Advanced N-N Axial Chiral Bisindole Phosphine Ligand Synthesis for High-Value Pharmaceutical Intermediates
Advanced N-N Axial Chiral Bisindole Phosphine Ligand Synthesis for High-Value Pharmaceutical Intermediates
The landscape of asymmetric catalysis is continuously evolving, driven by the relentless demand for higher purity and more efficient synthetic routes in the pharmaceutical and fine chemical sectors. A groundbreaking development detailed in patent CN116041393A introduces a novel class of N-N axial chiral bisindole phosphine ligands, specifically represented by Formula 4, which promises to redefine stereoselective control in transition metal-catalyzed reactions. Unlike traditional binaphthyl-based scaffolds that have dominated the field for decades, this new architecture leverages the unique electronic and steric properties of the indole backbone to create a more versatile chiral environment. For R&D directors and procurement specialists seeking a reliable chiral ligand supplier, this technology represents a significant leap forward, offering a pathway to access complex chiral intermediates with unprecedented precision and reduced operational complexity.
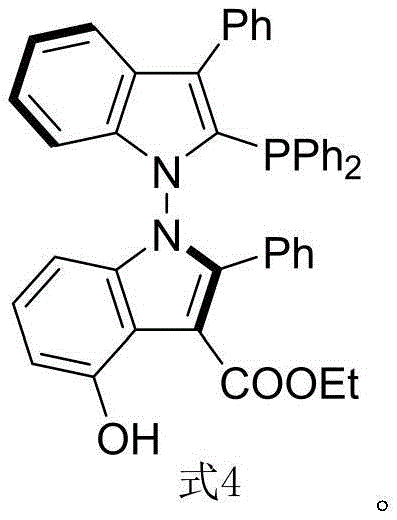
The strategic importance of this innovation lies in its potential to streamline the manufacturing of high-value active pharmaceutical ingredients (APIs). By shifting away from rigid, sterically hindered backbones to a more tunable N-N axis system, chemists can access a broader range of dihedral angles and hydrogen-bonding interactions. This flexibility is crucial for optimizing catalytic cycles in difficult transformations, such as asymmetric allylation, where subtle changes in the ligand sphere can drastically alter reaction outcomes. As we delve deeper into the technical specifics, it becomes clear that this patent offers not just a new molecule, but a robust platform for cost reduction in pharmaceutical intermediate manufacturing through improved efficiency and selectivity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of axially chiral phosphine ligands has relied heavily on binaphthyl (BINAP) or biphenyl scaffolds, which, while effective, suffer from inherent limitations in conformational flexibility and functionalization potential. Traditional methods often require harsh reaction conditions, precious metal catalysts for the axial chirality induction, and multi-step resolution processes that significantly drive up the cost of goods sold (COGS). Furthermore, the steric bulk of these conventional ligands can sometimes impede substrate access in crowded transition states, limiting their utility in the synthesis of bulky or complex drug candidates. For supply chain managers, these factors translate into longer lead times, higher dependency on scarce noble metals, and increased waste generation, all of which pose risks to commercial scale-up of complex polymer additives and pharmaceutical precursors.
The Novel Approach
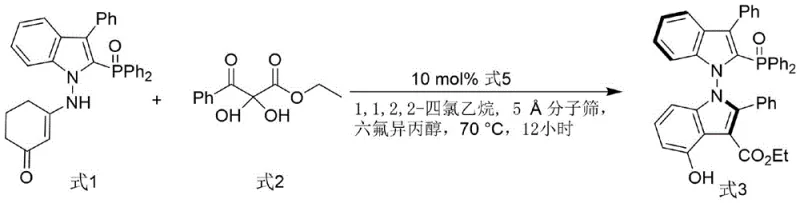
In stark contrast, the methodology disclosed in CN116041393A utilizes an organocatalytic strategy to construct the N-N axial chirality, bypassing the need for stoichiometric chiral auxiliaries or expensive metal templates in the ligand synthesis itself. The core transformation involves the coupling of an indole-derived enamine with a 2,3-diketoester, catalyzed by a chiral phosphoramide under mild thermal conditions. This approach not only simplifies the synthetic route but also introduces valuable hydrogen-bonding donors directly into the ligand framework, enhancing its ability to activate substrates through secondary interactions. The result is a ligand system that is both easier to produce and more effective in application, addressing the critical industry need for high-purity OLED material and pharmaceutical precursors with minimal environmental footprint.
Mechanistic Insights into Organocatalytic N-N Bond Formation
The mechanistic elegance of this synthesis lies in the dual activation mode facilitated by the chiral phosphoramide catalyst and the hexafluoroisopropanol (HFIP) additive. The phosphoramide acts as a Brønsted acid catalyst, activating the electrophilic diketoester through hydrogen bonding, while simultaneously organizing the nucleophilic indole enamine within a well-defined chiral pocket. The presence of HFIP further enhances this network by stabilizing charged intermediates and reinforcing the hydrogen-bonding array, which is critical for achieving high levels of stereoinduction. This cooperative catalysis ensures that the newly formed N-N axis adopts the desired configuration with high fidelity, yielding the intermediate Formula 3 with enantiomeric excess values that can reach up to 82% initially and 99% after recrystallization. Such precise control is essential for maintaining the integrity of the chiral information throughout the subsequent reduction steps.
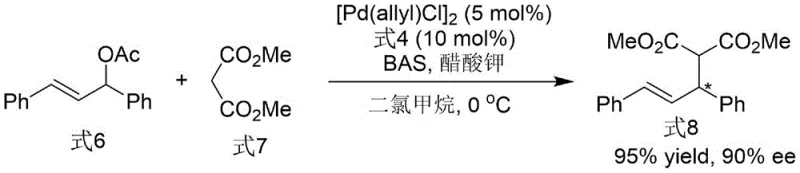
Following the construction of the chiral backbone, the conversion of the phosphine oxide moiety to the active phosphine species is achieved through a deoxygenation protocol using trichlorosilane and triethylamine. This step is pivotal, as it transforms the stable, air-resistant oxide precursor into the highly reactive phosphine ligand required for transition metal coordination. The retention of chirality during this reduction is paramount, and the patent data confirms that the optical purity is preserved, delivering the final Formula 4 ligand with 99% ee. This robustness against racemization underlines the stability of the N-N axis and validates the ligand's suitability for demanding applications like the asymmetric allylation shown below, where it delivers 90% ee in the formation of quaternary carbon centers.
How to Synthesize N-N Axial Chiral Bisindole Phosphine Ligand Efficiently
The practical implementation of this technology is designed for seamless integration into existing laboratory and pilot plant workflows. The process begins with the condensation of readily available indole enamines and diketoesters, utilizing molecular sieves to drive the equilibrium forward by removing water. The reaction is conducted in 1,1,2,2-tetrachloroethane, a solvent chosen for its ability to dissolve the polar intermediates while withstanding the reaction temperature of 70°C. Following the initial coupling, the crude intermediate is purified via silica gel chromatography and recrystallized to ensure maximum optical purity before proceeding to the final reduction. This streamlined workflow minimizes unit operations and solvent exchanges, directly contributing to reducing lead time for high-purity pharmaceutical intermediates.
- Couple indole-derived enamine with 2,3-diketoester using chiral phosphoramide catalyst, molecular sieves, and hexafluoroisopropanol in 1,1,2,2-tetrachloroethane at 70°C.
- Reduce the resulting N-N axial chiral bisindole derivative (Formula 3) using trichlorosilane and triethylamine in toluene at 120°C to yield the final phosphine ligand (Formula 4).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel ligand synthesis offers tangible strategic benefits beyond mere technical performance. The shift from metal-catalyzed axial chirality induction to an organocatalytic process eliminates the need for costly and potentially toxic heavy metals during the ligand manufacturing phase. This change significantly simplifies the purification train, reducing the burden on waste treatment facilities and lowering the overall cost of raw materials. Furthermore, the use of commodity chemicals like indole derivatives and simple esters as starting materials ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized chiral building blocks that often face market volatility.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts in the ligand synthesis step translates directly into lower production costs and reduced dependency on fluctuating metal markets. By relying on organic small-molecule catalysis, the process avoids the expensive downstream processing required to remove trace metal contaminants, which is a regulatory necessity for pharmaceutical grade materials. This efficiency gain allows for a more competitive pricing structure for the final ligand, enabling downstream users to achieve significant savings in their own API manufacturing costs without compromising on quality or performance standards.
- Enhanced Supply Chain Reliability: The reliance on broadly available chemical feedstocks rather than niche chiral pool resources enhances the robustness of the supply chain. Since the key starting materials are common intermediates in the fine chemical industry, sourcing bottlenecks are minimized, ensuring consistent availability even during periods of high global demand. This reliability is critical for long-term project planning and helps manufacturers maintain continuous production schedules, thereby securing the supply of critical medicines and advanced materials to the end market without interruption.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of hazardous reagents make this process inherently safer and easier to scale from gram to kilogram quantities. The use of molecular sieves as a dehydration agent is a green chemistry principle that reduces solvent waste compared to azeotropic distillation methods. Additionally, the high atom economy of the coupling reaction minimizes byproduct formation, aligning with increasingly stringent environmental regulations and corporate sustainability goals, thus facilitating smoother regulatory approvals for commercial manufacturing sites.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this technology into their pipelines, we have compiled answers to common inquiries regarding the ligand's performance and handling. These insights are derived directly from the experimental data provided in the patent, ensuring accuracy and relevance for process development scientists. Understanding these nuances is key to leveraging the full potential of the N-N axial chiral bisindole framework in your specific synthetic challenges.
Q: What distinguishes N-N axial chiral bisindole ligands from traditional BINAP derivatives?
A: N-N axial chiral bisindole ligands offer a wider dihedral angle control space and additional hydrogen bond activation sites compared to binaphthyl-derived ligands, leading to superior enantioselectivity in specific transition metal-catalyzed reactions.
Q: Is the synthesis of this ligand suitable for industrial scale-up?
A: Yes, the process utilizes mild reaction conditions (70°C and 120°C), commercially available starting materials, and avoids expensive transition metals in the ligand synthesis itself, making it highly suitable for cost-effective large-scale production.
Q: What level of enantiomeric excess (ee) can be achieved with this method?
A: The patented method achieves up to 99% ee after recrystallization of the intermediate, ensuring the final ligand provides excellent stereoselective control in downstream asymmetric allylation reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-N Axial Chiral Bisindole Phosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced chiral ligands like Formula 4 in accelerating drug discovery and process optimization. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of ligand delivered meets the highest international standards for enantiomeric excess and chemical purity required by top-tier pharmaceutical companies.
We invite you to collaborate with our technical team to explore how this novel ligand can enhance your asymmetric synthesis campaigns. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your process. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in innovation and supply chain excellence.
