Advancing Pharmaceutical Intermediate Synthesis via Additive-Free Radical Cyclization Technology
Introduction to Green Radical Cyclization Technology
The landscape of organic synthesis is rapidly shifting towards methodologies that prioritize atom economy and environmental sustainability without compromising on yield or selectivity. Patent CN111233732A introduces a groundbreaking approach to the construction of complex heterocyclic scaffolds through a free radical reaction method involving 1,6-dienes and alcohols under an additive-free system. This technology represents a significant leap forward for the production of high-purity pharmaceutical intermediates, specifically functionalized pyrrolidinone derivatives which are ubiquitous motifs in bioactive molecules. By eliminating the need for transition metal catalysts and external bases, this process addresses critical pain points in modern process chemistry, such as heavy metal contamination and excessive waste generation. The core innovation lies in the direct oxidative functionalization of the alcohol α-C(sp3)-H bond, which serves as the initiating event for a highly regioselective radical cyclization cascade.
Furthermore, the operational simplicity of this method cannot be overstated, as it proceeds efficiently under mild thermal conditions and tolerates ambient air atmospheres. This stands in stark contrast to traditional radical protocols that often demand rigorous exclusion of oxygen and moisture to prevent side reactions or catalyst deactivation. The ability to utilize simple alcohols not merely as solvents but as active reactants embodies the "multi-purpose" strategy that is gaining traction in green chemistry circles. For a reliable pharmaceutical intermediates supplier, adopting such a streamlined protocol translates directly into enhanced process robustness and reduced regulatory hurdles associated with residual metal limits in final drug substances. The following analysis delves into the mechanistic nuances and commercial implications of this transformative synthetic route.
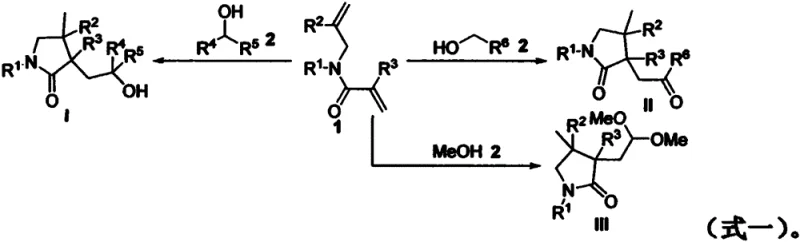
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of pyrrolidinone rings via the cyclization of 1,n-dienes has relied heavily on transition metal catalysis, utilizing precious metals such as palladium, rhodium, or copper to facilitate the bond-forming events. While effective, these conventional methods suffer from inherent drawbacks that complicate large-scale manufacturing, primarily the difficulty in removing trace metal residues to meet stringent pharmacopeial standards. Additionally, many traditional radical processes require stoichiometric amounts of hazardous reagents or harsh Lewis acids to activate the substrates, leading to significant safety concerns and increased waste disposal costs. The reliance on inert atmospheres, such as argon or nitrogen, further escalates the operational complexity and capital expenditure required for reactor setups, making these processes less attractive for cost reduction in fine chemical manufacturing. Moreover, the lack of regioselectivity in non-catalyzed thermal radical reactions often results in complex mixtures of isomers, necessitating energy-intensive purification steps that erode overall process efficiency.
The Novel Approach
The methodology disclosed in CN111233732A circumvents these challenges by employing a metal-free, base-free oxidative system that leverages the intrinsic reactivity of organic peroxides. By using tert-butyl peroxybenzoate (TBPB) as the sole oxidant, the reaction initiates a radical chain mechanism that selectively targets the α-C-H bond of the alcohol, generating a carbon-centered radical that adds intramolecularly to the pendant alkene. This novel approach not only achieves excellent yields, often exceeding 90% for optimized substrates, but also ensures high diastereoselectivity, typically favoring a single isomer with a ratio greater than 20:1. The tolerance for air atmosphere is a particularly disruptive feature, as it removes the necessity for expensive degassing equipment and allows for simpler batch processing. This shift from metal-dependent catalysis to organic oxidative radical chemistry aligns perfectly with the industry's drive towards sustainable and economically viable synthetic routes for complex organic scaffolds.
Mechanistic Insights into Oxidative Radical Cyclization
Understanding the underlying mechanism is crucial for R&D teams aiming to adapt this technology for diverse substrate libraries. The reaction proceeds through a well-defined radical pathway initiated by the thermal decomposition of the peroxide oxidant, which generates tert-butoxy radicals. These reactive species abstract a hydrogen atom from the α-position of the alcohol substrate, creating a nucleophilic α-hydroxyalkyl radical. This key intermediate then undergoes a 5-exo-trig cyclization onto the electron-deficient alkene of the 1,6-diene system, forming a new C-C bond and a transient alkyl radical on the pyrrolidinone ring. Subsequent oxidation and proton loss, or hydrogen atom transfer from the solvent, terminates the cycle to yield the stable cyclized product. The elegance of this mechanism lies in its ability to differentiate between secondary and primary alcohols; secondary alcohols stop at the hydroxyl stage, whereas primary alcohols undergo further oxidation to ketones due to the instability of the initial adduct under the reaction conditions.
To validate this radical hypothesis, control experiments were conducted using known radical scavengers such as TEMPO and BHT. As illustrated in the experimental data, the addition of 3.0 equivalents of these inhibitors completely suppressed product formation, yielding only trace amounts of the desired compound. This confirms that the transformation is indeed driven by a free radical process rather than an ionic or concerted mechanism. The proposed catalytic cycle, depicted below, highlights the role of the oxidant in regenerating the active radical species and driving the reaction to completion. Such mechanistic clarity provides confidence in scaling the process, as the kinetic parameters are well-understood and manageable within standard chemical engineering frameworks. This level of insight is invaluable for ensuring consistent quality and reducing lead time for high-purity intermediates during technology transfer phases.
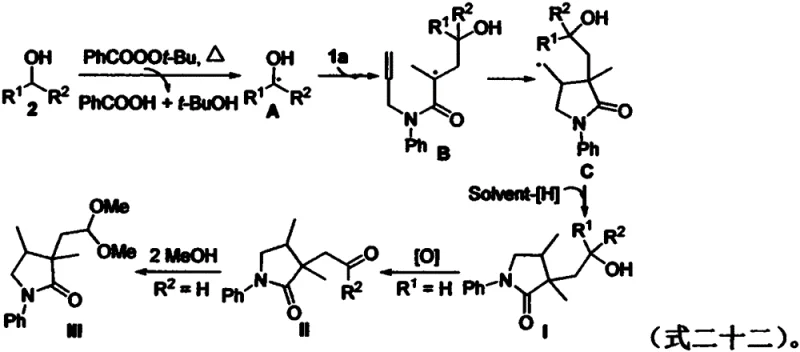
How to Synthesize Functionalized Pyrrolidinones Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and safety. The general procedure involves charging a reaction vessel with the 1,6-diene substrate, the alcohol reactant which often serves as the solvent, and the oxidant TBPB. The mixture is then heated to a moderate temperature of 80°C, which is sufficient to activate the peroxide without causing excessive decomposition or safety hazards. Monitoring the reaction progress via TLC or GC is recommended to determine the optimal endpoint, typically around 15 hours for complete conversion. Following the reaction, a straightforward workup involving ethyl acetate extraction and drying over anhydrous sodium sulfate effectively isolates the crude product, which can be purified by standard column chromatography. Detailed standardized synthesis steps are provided in the guide below.
- Charge a Schlenk flask with the 1,6-diene substrate, the chosen alcohol solvent/reactant, and tert-butyl peroxybenzoate (TBPB) as the oxidant.
- Heat the reaction mixture to 80°C under an air atmosphere and stir for approximately 15 hours until TLC monitoring indicates complete consumption of the starting material.
- Perform standard aqueous workup using ethyl acetate extraction, dry the organic phase, and purify the crude residue via column chromatography to isolate the cyclized product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this additive-free radical cyclization technology offers substantial strategic benefits that extend beyond mere chemical efficiency. The elimination of transition metal catalysts removes a significant cost center associated with both the purchase of expensive metals like palladium and the downstream processing required to scavenge them from the final product. This simplification of the purification train directly contributes to cost reduction in fine chemical manufacturing by reducing solvent consumption, filtration media usage, and overall processing time. Furthermore, the use of commodity chemicals such as simple alcohols and commercially available peroxides ensures a stable and resilient supply chain, mitigating risks associated with the sourcing of specialized reagents. The robustness of the reaction under air atmosphere further enhances supply chain reliability by reducing dependency on high-purity inert gases and specialized pressure-rated equipment.
- Cost Reduction in Manufacturing: The absence of precious metal catalysts fundamentally alters the cost structure of the synthesis, eliminating the need for expensive metal salts and the specialized ligands often required to maintain their activity. Additionally, the simplified workup procedure, which avoids complex metal scavenging resins or multiple recrystallization steps, significantly lowers the operational expenditure per kilogram of product. The high atom economy of the reaction ensures that a larger proportion of the raw material mass is incorporated into the final product, minimizing waste disposal fees and maximizing raw material utilization efficiency. These factors collectively drive down the cost of goods sold (COGS), making the final intermediates more competitive in the global market.
- Enhanced Supply Chain Reliability: By relying on widely available starting materials such as N-allyl acrylamides and common alcohols, the manufacturing process becomes less susceptible to supply disruptions that often plague specialty chemical markets. The tolerance for air atmosphere means that production does not rely on a continuous supply of nitrogen or argon, reducing logistical dependencies and infrastructure costs at the manufacturing site. This resilience is critical for maintaining consistent delivery schedules to downstream pharmaceutical clients, ensuring that project timelines are met without delay. The scalability of the process from gram to multi-kilogram scales without significant re-optimization further strengthens the supply chain's ability to respond to fluctuating demand.
- Scalability and Environmental Compliance: The green chemistry credentials of this method align perfectly with increasingly stringent environmental regulations, facilitating easier permitting and compliance reporting for manufacturing facilities. The reduction in hazardous waste streams, particularly those containing heavy metals, simplifies effluent treatment and lowers the environmental footprint of the production process. The mild reaction conditions (80°C) and the use of relatively safe oxidants enhance process safety, reducing the risk of thermal runaways and allowing for safer scale-up in existing reactor trains. This combination of safety, sustainability, and scalability makes the technology an ideal candidate for commercial scale-up of complex organic scaffolds in a regulated environment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this radical cyclization technology. These answers are derived directly from the experimental data and scope defined in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for project managers and technical leads evaluating the feasibility of integrating this route into their existing portfolios. The responses cover aspects ranging from substrate compatibility to safety considerations, ensuring a comprehensive overview for decision-makers.
Q: Does this radical cyclization process require expensive transition metal catalysts?
A: No, the method described in patent CN111233732A operates under a completely additive-free system regarding metals. It utilizes an organic peroxide oxidant (TBPB) to initiate the radical cascade, eliminating the need for costly palladium, rhodium, or copper catalysts and the subsequent rigorous purification steps required to remove trace metal residues.
Q: What is the substrate scope regarding the alcohol component in this reaction?
A: The process demonstrates excellent versatility with different alcohol classes. Secondary alcohols yield hydroxyl-functionalized pyrrolidinones, while primary alcohols (excluding methanol) are oxidized further to provide ketone-functionalized products. Uniquely, when methanol is used as the substrate, the reaction produces dimethyl acetal derivatives, showcasing distinct regioselectivity based on the alcohol structure.
Q: Is an inert atmosphere strictly required for this oxidative cyclization?
A: While the reaction can be performed under nitrogen or other inert gases, the patent explicitly highlights that an air atmosphere (1 atm) is sufficient and preferred for economic reasons. This tolerance to oxygen significantly simplifies the operational setup, removing the need for extensive degassing procedures or specialized glovebox techniques often associated with sensitive radical chemistries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,6-Diene Cyclization Product Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of additive-free radical cyclization in accelerating the development of next-generation therapeutics. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of radical chemistry, including rigorous QC labs that enforce stringent purity specifications to meet global regulatory standards. We are committed to delivering high-quality intermediates that empower your R&D teams to focus on innovation rather than supply chain constraints.
We invite you to collaborate with our technical procurement team to explore how this technology can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic benefits of switching to this metal-free protocol for your target molecules. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your unique chemical challenges. Let us help you leverage this cutting-edge chemistry to achieve your commercial goals with speed, precision, and reliability.
