Advanced P-Chiral Aminodiphosphine Ligands for Scalable Asymmetric Hydrogenation
The landscape of asymmetric synthesis is undergoing a significant transformation driven by the need for more robust and scalable chiral catalysts. Patent CN102781948A introduces a groundbreaking class of enantiomerically enriched aminodiphosphine ligands where chirality is located directly at the phosphorus atom. This innovation addresses critical stability issues found in prior art, offering a pathway to highly efficient asymmetric hydrogenation processes. These ligands, characterized by a phosphorus-nitrogen-phosphorus (P-N-P) backbone, represent a substantial leap forward in ligand design for the production of high-value chiral compounds.
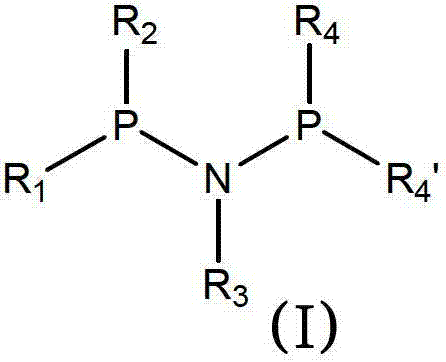
Traditionally, the reliance on phosphorus-carbon-phosphorus (PCP) backbones has presented significant logistical challenges for industrial applications. Conventional chiral PCP diphosphine ligands are frequently described as oils that are highly sensitive to oxidation upon exposure to air. This inherent instability necessitates their immediate conversion into metal complexes, often within a matter of hours, to prevent degradation. Such constraints impose severe limitations on supply chain flexibility, requiring just-in-time synthesis and complicating inventory management for large-scale manufacturing operations. Furthermore, the purification of these oily intermediates typically demands resource-intensive chromatographic techniques, which are difficult to scale and generate substantial solvent waste.
In stark contrast, the novel approach detailed in the patent utilizes a P-N-P framework that yields ligands capable of existing as stable solid salts. This physical state change from oil to solid is not merely cosmetic; it fundamentally alters the handling and storage profile of the material. The ability to isolate these ligands as crystalline salts, such as hydrochlorides or tetrafluoroborates, ensures resistance to air oxidation and significantly extends shelf life. This stability eliminates the urgency for immediate metal complexation, allowing for batch production, quality control testing, and strategic stockpiling. Moreover, the synthetic route leverages diastereomeric crystallization of borane complexes, effectively bypassing the need for preparative HPLC and enabling a more cost-effective and environmentally friendly purification process suitable for commercial scale-up.
Mechanistic Insights into Rhodium-Catalyzed Asymmetric Hydrogenation
The efficacy of these aminodiphosphine ligands stems from their unique ability to coordinate strongly and diastereoselectively with transition metals, particularly rhodium. When combined with precursors like [Rh(COD)2]BF4, the phosphorus atoms of the ligand displace labile ligands to form a cationic rhodium complex. The chirality at the phosphorus centers creates a highly defined chiral environment around the metal active site. This precise spatial arrangement is crucial for differentiating between the enantiotopic faces of prochiral substrates during the hydrogenation process. The nitrogen bridge in the P-N-P structure provides additional electronic tuning and conformational rigidity compared to carbon bridges, enhancing the catalyst's ability to induce high enantioselectivity even at low catalyst loadings.
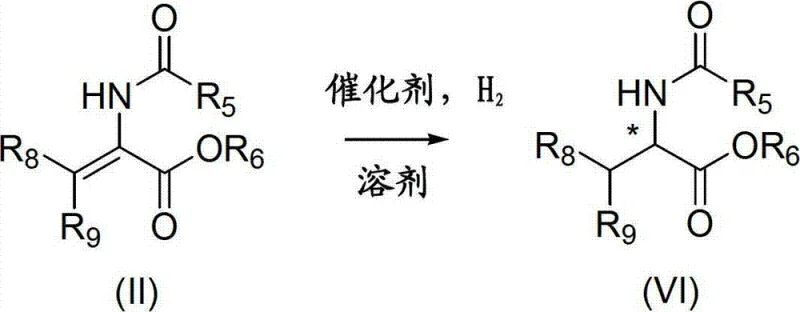
Impurity control is inherently managed through the robust nature of the catalyst system and the purification strategy of the ligand itself. Since the ligand can be purified to high enantiomeric excess (ee > 99%) via crystallization prior to metal complexation, the resulting catalyst starts with a high degree of optical purity. This minimizes the formation of the unwanted enantiomer during the reaction. Additionally, the stability of the ligand salts reduces the risk of oxidative byproducts that could poison the catalyst or contaminate the final product. The process tolerates a wide range of functional groups on the substrate, including esters and amides, ensuring that the final chiral amino acid products maintain high chemical purity alongside their stereochemical integrity.
How to Synthesize Enantiomerically Enriched Aminodiphosphine Ligands Efficiently
The synthesis of these advanced ligands follows a logical progression designed to maximize yield and optical purity while minimizing operational complexity. The process begins with the preparation of chiral aminomonophosphine borane complexes, which serve as stable building blocks. These intermediates are then coupled with chlorophosphines under basic conditions to construct the P-N-P skeleton. The borane protecting groups are subsequently removed to reveal the active phosphine centers. This modular approach allows for the variation of substituents on the phosphorus atoms to fine-tune catalytic performance. For a detailed breakdown of the standardized synthesis steps and reaction conditions, please refer to the guide below.
- React enantiomerically enriched aminomonophosphine borane complexes with chlorophosphines in the presence of a strong base to form the aminodiphosphine intermediate.
- Remove the borane protecting group from the intermediate using acid or base treatment to generate the free ligand or its salt.
- Isolate the final enantiomerically enriched ligand through crystallization, avoiding the need for chromatographic purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from unstable oily ligands to stable solid PNP ligands offers profound operational benefits. The primary advantage lies in the decoupling of ligand synthesis from catalyst usage. Because the ligands are stable solids, they can be manufactured in large batches, stored for extended periods, and shipped globally without the degradation risks associated with air-sensitive oils. This reliability significantly reduces the risk of production stoppages due to reagent failure. Furthermore, the elimination of chromatographic purification in favor of crystallization drastically reduces solvent consumption and waste disposal costs, aligning with green chemistry initiatives and reducing the overall cost of goods sold.
- Cost Reduction in Manufacturing: The shift to crystallization-based purification represents a major economic advantage over chromatography. Chromatographic separation is notoriously expensive at scale due to high solvent volumes, silica gel costs, and low throughput. By utilizing crystallization, the manufacturing process becomes significantly more streamlined and cost-efficient. Additionally, the stability of the ligands means less material is lost to degradation during storage and handling, improving overall material yield. The ability to use low catalyst loadings while maintaining high selectivity further reduces the consumption of precious metals like rhodium, contributing to substantial cost savings in chiral pharmaceutical intermediates manufacturing.
- Enhanced Supply Chain Reliability: Supply chain continuity is critical for pharmaceutical production. The air-stable nature of these ligand salts ensures that inventory remains viable over long periods, allowing for strategic stocking and buffering against supply disruptions. Unlike sensitive oils that require inert atmosphere shipping and immediate use, these solids can be handled with standard protocols. This simplifies logistics, reduces shipping costs associated with specialized containment, and ensures that raw materials are available when needed. The robustness of the supply chain is further strengthened by the use of commercially available starting materials for the ligand synthesis.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden inefficiencies, particularly in purification steps. The crystallization method described is inherently scalable, avoiding the bottlenecks of column chromatography. This facilitates the commercial scale-up of complex chiral ligands from gram to ton quantities. From an environmental perspective, the reduction in solvent waste and the avoidance of silica gel disposal lower the environmental footprint of the manufacturing process. This compliance with stricter environmental regulations reduces liability and supports sustainable manufacturing goals, making the technology attractive for long-term industrial adoption.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on the practical application of these ligands in asymmetric synthesis. Understanding these details is essential for evaluating the feasibility of integrating this catalytic system into existing production workflows.
Q: What distinguishes these PNP ligands from traditional PCP ligands?
A: Unlike traditional PCP ligands which are often oxidation-sensitive oils requiring immediate conversion, these PNP ligands can be isolated as stable solid salts with extended shelf life.
Q: How is high enantiomeric purity achieved without chromatography?
A: The process utilizes diastereomeric crystallization of borane complexes, allowing for the separation of isomers and achieving high ee values (>99%) without expensive chromatographic steps.
Q: What metals are compatible with these ligands for catalysis?
A: The ligands effectively coordinate with transition metals such as Rhodium (Rh), Ruthenium (Ru), Iridium (Ir), and Copper (Cu) to form active catalysts for asymmetric hydrogenation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Pharmaceutical Intermediates Supplier
The technological potential of P-chiral aminodiphosphine ligands is immense, offering a route to high-purity chiral amino acids essential for modern drug development. NINGBO INNO PHARMCHEM stands ready to support your transition to this advanced catalytic technology. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of ligand or catalyst meets the exacting standards required for pharmaceutical applications.
We invite you to engage with our technical procurement team to discuss how this technology can optimize your specific synthesis routes. By requesting a Customized Cost-Saving Analysis, you can gain insights into the potential economic benefits for your project. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your target molecules, ensuring a seamless integration of these high-performance ligands into your supply chain.
