Advanced Palladium-Catalyzed Synthesis of Alkyne-Functionalized Arylamine Derivatives for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of Alkyne-Functionalized Arylamine Derivatives for Commercial Scale-Up
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more efficient, selective, and environmentally benign pathways to complex molecular scaffolds. Patent CN103408445A introduces a groundbreaking methodology for the preparation of novel arylamine derivatives, specifically those incorporating alkyne functionalities within a benzocyclobutanone framework. This technology represents a significant leap forward from conventional aromatic amine synthesis, which has historically relied on energy-intensive reduction processes or harsh coupling conditions. By leveraging a sophisticated dual-step palladium-catalyzed sequence, this invention unlocks access to highly functionalized intermediates that are critical for the development of next-generation organic electroluminescent materials and pharmaceutical active ingredients. For R&D directors and procurement strategists alike, understanding the mechanistic elegance and operational simplicity of this route is essential for securing a competitive edge in the supply of high-value specialty chemicals.
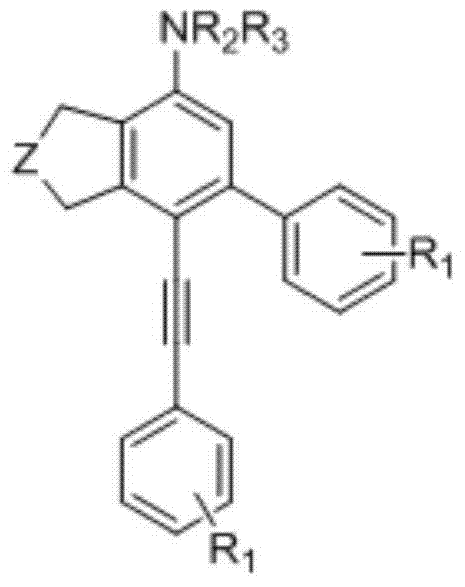
In the context of modern industrial chemistry, the ability to introduce alkyne bonds into an arylamine skeleton offers profound versatility. Alkynes serve as pivotal handles for further derivatization, allowing chemists to build complex molecular architectures through click chemistry or additional cross-coupling reactions. The patent explicitly highlights that while traditional arylamines are ubiquitous, their alkyne-functionalized counterparts occupy a unique niche due to their enhanced reactivity and electronic properties. This structural innovation is not merely academic; it addresses a tangible market demand for precursors that can be readily converted into high-performance hole-transport materials for OLED displays or specialized bioactive compounds. As a reliable arylamine derivative supplier, recognizing the strategic value of such versatile intermediates is crucial for long-term portfolio planning.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of aromatic amines has been dominated by the reduction of nitro compounds, a process fraught with significant environmental and safety challenges. Traditional routes often involve the use of iron filings in acidic media, sulfide reduction, or high-pressure catalytic hydrogenation, all of which generate substantial quantities of hazardous waste and require rigorous safety protocols. Furthermore, the synthesis of diarylamines, a closely related class of compounds, has typically relied on self-condensation of primary arylamines or coupling with aryl halides under extreme thermal conditions. 
 These legacy methods, as illustrated in the background art of the patent, frequently suffer from poor atom economy, limited substrate scope, and the formation of difficult-to-remove impurities. For a procurement manager, these inefficiencies translate directly into higher costs of goods sold (COGS) and extended lead times due to complex downstream processing requirements.
These legacy methods, as illustrated in the background art of the patent, frequently suffer from poor atom economy, limited substrate scope, and the formation of difficult-to-remove impurities. For a procurement manager, these inefficiencies translate directly into higher costs of goods sold (COGS) and extended lead times due to complex downstream processing requirements.
The Novel Approach
In stark contrast to these archaic methodologies, the process described in CN103408445A utilizes a modern organometallic strategy that prioritizes selectivity and mild reaction conditions. Instead of brute-force reduction, this novel approach employs a precise palladium-catalyzed cascade that constructs the desired arylamine骨架 (skeleton) with high fidelity. The elimination of heavy metal waste streams associated with iron or sulfide reductions significantly simplifies the environmental compliance burden. Moreover, the ability to conduct the key transformation at moderate temperatures (100°C) rather than the extreme heat or pressure required by older technologies drastically reduces energy consumption. This shift towards catalytic precision not only enhances the purity profile of the final product but also streamlines the overall manufacturing workflow, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Palladium-Catalyzed Cyclization and Amination
The core of this technological breakthrough lies in a meticulously orchestrated two-step catalytic cycle that transforms simple diyne precursors into complex polycyclic arylamines. The first stage involves a Sonogashira-type coupling where a 1,6-diyne reacts with phenylacetylene bromide in the presence of a Pd(PPh3)2Cl2 and CuI catalyst system. This step is conducted under anhydrous and oxygen-free conditions in acetonitrile at room temperature, demonstrating remarkable chemoselectivity. The resulting intermediate, referred to as Compound 1, serves as the critical precursor for the subsequent cyclization event. The second stage is where the true magic occurs: an intramolecular palladium-catalyzed amination/cyclization. Here, Compound 1 reacts with tri-n-butylamine in DMF solvent, facilitated by a Pd(OAc)2 and triphenylphosphine catalyst system. 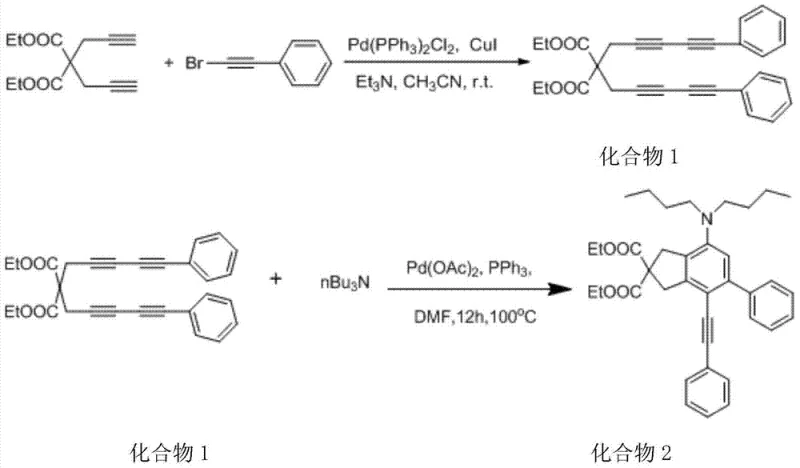 This transformation effectively closes the ring and installs the amine functionality simultaneously, a feat that would be incredibly difficult to achieve via stepwise classical synthesis.
This transformation effectively closes the ring and installs the amine functionality simultaneously, a feat that would be incredibly difficult to achieve via stepwise classical synthesis.
From an impurity control perspective, this mechanism offers distinct advantages over non-catalytic routes. The use of specific ligand-to-metal ratios, such as the 2:1 molar ratio of triphenylphosphine to palladium acetate specified in the patent, ensures that the catalytic cycle remains stable and minimizes the formation of side products like homocoupled alkynes or dehalogenated byproducts. The reaction proceeds cleanly to form the target benzocyclobutanone derivative, which can then be isolated via standard aqueous workup and column chromatography. The patent reports a column chromatography yield of approximately 88%, indicating a highly efficient conversion that maximizes raw material utilization. For R&D teams, this level of predictability and cleanliness is invaluable, as it reduces the need for extensive method development to remove trace impurities that could compromise downstream biological testing or material performance.
How to Synthesize Alkyne-Functionalized Arylamine Derivatives Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters outlined in the patent to ensure reproducibility and high yield. The process begins with the preparation of the alkyne-rich precursor under inert atmosphere, followed by the thermal cyclization step which drives the formation of the rigid arylamine core. Detailed standard operating procedures regarding reagent stoichiometry, solvent quality, and temperature ramping are critical for success. The following guide summarizes the key operational milestones derived from the patented examples, providing a roadmap for laboratory scale-up and process validation.
- Precursor Synthesis: Conduct a Sonogashira coupling between 1,6-diyne and phenylacetylene bromide using Pd(PPh3)2Cl2 and CuI catalysts in acetonitrile at room temperature for 24 hours.
- Palladium Catalysis & Cyclization: React the precursor with tri-n-butylamine in DMF using a Pd(OAc)2 and PPh3 catalyst system at 100°C for 12 to 24 hours.
- Purification: Perform aqueous workup followed by ethyl acetate extraction and column chromatography to isolate the final arylamine derivative with approximately 88% yield.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this palladium-catalyzed technology offers a compelling value proposition centered around operational efficiency and risk mitigation. By transitioning away from hazardous reduction chemistries towards catalytic coupling, manufacturers can significantly reduce the regulatory burden associated with waste disposal and worker safety. The mild reaction conditions (100°C) allow for the use of standard glass-lined or stainless steel reactors without the need for specialized high-pressure equipment, thereby lowering capital expenditure barriers for production. Furthermore, the high selectivity of the reaction minimizes the generation of complex impurity profiles, which often act as bottlenecks in purification and delay batch release.
- Cost Reduction in Manufacturing: The elimination of stoichiometric reducing agents such as iron powder or sodium sulfide removes a major source of solid waste, leading to substantial cost savings in waste treatment and disposal. Additionally, the high isolated yield reported in the patent implies better raw material efficiency, meaning less starting material is required to produce the same amount of finished goods. The use of common solvents like DMF and acetonitrile, which are readily available in the global supply chain, further stabilizes input costs and prevents supply disruptions associated with exotic reagents.
- Enhanced Supply Chain Reliability: The robustness of the palladium catalytic system ensures consistent batch-to-batch quality, a critical factor for maintaining long-term contracts with pharmaceutical clients. Because the reaction does not rely on sensitive biological enzymes or unstable reagents, the process is less susceptible to variations in ambient conditions, ensuring reliable delivery schedules. The modular nature of the two-step synthesis also allows for flexible production planning, where the precursor can be stockpiled and converted to the final amine on demand, optimizing inventory management.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram quantities is straightforward due to the homogeneous nature of the catalytic system and the absence of exothermic runaway risks common in nitro reductions. The process aligns well with green chemistry principles by maximizing atom economy and minimizing the use of toxic heavy metals in the final product through efficient purification. This environmental stewardship not only future-proofs the supply chain against tightening regulations but also enhances the brand reputation of the manufacturer as a sustainable partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these specialized arylamine derivatives. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners.
Q: What are the key advantages of this palladium-catalyzed method over traditional reduction routes?
A: Unlike traditional methods requiring harsh reducing agents like iron filings or hydrogen sulfide, this novel route operates under mild conditions (100°C) and utilizes precise palladium catalysis, resulting in significantly cleaner reaction profiles and easier purification.
Q: What is the typical yield and purity profile for these arylamine derivatives?
A: The patent data indicates that following the optimized column chromatography purification step, the process achieves an isolated yield of approximately 88%, ensuring high material throughput suitable for fine chemical applications.
Q: Can this synthesis method be scaled for industrial production of OLED or pharma intermediates?
A: Yes, the use of standard solvents like DMF and acetonitrile, combined with widely available palladium catalysts, makes this route highly adaptable for commercial scale-up, addressing the growing demand for functionalized hole-transport materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arylamine Derivatives Supplier
The synthesis of alkyne-functionalized arylamines described in CN103408445A exemplifies the kind of sophisticated chemistry that defines the modern fine chemical industry. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such complex patent methodologies into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to manufacturing plant is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch meets the exacting standards required for pharmaceutical and electronic material applications.
We invite you to collaborate with us to leverage this innovative technology for your specific product needs. Whether you require a Customized Cost-Saving Analysis for your existing supply chain or need to evaluate the feasibility of this route for a new project, our technical procurement team is ready to assist. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to quality and innovation can drive value for your organization.
