. For R&D directors and procurement strategists, this patent offers a compelling alternative route to critical building blocks like 3-hexenedioic acid, which serves as a versatile precursor for nylon production, pharmaceutical intermediates, and specialty polymers. The ability to operate without additional homogeneous catalysts not only simplifies the reaction mixture but also eliminates the complex and costly downstream removal of heavy metal residues, thereby streamlining the entire production workflow. Furthermore, the mild reaction conditions and the use of recyclable electrolytes position this technology as a robust solution for companies aiming to reduce their carbon footprint while maintaining high operational efficiency and product quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for adipic acid and related diacids, such as the KA oil method involving nitric acid oxidation, are plagued by severe environmental and safety drawbacks that increasingly conflict with modern regulatory frameworks. The oxidation of cyclohexanol and cyclohexanone with nitric acid generates substantial quantities of nitrous oxide (N2O), a potent greenhouse gas that requires expensive abatement technologies to mitigate, thereby inflating the overall production cost and complicating facility permitting. Alternative biological pathways, while theoretically green, often suffer from low volumetric productivity, high sensitivity to contamination, and the requirement for sterile conditions that are difficult and energy-intensive to maintain on a multi-ton commercial scale. Additionally, methods relying on hydrocyanation or carbonylation frequently utilize toxic reagents like hydrogen cyanide or carbon monoxide under high pressure with complex palladium or nickel catalyst systems, posing significant occupational health risks and necessitating rigorous safety protocols. The separation of products from these complex catalytic mixtures is often arduous, requiring multiple extraction and distillation steps that lead to yield losses and increased solvent consumption. Consequently, the industry faces a persistent challenge in finding a synthesis route that balances economic viability with stringent environmental compliance and operational safety.
The Novel Approach
The electrochemical carboxylation method described in this patent circumvents these historical bottlenecks by employing a diaphragm-free high-pressure electrolytic cell where the reaction is driven by electrical energy rather than thermal activation or stoichiometric oxidants. By dissolving 1,3-diene substrates directly into an organic electrolyte solution saturated with carbon dioxide, the process facilitates a direct C-C bond formation at the electrode surface, bypassing the need for pre-functionalized starting materials or toxic gaseous reagents like phosgene. The absence of a diaphragm simplifies the cell design and reduces capital expenditure, while the use of simple metal electrodes such as silver or iron eliminates the dependency on scarce and volatile precious metal catalysts. This approach ensures that the reaction proceeds with high atom economy, as the carbon dioxide is incorporated directly into the product skeleton, and the only by-products are minimal, easily separable species. Moreover, the operational flexibility allows for the tuning of product distribution by adjusting parameters such as current density and pressure, providing manufacturers with a versatile platform capable of adapting to varying market demands for specific diacid isomers without retooling the entire production line.
Mechanistic Insights into Electrochemical Carboxylation
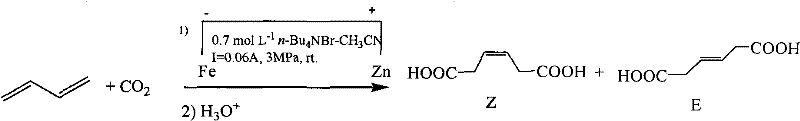
The core mechanism of this synthesis relies on the electro-reduction of carbon dioxide at the cathode surface to generate reactive radical anions or carbanion intermediates that subsequently attack the electron-deficient double bonds of the 1,3-diene substrate. In the high-pressure environment maintained between 1 MPa and 10 MPa, the solubility of CO2 in the organic solvent is significantly enhanced, ensuring a high local concentration of the electrophile at the electrode interface which drives the kinetics of the carboxylation forward. The choice of cathode material plays a pivotal catalytic role; for instance, silver cathodes have been demonstrated to provide superior selectivity and yield compared to zinc or copper, likely due to favorable adsorption energies for the CO2 intermediate that lower the activation barrier for the C-C coupling step. The organic ammonium salt serves a dual purpose as both the supporting electrolyte to ensure conductivity and a phase transfer agent that stabilizes the charged intermediates in the organic phase, preventing premature protonation or side reactions. This delicate balance of electrochemical potential and mass transfer allows for the precise formation of the 3-ene-1,6-diacid backbone with minimal over-reduction or polymerization of the diene starting material.
Impurity control in this electrochemical system is inherently robust due to the high selectivity of the electrode surface and the simplicity of the reaction matrix, which lacks the complex ligand spheres found in homogeneous transition metal catalysis. The primary impurities typically arise from isomeric variations, such as the formation of Z versus E isomers of the double bond, which can be managed through careful control of reaction temperature and electrolysis time. Since the electrolyte does not participate chemically in the bond-forming steps but merely facilitates charge transport, it remains chemically stable over multiple cycles, preventing the accumulation of degradation products that could foul the electrode or contaminate the product. Post-reaction workup involves a straightforward filtration to remove any solid precipitates followed by acidification, which protonates the carboxylate salts to release the free diacid, allowing for easy extraction into organic solvents. The final purification via recrystallization yields a product of exceptional purity, free from the heavy metal traces that are notoriously difficult to remove from products synthesized via traditional palladium or nickel-catalyzed routes, thus meeting the stringent specifications required for pharmaceutical and electronic grade applications.
How to Synthesize 3-Hexenedioic Acid Efficiently
The synthesis of 3-hexenedioic acid via this electrochemical protocol involves a streamlined sequence of operations that begins with the preparation of the conductive medium and concludes with the isolation of the crystalline diacid product. Operators must first charge a high-pressure electrolytic cell with a mixture of an organic solvent, such as acetonitrile or dimethyl sulfoxide, and a quaternary ammonium salt like tetrabutylammonium bromide to establish the necessary ionic conductivity. Once the cell is sealed, 1,3-butadiene gas is introduced followed by carbon dioxide until the internal pressure reaches the optimal range of 3 MPa to 10 MPa, ensuring the liquid phase is saturated with both reactants to maximize collision frequency at the electrode. The electrolysis is then conducted under constant current conditions using a silver cathode and an aluminum or iron anode, with the total charge passed carefully monitored to prevent over-electrolysis which could lead to side reactions. Following the completion of the reaction, the mixture is filtered to separate the crude product precipitate, which is then subjected to acidification with mineral acids and extracted to recover the target diacid in high yield.
- Prepare the electrolyte by mixing organic ammonium salts and organic solvents in a diaphragm-free high-pressure electrolytic cell.
- Introduce 1,3-diene compounds and pressurize with carbon dioxide gas to 1-10 MPa to ensure saturation.
- Apply constant current electrolysis using metal electrodes, followed by filtration, acidification, and recrystallization to isolate the pure diacid.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this electrochemical synthesis route offers transformative advantages by decoupling production costs from the volatility of precious metal markets and reducing dependency on complex catalyst supply chains. The elimination of expensive transition metal catalysts such as palladium or rhodium complexes removes a significant variable cost component and mitigates the risk of supply disruptions associated with these geographically concentrated resources. Furthermore, the ability to recycle the electrolyte solution multiple times without significant loss of performance drastically reduces the consumption of organic solvents and salts, leading to substantial savings in raw material procurement and waste disposal fees. The process operates under relatively mild temperatures compared to thermal oxidation methods, which translates to lower energy consumption for heating and cooling utilities, thereby improving the overall operating margin of the manufacturing facility. Additionally, the simplified downstream processing, which avoids complex chromatographic purification steps often required to remove metal catalysts, shortens the production cycle time and increases the throughput capacity of existing equipment.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the use of commodity-grade raw materials like butadiene and carbon dioxide, which are abundantly available and priced significantly lower than specialized functionalized precursors used in alternative synthetic routes. By removing the need for stoichiometric oxidants like nitric acid or hazardous reagents like hydrogen cyanide, the facility avoids the high costs associated with handling, storage, and neutralization of dangerous chemicals, resulting in a leaner and more cost-effective operation. The high selectivity of the reaction minimizes the formation of by-products, which means that less material is lost to waste streams and more of the input feedstock is converted into saleable product, enhancing the overall yield efficiency. Moreover, the longevity of the metal electrodes reduces the frequency of replacement parts procurement, contributing to lower maintenance expenditures and more predictable budget planning for plant managers.
- Enhanced Supply Chain Reliability: Implementing this technology diversifies the supply base for critical diacid intermediates by enabling production from widely available petrochemical feedstocks rather than relying on niche agricultural or fermentation-derived sources that are susceptible to seasonal fluctuations. The robustness of the electrochemical cell design allows for continuous or semi-continuous operation modes, which ensures a steady and reliable output of material to meet just-in-time delivery requirements for downstream customers. Since the process does not rely on sensitive biological cultures or enzymes that require strict cold-chain logistics and sterile environments, the risk of batch failure due to contamination is virtually eliminated, guaranteeing consistent supply continuity. This stability is crucial for long-term contracts with automotive or pharmaceutical clients who require assured availability of raw materials to maintain their own production schedules without interruption.
- Scalability and Environmental Compliance: The modular nature of electrochemical reactors facilitates straightforward scale-up from pilot plant to full commercial production without the nonlinear engineering challenges often encountered when scaling thermal batch processes. The inherent green chemistry attributes of utilizing CO2 as a feedstock align perfectly with corporate sustainability goals and increasingly strict environmental regulations regarding carbon emissions and volatile organic compound (VOC) releases. By generating minimal hazardous waste and avoiding the emission of nitrous oxides, facilities utilizing this method can secure environmental permits more easily and potentially qualify for green manufacturing incentives or carbon credits. This regulatory advantage future-proofs the investment against tightening environmental legislation, ensuring that the production asset remains compliant and operational for the long term without requiring costly retrofits for pollution control equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and performance of this electrochemical synthesis technology, providing clarity for stakeholders evaluating its adoption. These answers are derived directly from the experimental data and process descriptions detailed in the patent documentation, ensuring accuracy and relevance for industrial application assessments. Understanding these specifics is vital for engineering teams planning the integration of this process into existing manufacturing lines or for procurement officers negotiating supply agreements based on this methodology. The insights provided here cover aspects ranging from reaction selectivity and electrode durability to waste management and product specification compliance.
Q: What are the primary advantages of this electrochemical method over traditional nitric acid oxidation?
A: Unlike traditional nitric acid oxidation which generates hazardous nitrogen oxides, this electrochemical method utilizes carbon dioxide as a C1 synthon under mild conditions, eliminating toxic gas emissions and reducing environmental compliance costs significantly.
Q: Is the electrolyte solution reusable in this industrial process?
A: Yes, the patent specifies that the filtrate containing the organic ammonium salt and solvent can be recycled and reused directly in subsequent batches, which drastically lowers the consumption of auxiliary chemicals and reduces liquid waste treatment burdens.
Q: What level of selectivity can be achieved for the Z and E isomers?
A: The process demonstrates high selectivity for 3-ene-1,6-dioic acid isomers with minimal by-product formation, particularly when using silver cathodes, simplifying the downstream purification and crystallization steps required to meet high-purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hexenedioic Acid Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting advanced synthetic methodologies, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring this innovative electrochemical technology to the global market. Our state-of-the-art facilities are equipped with high-pressure electrochemical reactors and rigorous QC labs capable of verifying the stringent purity specifications required for high-performance polymer and pharmaceutical applications. We understand the critical importance of supply chain resilience and are committed to delivering high-purity 3-hexenedioic acid and related derivatives with consistent quality and reliability. By partnering with us, clients gain access to a sustainable and cost-optimized supply source that aligns with modern environmental standards while meeting the demanding technical requirements of next-generation materials.
We invite interested parties to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your unique volume and purity needs. Our experts are ready to provide a Customized Cost-Saving Analysis that demonstrates how switching to this electrochemically synthesized intermediate can optimize your overall manufacturing economics. Whether you require metric ton quantities for polymer production or kilogram scales for R&D, NINGBO INNO PHARMCHEM is prepared to support your growth with flexible manufacturing solutions and unwavering commitment to quality excellence.
