Revolutionizing Fluorinated Intermediate Production: Visible-Light C-F Bond Carboxylation for Commercial Scale
The landscape of organic synthesis is undergoing a transformative shift with the advent of sustainable methodologies that address the inherent inertness of strong chemical bonds. Patent CN115073250A introduces a groundbreaking protocol for the synthesis of alpha-aryl acetic acid and alpha-fluoro carboxylic acid compounds through a visible-light-mediated sp3 carbon-fluorine bond carboxylation reaction. This technology represents a significant leap forward in fluorine chemistry, enabling the direct utilization of carbon dioxide as a C1 synthon to functionalize robust C-F bonds under exceptionally mild conditions. By leveraging organic photocatalysts and silane reductants, this method bypasses the need for harsh thermal conditions or stoichiometric organometallic reagents, offering a greener and more atom-economical pathway to valuable fluorinated scaffolds. For R&D teams focused on developing next-generation therapeutics, this approach provides a powerful tool for constructing complex molecular architectures that were previously difficult or impossible to access efficiently.
Furthermore, the universality of this reaction system extends across a broad spectrum of substrates, including mono-, di-, and tri-fluorinated benzyl derivatives as well as difluoroesters and amides. The ability to selectively cleave specific C-F bonds in the presence of other sensitive functionalities underscores the precision of this catalytic system. From a commercial perspective, the reliance on inexpensive feedstocks like CO2 and commercially available silanes positions this technology as a highly viable candidate for cost reduction in fluorinated fine chemical manufacturing. As the demand for fluorinated pharmaceutical intermediates continues to surge, mastering such efficient activation strategies becomes paramount for maintaining competitiveness in the global supply chain. This patent not only fills a critical gap in sp3 C-H/C-F functionalization literature but also lays the groundwork for scalable industrial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the functionalization of carbon-fluorine bonds has been regarded as one of the most formidable challenges in synthetic organic chemistry due to the exceptional strength and low polarizability of the C-F bond. Conventional strategies often rely on the use of stoichiometric amounts of highly reactive organometallic reagents, such as Grignard or organolithium species, which require stringent anhydrous conditions and cryogenic temperatures to prevent decomposition. These methods are not only hazardous and difficult to handle on a large scale but also exhibit poor functional group tolerance, frequently leading to side reactions with esters, ketones, or nitriles present in the molecule. Moreover, transition-metal-catalyzed cross-coupling approaches, while effective, often necessitate the use of expensive noble metals like palladium or iridium, introducing significant cost burdens and potential toxicity issues related to residual metal contamination in the final API.
Additionally, existing carboxylation methods utilizing CO2 typically focus on sp2 hybridized carbons or activated benzylic positions, leaving the more inert sp3 C-F bonds largely untouched. The high bond dissociation energy of sp3 C-F bonds renders them unreactive towards standard nucleophilic attack or oxidative addition under mild conditions. Consequently, chemists have been forced to employ multi-step synthetic sequences involving pre-functionalization or protecting group strategies, which drastically reduce overall yield and increase waste generation. The inability to directly convert readily available fluorinated precursors into carboxylic acids using a simple, one-pot procedure has long been a bottleneck in the efficient production of fluorinated building blocks. This inefficiency translates directly into higher production costs and longer lead times for procurement teams seeking reliable sources of complex intermediates.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in Patent CN115073250A utilizes a photoredox catalytic cycle driven by visible light to activate the inert sp3 C-F bond at room temperature. By employing an organic photocatalyst such as 4CzIPN in conjunction with a silane reducing agent like triethylsilane and a mild base like cesium carbonate, the system generates radical intermediates capable of cleaving the C-F bond selectively. This radical pathway avoids the high energy barriers associated with ionic mechanisms, allowing the reaction to proceed under ambient pressure of CO2 (1 atm) without the need for specialized high-pressure equipment. The use of earth-abundant elements and organic catalysts eliminates the dependency on scarce transition metals, thereby simplifying the downstream purification process and ensuring high-purity products suitable for pharmaceutical applications.
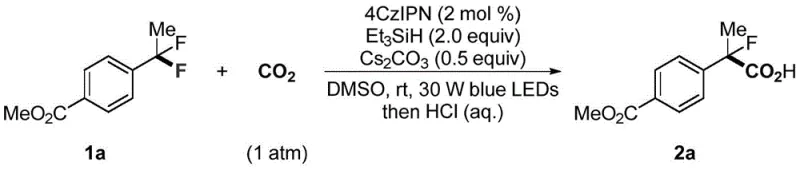
Moreover, this methodology demonstrates remarkable versatility, successfully accommodating a wide array of electronic and steric environments on the aromatic ring. Electron-withdrawing groups, which typically deactivate substrates in nucleophilic substitutions, are well-tolerated and often lead to improved yields in this radical manifold. The reaction conditions are sufficiently mild to preserve sensitive moieties such as esters, ethers, and heterocycles, enabling the direct late-stage carboxylation of complex drug-like molecules. For supply chain managers, this translates to a streamlined process that reduces the number of synthetic steps and minimizes the consumption of hazardous reagents. The ability to utilize CO2, a greenhouse gas, as a raw material further enhances the environmental profile of the process, aligning with modern sustainability goals and regulatory requirements for green chemistry in the fine chemical industry.
Mechanistic Insights into Photocatalytic sp3 C-F Bond Activation
The mechanistic underpinning of this transformation involves a sophisticated interplay between single electron transfer (SET) and hydrogen atom transfer (HAT) processes initiated by the excited state of the photocatalyst. Upon irradiation with blue LEDs, the organic photocatalyst (e.g., 4CzIPN) enters an excited state, which is then reductively quenched by the silane-base complex to generate a reduced photocatalyst species and a silyl radical. This silyl radical plays a crucial role in abstracting a fluorine atom or facilitating the formation of a carbon-centered radical via a radical-polar crossover mechanism. The resulting benzyl radical is subsequently reduced by the reduced photocatalyst to form a nucleophilic carbanion intermediate, which rapidly attacks the electrophilic carbon of the dissolved CO2. This sequence effectively converts a strong C-F bond into a new C-C bond, installing the carboxyl group with high regioselectivity.
Crucially, the choice of base and solvent significantly influences the efficiency of the radical generation and stabilization steps. Cesium carbonate acts not only as a base to activate the silane but also helps in stabilizing the carboxylate intermediate formed after CO2 insertion. The use of polar aprotic solvents like DMSO ensures adequate solubility of the ionic species and facilitates the diffusion of CO2 into the reaction medium. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters such as light intensity, catalyst loading, and reaction time to maximize yield and minimize byproduct formation. For R&D directors, this level of mechanistic clarity provides confidence in the robustness of the process, enabling rational optimization for specific substrate classes without extensive trial-and-error experimentation.
Furthermore, the selectivity of the C-F bond cleavage is governed by the stability of the intermediate radicals and the bond dissociation energies of the specific C-F bonds present. In polyfluorinated substrates, the system exhibits a preference for cleaving the bond that leads to the most stable radical intermediate, often allowing for differentiated functionalization in molecules with multiple fluorine atoms. This selectivity is vital for synthesizing specific isomers required for biological activity. The suppression of competing protonation pathways, which would lead to defluorinated side products, is achieved through the careful balancing of the reducing agent and the CO2 pressure. This precise control over the reaction trajectory ensures that the desired alpha-fluoro carboxylic acid is obtained as the major product, simplifying isolation and enhancing the overall economic viability of the synthesis.
How to Synthesize Alpha-Fluoro Carboxylic Acids Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must adhere to strict protocols regarding atmosphere control and light exposure to ensure reproducibility and safety. The process begins with the preparation of a dry reaction vessel, typically a Schlenk tube, where the photocatalyst and fluorinated substrate are weighed out accurately. It is imperative to maintain an inert environment initially to prevent moisture from deactivating the silane reagent before the CO2 atmosphere is established. Once the solid components are in place, the vessel is subjected to vacuum-refill cycles with CO2 to ensure saturation of the headspace and solution. The addition of the liquid reagents, including the anhydrous solvent and the silane reducing agent, should be performed via syringe under positive CO2 pressure to maintain the integrity of the reaction environment throughout the setup phase.
- Prepare the reaction mixture by adding the fluorinated substrate (e.g., benzyl fluoride), organic photocatalyst (4CzIPN), and base (Cs2CO3) into a dry reaction vessel under inert atmosphere.
- Replace the atmosphere with CO2 gas, then add the reducing agent (Et3SiH) and anhydrous solvent (DMSO) while maintaining the CO2 pressure at 1 atm.
- Irradiate the mixture with blue LEDs (30W) at room temperature for 2-48 hours, followed by acidic workup and purification to isolate the target carboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this photocatalytic carboxylation technology offers profound strategic advantages for procurement and supply chain operations within the pharmaceutical and fine chemical sectors. By shifting away from precious metal catalysts and cryogenic reagents, manufacturers can achieve substantial cost savings in raw material acquisition and waste disposal. The elimination of heavy metals from the process flow removes the need for expensive scavenging resins and rigorous metal testing, which are often bottlenecks in API release. Furthermore, the use of CO2 as a feedstock transforms a waste product into a value-added resource, insulating the supply chain from the volatility of petrochemical-derived C1 sources. This resilience is critical for maintaining continuous production schedules in the face of global raw material shortages.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with organic photocatalysts significantly lowers the bill of materials for each batch. Organic catalysts like 4CzIPN are generally cheaper to synthesize and easier to handle than their iridium or palladium counterparts. Additionally, the reaction proceeds at room temperature, eliminating the energy costs associated with heating or cooling large reactors. The simplified workup procedure, which avoids complex extraction steps required for metal removal, further reduces labor and solvent consumption. These cumulative efficiencies translate into a lower cost of goods sold (COGS), allowing companies to offer more competitive pricing for high-purity pharmaceutical intermediates without sacrificing margin.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as silanes, carbonates, and CO2 ensures a stable and diversified supply base. Unlike specialized organometallic reagents that may have long lead times or single-source suppliers, the reagents for this process are widely available from multiple vendors globally. This redundancy mitigates the risk of supply disruptions caused by geopolitical events or manufacturing incidents at specific facilities. Moreover, the mild reaction conditions reduce the wear and tear on equipment, extending the lifespan of reactors and minimizing unplanned maintenance downtime. For supply chain heads, this reliability means more predictable delivery timelines and the ability to scale production rapidly in response to market demand fluctuations.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging due to light penetration limits, but advancements in flow chemistry and LED technology have made kilogram-scale photoredox synthesis feasible. The low pressure (1 atm) and ambient temperature conditions simplify the engineering requirements for scale-up, avoiding the need for high-pressure autoclaves. From an environmental standpoint, the atom economy of incorporating CO2 and the absence of toxic metal waste align perfectly with increasingly stringent environmental regulations. This compliance reduces the regulatory burden and potential fines associated with hazardous waste disposal, making the process not only economically attractive but also socially responsible and sustainable for long-term operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this sp3 C-F bond carboxylation technology. These answers are derived directly from the experimental data and scope defined in the patent documentation, providing clarity on the method's capabilities and limitations. Understanding these details is essential for project managers evaluating the feasibility of integrating this route into existing manufacturing pipelines. The responses cover aspects ranging from substrate compatibility to process safety, ensuring a comprehensive overview for decision-makers.
Q: What are the advantages of using visible-light photocatalysis for C-F bond activation compared to traditional transition metal methods?
A: Visible-light photocatalysis eliminates the need for expensive and toxic transition metal catalysts like palladium or nickel. It operates under mild conditions (room temperature, 1 atm CO2), significantly reducing energy consumption and simplifying the removal of metal residues, which is critical for pharmaceutical grade purity.
Q: Can this method tolerate sensitive functional groups present in complex drug molecules?
A: Yes, the patent demonstrates excellent functional group tolerance. Substrates containing esters, ethers, cyano groups, and even other halogens can undergo carboxylation without degradation, making it suitable for late-stage functionalization of complex bioactive molecules.
Q: Is the process scalable for industrial production of fluorinated intermediates?
A: The process utilizes cheap and abundant raw materials like CO2 and silanes, and operates at atmospheric pressure. The absence of cryogenic conditions or high-pressure equipment facilitates easier scale-up from gram to kilogram levels, ensuring a reliable supply chain for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Fluoro Carboxylic Acids Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of photocatalytic C-F activation in the synthesis of advanced pharmaceutical intermediates. Our team of expert chemists has extensively studied the nuances of this technology, allowing us to offer customized synthesis solutions that leverage these mild and efficient conditions. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and risk-free. Our state-of-the-art facilities are equipped with specialized photoreactors and rigorous QC labs capable of meeting the most stringent purity specifications required by global regulatory bodies.
We invite you to collaborate with us to optimize your supply chain for fluorinated building blocks. By partnering with our technical procurement team, you can gain access to a Customized Cost-Saving Analysis tailored to your specific molecule, highlighting potential efficiencies in your current route. We encourage you to reach out today to request specific COA data and route feasibility assessments for your target alpha-fluoro carboxylic acids. Let us help you harness the power of green chemistry to drive innovation and profitability in your drug development programs.
