Advanced Visible-Light Driven Synthesis of Glutaric Acid Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking sustainable methodologies to construct complex dicarboxylic acid scaffolds, which serve as critical building blocks for polyesters, polyamides, and active pharmaceutical ingredients. Patent CN113444005B introduces a groundbreaking strategy for synthesizing novel glutaric acid compounds from bisaryl-substituted non-activated olefins through a visible-light-driven aryl migration mechanism. This technology represents a paradigm shift from traditional harsh oxidative conditions to a mild, environmentally benign protocol that utilizes carbon dioxide as a renewable C1 resource. By leveraging photoredox catalysis, this method achieves high yields and exceptional functional group compatibility without the need for expensive transition metals or high-pressure equipment. For R&D directors and procurement specialists, this innovation offers a pathway to access high-purity pharmaceutical intermediates with a significantly reduced environmental footprint and optimized cost structure.
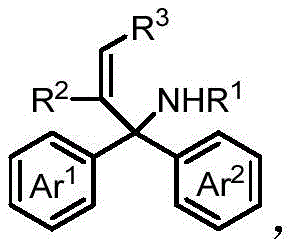
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of diacids has relied heavily on the selective oxidation of alcohols, cyclohexanol, or cyclohexanone using concentrated nitric acid, a process fraught with significant safety and environmental hazards. The generation of nitrous oxide (N2O), a potent greenhouse gas, during these oxidation reactions directly contradicts modern green chemistry principles and imposes heavy regulatory burdens on manufacturing facilities. Furthermore, these classical oxidative protocols are inherently limited to the synthesis of symmetric diacids, restricting the structural diversity available to medicinal chemists designing new drug candidates. Alternative methods involving transition metal-catalyzed dicarbonylation often require costly precious metal catalysts such as palladium or rhodium, which introduce challenges related to residual metal removal and stringent purity specifications required for pharmaceutical applications. These limitations necessitate a more versatile and sustainable approach to constructing complex diacid frameworks.
The Novel Approach
The methodology disclosed in CN113444005B overcomes these historical bottlenecks by employing a visible-light-mediated carboxylation strategy that operates under remarkably mild conditions. Instead of relying on stoichiometric oxidants, this process utilizes carbon dioxide, an abundant and non-toxic feedstock, to install carboxyl groups directly onto non-activated olefin substrates via a radical-mediated aryl migration. The reaction proceeds at room temperature under visible light irradiation, eliminating the need for energy-intensive heating or high-pressure reactors typically associated with CO2 fixation. This approach not only expands the substrate scope to include asymmetric and highly functionalized olefins but also ensures that the resulting glutaric acid products are obtained with high regioselectivity and diastereoselectivity. The elimination of heavy metal catalysts further simplifies the downstream purification process, making it an ideal candidate for cost reduction in fine chemical manufacturing.
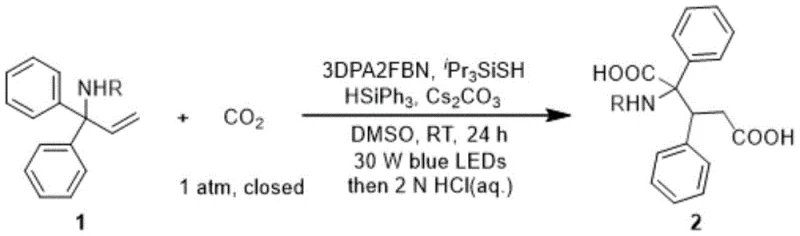
Mechanistic Insights into Visible-Light Driven Aryl Migration
The core of this technological advancement lies in its sophisticated radical mechanism, which orchestrates the activation of inert C-H bonds and the subsequent insertion of carbon dioxide through a series of single-electron transfer (SET) processes. Upon irradiation, the photosensitizer enters an excited state and undergoes reductive quenching by a hydrogen abstraction reagent, generating a radical species capable of activating the silane reducing agent. This activation sequence produces a key carbon dioxide radical anion intermediate, which adds to the non-activated olefin substrate to initiate the carboxylation cascade. The subsequent intramolecular 1,2-aryl migration is the defining step that constructs the complex glutaric acid skeleton, transforming a simple olefin into a highly functionalized diacid precursor. Understanding this mechanistic pathway is crucial for R&D teams aiming to optimize reaction parameters for specific substrate classes.
Impurity control is inherently managed by the selectivity of the radical intermediates and the mildness of the reaction conditions, which minimize side reactions such as polymerization or over-oxidation common in thermal processes. The use of specific bases and hydrogen abstraction reagents ensures that the radical chain propagation is efficient while suppressing unproductive pathways that could lead to byproduct formation. For quality assurance teams, this means that the crude reaction mixtures are cleaner, reducing the burden on chromatographic purification steps and improving overall material throughput. The ability to tolerate diverse functional groups, including halogens, ethers, and heterocycles, without compromising the integrity of the carboxylation process underscores the robustness of this catalytic system. This level of precision in bond construction is essential for producing high-purity pharmaceutical intermediates that meet rigorous regulatory standards.
How to Synthesize Novel Glutaric Acid Compounds Efficiently
Implementing this synthesis route requires careful attention to the preparation of the reaction environment, specifically the exclusion of oxygen and moisture which can quench the radical intermediates. The standard protocol involves charging a dried reaction vessel with the bisaryl-substituted olefin substrate, a suitable organic or metal-complex photosensitizer, a hydrosilane reducing agent, and an inorganic base. Following the establishment of a carbon dioxide atmosphere, a hydrogen abstraction reagent and anhydrous solvent are introduced, and the mixture is subjected to visible light irradiation at room temperature for a defined period ranging from 0.2 to 48 hours. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Prepare the reaction mixture by combining bisaryl-substituted non-activated olefin substrate, photosensitizer, reducing agent, and base in a dried vessel.
- Introduce carbon dioxide atmosphere and add hydrogen abstraction reagent and solvent, ensuring strict exclusion of oxygen and moisture.
- Irradiate the mixture with visible light at room temperature for 0.2 to 48 hours, followed by acidification and purification to isolate the glutaric acid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this photocatalytic technology offers substantial benefits by decoupling production costs from the volatility of precious metal markets and reducing dependency on hazardous oxidants. The substitution of expensive transition metal catalysts with organic dyes or earth-abundant metal complexes drastically lowers the raw material expenditure and eliminates the need for costly metal scavenging resins during purification. Additionally, the utilization of carbon dioxide as a carbon source provides a stable and inexpensive alternative to traditional carboxylating agents, contributing to significant cost savings in manufacturing overheads. The mild reaction conditions also translate to lower energy consumption, as there is no requirement for high-temperature heating or cryogenic cooling, further enhancing the economic viability of the process for large-scale production.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and hazardous oxidants removes major cost drivers associated with raw material procurement and waste disposal compliance. By avoiding the use of nitric acid, facilities can bypass the expensive scrubbing systems required to handle nitrogen oxide emissions, leading to substantial operational expenditure reductions. Furthermore, the simplified workup procedure reduces solvent usage and processing time, allowing for higher throughput within existing infrastructure without the need for capital-intensive equipment upgrades.
- Enhanced Supply Chain Reliability: The reliance on readily available commodity chemicals such as carbon dioxide, silanes, and organic bases ensures a resilient supply chain that is less susceptible to geopolitical disruptions affecting rare metal availability. The robustness of the reaction across a wide range of substrates means that a single manufacturing line can be adapted to produce various glutaric acid derivatives, increasing flexibility in responding to market demand fluctuations. This versatility allows procurement managers to consolidate suppliers and reduce the complexity of the vendor base while maintaining a steady flow of critical intermediates.
- Scalability and Environmental Compliance: Operating at ambient temperature and pressure significantly de-risks the scale-up process, as heat transfer and pressure containment become manageable engineering challenges rather than critical barriers. The green nature of the process aligns with increasingly stringent environmental regulations, minimizing the risk of production shutdowns due to compliance issues and enhancing the corporate sustainability profile. This environmental compatibility facilitates smoother regulatory approvals for new drug filings, accelerating time-to-market for downstream pharmaceutical products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible-light carboxylation technology in industrial settings. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation to provide clarity on feasibility and performance. Understanding these aspects is vital for stakeholders evaluating the integration of this method into their existing production portfolios.
Q: What are the primary advantages of this visible-light method over traditional nitric acid oxidation?
A: Unlike traditional methods that generate greenhouse gases like N2O and are limited to symmetric diacids, this photocatalytic approach utilizes CO2 under mild conditions to produce complex, asymmetric glutaric acid structures with high functional group tolerance.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the reaction operates at room temperature and atmospheric pressure with readily available reagents, eliminating the need for high-energy inputs or expensive transition metal catalysts, which facilitates easier commercial scale-up.
Q: What is the substrate scope for this carboxylation reaction?
A: The method demonstrates broad applicability across various bisaryl-substituted non-activated olefins, including those with electron-donating or withdrawing groups, heterocycles, and fused rings, maintaining high yields and selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glutaric Acid Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this visible-light driven synthesis in reshaping the landscape of dicarboxylic acid production for the global pharmaceutical industry. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust manufacturing processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that guarantee every batch of glutaric acid derivatives meets the highest international standards for safety and efficacy.
We invite forward-thinking organizations to collaborate with us to leverage this cutting-edge technology for their next-generation drug development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can optimize your supply chain and accelerate your project timelines.
