Scalable Manufacturing of Key Orelabrutinib Intermediates via Novel Condensation and Cyclization Strategies
Introduction to the Novel Synthetic Route
The pharmaceutical industry continuously seeks robust and scalable pathways for complex oncology intermediates, particularly for next-generation BTK inhibitors. Patent CN113563305B discloses a groundbreaking preparation method for 2-(4-phenoxyphenyl)-6-(N-substituted oxycarbonyl piperidine-4-) nicotinamide, a pivotal advanced intermediate in the synthesis of Orelabrutinib. This specific chemical architecture serves as the core scaffold for the drug, necessitating a manufacturing process that guarantees exceptional purity and structural integrity. The disclosed technology addresses the historical lack of efficient industrial methods for this specific derivative, offering a solution that leverages inexpensive starting materials like 3-nitropropionaldehyde. By integrating acid-catalyzed condensation with a sophisticated temperature-programmed reductive cyclization, this methodology establishes a new benchmark for producing high-purity pharmaceutical intermediates suitable for global regulatory standards.
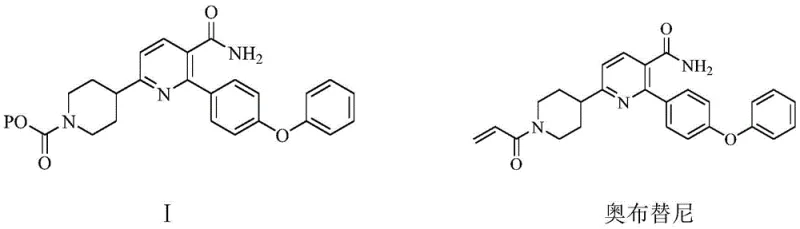
The strategic importance of this intermediate cannot be overstated, as it directly influences the quality profile of the final active pharmaceutical ingredient. The patent outlines a sequence that not only simplifies the operational workflow but also inherently minimizes the generation of difficult-to-remove impurities. For R&D directors and process chemists, this represents a significant opportunity to optimize the supply chain for BTK inhibitor production. The ability to synthesize this complex heterocyclic system with high selectivity using commodity chemicals suggests a pathway that is both economically viable and technically superior to traditional multi-step approaches that often suffer from low overall yields.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted nicotinamides with complex piperidine side chains has been plagued by challenges related to regioselectivity and harsh reaction conditions. Conventional routes often rely on expensive precursors or require multiple protection and deprotection steps that drastically increase the cost of goods and extend the manufacturing lead time. Furthermore, traditional cyclization methods frequently struggle with the formation of isomeric byproducts, where the amino group reacts with the wrong carbonyl functionality, leading to structurally similar impurities that are notoriously difficult to separate via crystallization. These inefficiencies result in substantial material loss and complicate the purification process, creating bottlenecks in the supply chain that can delay clinical trials and commercial launch timelines. The reliance on transition metals that are difficult to remove also poses significant regulatory hurdles regarding residual metal limits in the final drug substance.
The Novel Approach
In stark contrast, the methodology described in CN113563305B introduces a streamlined, convergent strategy that bypasses many of these traditional pitfalls. The novel approach utilizes a tandem sequence where a nitro-enamide intermediate is coupled with a protected piperidone under carefully controlled alkaline conditions. This is followed by a ingenious one-pot reduction-cyclization-isomerization cascade. By employing temperature programming, the process selectively reduces the nitro group at lower temperatures to generate the reactive amine in situ, which then undergoes cyclization. Subsequent heating drives the isomerization to the thermodynamically stable aromatic pyridine ring. This integration of steps eliminates the need for isolating unstable intermediates, thereby reducing solvent consumption and processing time. The use of a co-catalyst system further refines the selectivity, ensuring that the cyclization occurs exclusively at the desired position to yield the target nicotinamide scaffold with exceptional fidelity.
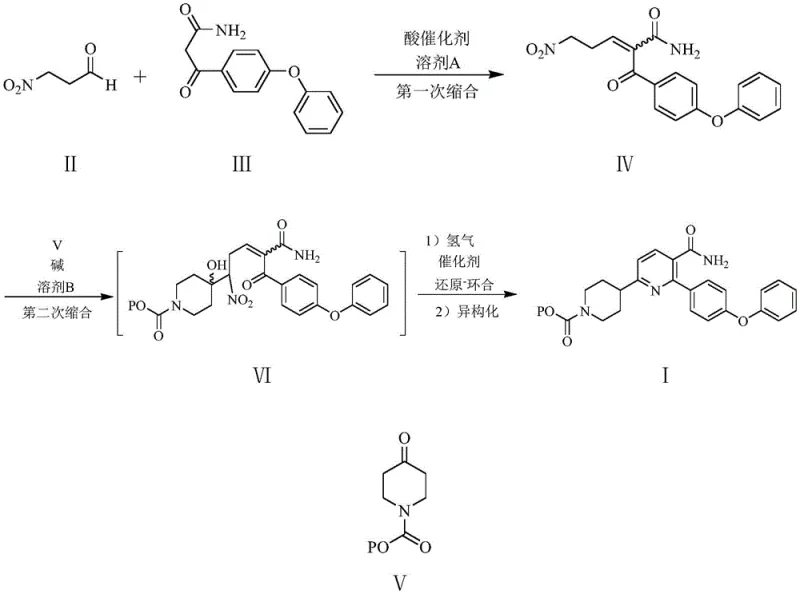
Mechanistic Insights into Temperature-Programmed Reductive Cyclization
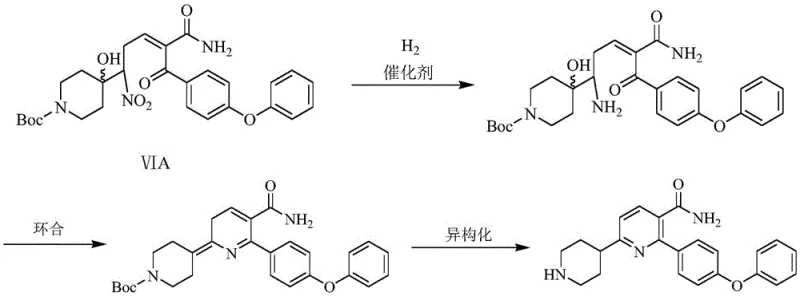
The core innovation of this patent lies in the mechanistic understanding and control of the final cyclization step, which transforms a linear precursor into the rigid pyridine core. The reaction begins with the catalytic hydrogenation of the nitro group on the side chain, converting it into a primary amine. Under the specific conditions employed, this newly formed amine is highly nucleophilic. However, the molecule contains multiple electrophilic sites, including the ketone carbonyl and the amide carbonyl. The patent reveals that at lower temperatures, the kinetic control favors the interaction of the amine with the ketone carbonyl adjacent to the phenoxyphenyl group, initiating the ring closure. This selectivity is crucial because reaction at the amide carbonyl would lead to an undesired isomer. The presence of a co-catalyst, such as triphenylphosphine, plays a subtle yet vital role by coordinating with the catalyst surface or the substrate to favor a specific conformation (trans-configuration) that aligns the reacting centers optimally for the desired cyclization pathway.
Following the initial ring closure, the intermediate exists in a dihydropyridine or similar non-aromatic state. To achieve the final stable aromatic system, the process employs a temperature ramp to 70-75°C. This thermal energy input drives the isomerization and aromatization steps, effectively locking the structure into the desired 2-(4-phenoxyphenyl)-6-(N-substituted oxycarbonyl piperidine-4-) nicotinamide configuration. The mechanistic pathway avoids the formation of the 2-aminopyridine byproduct, which typically arises if the reduction is performed at excessively high temperatures where the amine might attack the amide carbonyl instead. This precise control over reaction kinetics and thermodynamics ensures that the impurity profile remains minimal, significantly reducing the burden on downstream purification units and enhancing the overall process mass intensity.
How to Synthesize 2-(4-phenoxyphenyl)-6-(N-substituted oxycarbonyl piperidine-4-) nicotinamide Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for replicating this high-efficiency route in a pilot or commercial plant setting. The process is designed to be robust, tolerating slight variations in reagent quality while maintaining high performance. It begins with the preparation of the nitro-enamide backbone, followed by the critical coupling with the piperidine fragment. The final transformation relies on standard hydrogenation equipment but requires precise control over the thermal profile to ensure the correct isomer is obtained. For process engineers, the ability to perform the reduction and cyclization in a single vessel without intermediate isolation represents a significant operational advantage, reducing equipment footprint and cleaning validation requirements. The detailed standardized synthesis steps for implementing this proprietary technology are outlined below.
- Perform a first condensation between 3-nitropropionaldehyde and 3-(4-phenoxyphenyl)-3-oxo-propionamide using an acid catalyst in solvent A to obtain the nitro-enamide intermediate.
- Execute a second condensation by reacting the nitro-enamide with N-substituted oxycarbonyl piperidin-4-one under alkaline conditions, utilizing dropwise addition to control selectivity.
- Conduct a one-pot reduction-cyclization and isomerization using a palladium catalyst and co-catalyst under hydrogen pressure with precise temperature programming to form the final pyridine ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The primary advantage stems from the utilization of 3-nitropropionaldehyde and substituted acetophenones as starting materials, which are commodity chemicals available from multiple global suppliers. This diversification of the raw material base mitigates the risk of supply disruptions that often plague processes relying on bespoke or single-source intermediates. Furthermore, the high selectivity of the reaction sequence means that the crude product possesses high purity, often exceeding 99% after simple recrystallization. This reduction in impurity load translates directly into lower solvent usage for purification and higher recovery rates of the final product, effectively lowering the cost per kilogram of the manufactured intermediate.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts that require complex removal steps, combined with the use of inexpensive acid and base catalysts, significantly lowers the direct material costs. Additionally, the one-pot nature of the final cyclization step reduces the number of unit operations, leading to substantial savings in labor, energy, and solvent consumption. The high yield reported in the examples indicates that less raw material is wasted, further driving down the variable cost of production and improving the overall margin profile for the manufacturing campaign.
- Enhanced Supply Chain Reliability: By establishing a synthesis route that relies on widely available bulk chemicals rather than specialized building blocks, the supply chain becomes more resilient to market fluctuations. The robustness of the process, demonstrated by its tolerance to various solvents and catalysts, ensures consistent production output even if specific reagent grades vary slightly. This reliability is critical for maintaining continuous supply to downstream API manufacturers, preventing costly delays in drug formulation and packaging schedules.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard reaction types such as condensation and catalytic hydrogenation that are well-understood in large-scale chemical engineering. The reduction in waste generation, due to high atom economy and fewer purification steps, aligns with modern green chemistry principles and environmental regulations. This facilitates easier permitting and compliance with increasingly stringent waste discharge standards, ensuring long-term operational sustainability for the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific experimental data and mechanistic explanations provided in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for commercial production. The answers highlight the critical process parameters that must be controlled to achieve the reported success.
Q: What is the critical factor for controlling selectivity in the second condensation step?
A: The dropwise addition of N-substituted oxycarbonyl piperidin-4-one at low temperatures (-20 to 25°C) is critical. This prevents the self-condensation or polymerization of the piperidone, ensuring high yield and purity of the coupled intermediate.
Q: How does the temperature programming in the final step improve product quality?
A: The process utilizes a two-stage temperature profile: lower temperatures (25-30°C) facilitate the selective reduction of the nitro group and initial cyclization, while higher temperatures (70-75°C) drive the necessary isomerization to form the stable pyridine aromatic system, minimizing byproduct formation.
Q: Why is a co-catalyst like triphenylphosphine used in the hydrogenation step?
A: The co-catalyst assists in the 1,4-addition of the unsaturated ketone during reduction. It helps stabilize the specific conformation required for the amino group to cyclize with the correct carbonyl group, significantly enhancing the selectivity for the target nicotinamide structure over potential isomers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(4-phenoxyphenyl)-6-(N-substituted oxycarbonyl piperidine-4-) nicotinamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving oncology therapies. Our technical team has extensively analyzed the pathway described in CN113563305B and possesses the expertise to execute this complex synthesis with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our facilities are equipped with state-of-the-art hydrogenation reactors and rigorous QC labs capable of meeting stringent purity specifications required for GMP manufacturing of pharmaceutical intermediates.
We invite you to collaborate with us to leverage this advanced technology for your Orelabrutinib supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to discuss specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and cost-effective chemical strategy available.
