Advancing Deuterated Drug Discovery: Scalable Synthesis of Selective Mono-Deuterated 1,2,3-Triazoles
The landscape of deuterated drug development has been significantly transformed by the innovations disclosed in patent CN113461624B, which introduces a highly efficient preparation method for selective mono-deuterated markers of mono-1-substituted 1,2,3-triazoles. This technology addresses a critical bottleneck in medicinal chemistry where precise isotopic labeling is required for metabolic stability studies and novel deuterated drug entities. Unlike conventional methods that often yield complex mixtures of isotopologues, this invention enables the specific installation of a single deuterium atom at either the C4 or C5 position of the triazole ring with remarkable precision. The methodology relies on a sophisticated yet operationally simple base-catalyzed exchange mechanism that functions under mild thermal conditions, typically ranging from 0°C to 80°C. For pharmaceutical manufacturers and research organizations, this represents a pivotal advancement, as it allows for the creation of high-purity reference standards and active ingredients without the cumbersome purification steps associated with separating positional isomers. The ability to control deuteration sites so accurately opens new avenues for optimizing the pharmacokinetic profiles of triazole-containing drugs, such as the widely used beta-lactamase inhibitor tazobactam.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of deuterated 1,2,3-triazoles has been dominated by variations of the Huisgen cycloaddition reaction, particularly the copper-catalyzed azide-alkyne cycloaddition (CuAAC). While effective for forming the triazole core, these traditional pathways suffer from inherent limitations regarding isotopic specificity. For instance, earlier research by Novák et al. utilized calcium carbide and heavy water to generate deuterated acetylene in situ, which subsequently reacted with azides to form 4,5-dideuterated triazoles. Similarly, other established protocols involving decarboxylative cycloadditions or hydrogen-deuterium exchange on disubstituted triazoles often result in dual labeling at both the C4 and C5 positions. This lack of selectivity is a major drawback for R&D teams who require site-specific labels to trace metabolic pathways or to invoke the kinetic isotope effect at a precise location within the molecule. Furthermore, many of these legacy methods require harsh reaction conditions, expensive transition metal catalysts that necessitate rigorous removal to meet regulatory limits, or complex multi-step sequences that reduce overall process efficiency and increase the cost of goods sold.
The Novel Approach
In stark contrast to these legacy techniques, the method described in patent CN113461624B offers a streamlined, direct route to mono-deuterated species through a controlled exchange process. By simply treating a mono-1-substituted 1,2,3-triazole precursor with a specific base additive in a deuterated or protic solvent environment, chemists can selectively target either the C4 or C5 hydrogen atom for replacement. 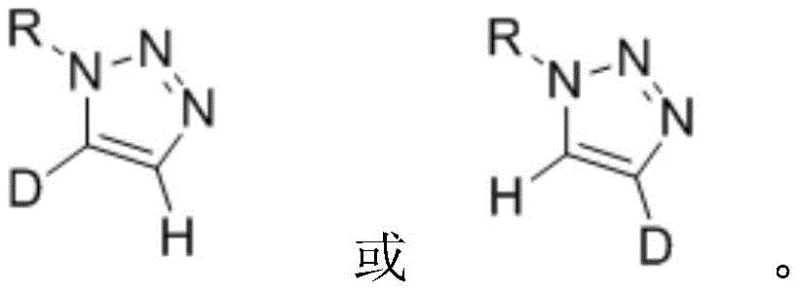 This approach eliminates the need for constructing the triazole ring from scratch with labeled building blocks, thereby saving significant synthetic effort. The versatility of this system is evident in its ability to produce both C5-deuterated markers via hydrogen-deuterium exchange and C4-deuterated markers via deuterium-hydrogen exchange starting from dideuterated precursors. The operational simplicity is further enhanced by the use of common laboratory bases such as potassium tert-butoxide or cesium carbonate, which are far more economical and easier to handle than specialized organometallic reagents. This shift from construction-based labeling to post-synthetic modification represents a paradigm shift in how deuterated heterocycles are manufactured for the life sciences industry.
This approach eliminates the need for constructing the triazole ring from scratch with labeled building blocks, thereby saving significant synthetic effort. The versatility of this system is evident in its ability to produce both C5-deuterated markers via hydrogen-deuterium exchange and C4-deuterated markers via deuterium-hydrogen exchange starting from dideuterated precursors. The operational simplicity is further enhanced by the use of common laboratory bases such as potassium tert-butoxide or cesium carbonate, which are far more economical and easier to handle than specialized organometallic reagents. This shift from construction-based labeling to post-synthetic modification represents a paradigm shift in how deuterated heterocycles are manufactured for the life sciences industry.
Mechanistic Insights into Base-Catalyzed Isotopic Exchange
The core of this technological breakthrough lies in the nuanced understanding of proton acidity within the 1,2,3-triazole ring system and how it can be manipulated by Brønsted bases. The mechanism involves the reversible deprotonation of the triazole ring at the C4 or C5 position to form a transient carbanion or stabilized anionic intermediate, which then captures a deuteron from the solvent or releases a deuteron into a protic medium. 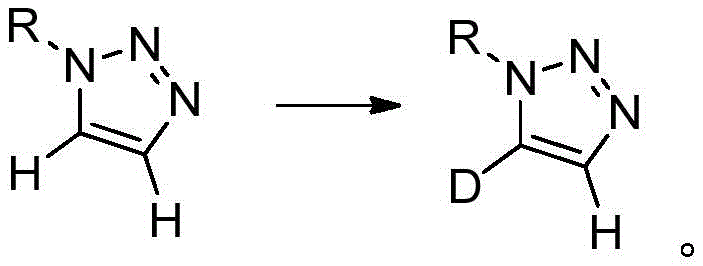 For the synthesis of C5-deuterated compounds, the reaction utilizes deuterated solvents like DMSO-d6 or mixtures containing heavy water, driving the equilibrium towards the deuterated product through mass action. Conversely, for C4-labeling, the process reverses this logic by starting with a dideuterated substrate and exposing it to a protic solvent environment with a base catalyst, effectively swapping one deuterium atom for a hydrogen atom with high regioselectivity. The choice of additive is critical; alkoxides like sodium tert-butoxide and inorganic bases like cesium carbonate act as efficient proton shuttles without promoting side reactions such as ring opening or N-alkyl migration. This mechanistic elegance ensures that sensitive functional groups on the N1 substituent, such as esters, halides, or ethers, remain intact throughout the process, preserving the structural integrity of complex drug intermediates.
For the synthesis of C5-deuterated compounds, the reaction utilizes deuterated solvents like DMSO-d6 or mixtures containing heavy water, driving the equilibrium towards the deuterated product through mass action. Conversely, for C4-labeling, the process reverses this logic by starting with a dideuterated substrate and exposing it to a protic solvent environment with a base catalyst, effectively swapping one deuterium atom for a hydrogen atom with high regioselectivity. The choice of additive is critical; alkoxides like sodium tert-butoxide and inorganic bases like cesium carbonate act as efficient proton shuttles without promoting side reactions such as ring opening or N-alkyl migration. This mechanistic elegance ensures that sensitive functional groups on the N1 substituent, such as esters, halides, or ethers, remain intact throughout the process, preserving the structural integrity of complex drug intermediates.
From an impurity control perspective, this exchange mechanism offers distinct advantages over cyclization methods. Because the triazole ring is pre-formed, there is no risk of generating regioisomeric byproducts (such as 1,4-disubstituted vs 1,5-disubstituted triazoles) that are common in cycloaddition reactions. The primary impurities are typically limited to unreacted starting material or over-exchanged species, both of which are easily managed through the described workup procedures involving extraction and column chromatography. The patent data demonstrates that by fine-tuning the molar ratio of the base additive—often keeping it as low as 0.05 to 0.2 equivalents relative to the substrate—chemists can minimize side reactions while maintaining high conversion rates. This level of control is essential for producing materials that meet the stringent purity specifications required for clinical trial applications, where the presence of unknown impurities can derail regulatory approval processes. The robustness of the mechanism across a wide temperature range (0°C to 80°C) further allows process engineers to optimize reaction kinetics without compromising selectivity.
How to Synthesize Selective Mono-Deuterated 1,2,3-Triazoles Efficiently
The practical implementation of this synthesis route is designed for ease of adoption in both laboratory and pilot plant settings. The general procedure involves dissolving the mono-1-substituted 1,2,3-triazole starting material in the chosen solvent system, adding the base catalyst under an inert atmosphere, and stirring for a defined period ranging from 1 to 8 hours.
- Dissolve the mono-1-substituted 1,2,3-triazole substrate in a suitable deuterated solvent (for C5 labeling) or non-deuterated solvent (for C4 labeling) within a reactor.
- Add a catalytic amount of base additive such as potassium tert-butoxide or cesium carbonate under a nitrogen atmosphere.
- Stir the reaction mixture at mild temperatures between 0°C and 80°C for 1 to 8 hours, followed by standard aqueous workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic benefits regarding cost structure and supply reliability. The elimination of expensive transition metal catalysts, such as copper complexes often required for click chemistry, removes a significant cost driver from the raw material bill. Moreover, the absence of heavy metals simplifies the downstream purification process, as there is no need for specialized scavenging resins or extensive washing protocols to meet residual metal specifications, which drastically reduces processing time and solvent consumption. The use of commodity chemicals like tert-butoxides and carbonates ensures that the supply chain is not vulnerable to the bottlenecks often associated with specialized reagents. This resilience is crucial for maintaining continuous production schedules for deuterated active pharmaceutical ingredients, where any interruption can have cascading effects on clinical trial timelines.
- Cost Reduction in Manufacturing: The economic model of this process is highly favorable due to the reliance on inexpensive, bulk-available base additives rather than precious metal catalysts. By avoiding the use of deuterated acetylene gas, which requires specialized handling and generation equipment, the capital expenditure for setting up production lines is significantly lowered. The high yields reported in the patent examples, often exceeding 95%, mean that raw material waste is minimized, directly improving the overall atom economy of the process. Furthermore, the mild reaction conditions reduce energy consumption for heating or cooling, contributing to lower utility costs per kilogram of produced material. These factors combine to create a cost-effective manufacturing route that makes deuterated drugs more commercially viable.
- Enhanced Supply Chain Reliability: Sourcing deuterated intermediates has historically been challenging due to the limited number of suppliers capable of performing complex isotopic labeling. This method democratizes the production capability by utilizing standard organic synthesis infrastructure. The starting materials, mono-1-substituted 1,2,3-triazoles, are widely available or easily synthesized from common precursors, reducing dependency on niche vendors. The robustness of the reaction conditions means that production is less susceptible to variability caused by minor fluctuations in temperature or reagent quality, ensuring consistent batch-to-batch quality. This reliability is paramount for long-term supply agreements where consistency is as valuable as price.
- Scalability and Environmental Compliance: Scaling isotopic labeling reactions often presents engineering challenges, but this base-catalyzed exchange is inherently scalable. The reaction does not generate hazardous gaseous byproducts or require high-pressure vessels, making it safer to operate at the 100 kg to multi-ton scale. From an environmental standpoint, the process generates less hazardous waste compared to metal-catalyzed alternatives, aligning with modern green chemistry principles. The solvents used, such as DMSO or THF, are readily recyclable, further reducing the environmental footprint. This alignment with sustainability goals is increasingly important for pharmaceutical companies aiming to reduce their Scope 3 emissions and meet corporate social responsibility targets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this selective deuteration technology. These insights are derived directly from the experimental data and claims within patent CN113461624B, providing a factual basis for decision-making.
Q: What distinguishes this mono-deuteration method from traditional click chemistry approaches?
A: Traditional copper-catalyzed azide-alkyne cycloaddition often results in 4,5-dideuterated products due to the use of deuterated acetylene. This patented method utilizes base-catalyzed exchange to achieve high selectivity for either the C4 or C5 position exclusively, avoiding mixed isotopologues.
Q: Can this process be scaled for commercial production of deuterated APIs?
A: Yes, the process operates under mild conditions (0-80°C) using commercially available bases and solvents, eliminating the need for specialized high-pressure equipment or expensive transition metal catalysts, which facilitates straightforward scale-up.
Q: What represents the typical deuteration efficiency for this technology?
A: Experimental data from the patent indicates exceptionally high deuteration rates, frequently exceeding 95% and reaching up to 100% in optimized examples, ensuring minimal isotopic dilution in the final active pharmaceutical ingredient.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mono-1-substituted 1,2,3-triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the selective mono-deuteration technology described in patent CN113461624B for the next generation of deuterated therapeutics. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to market supply is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying isotopic purity and structural integrity, adhering to the most stringent purity specifications required by global regulatory bodies. We understand that the successful commercialization of deuterated drugs depends not just on the chemistry, but on a partner who can guarantee supply continuity and quality consistency at every stage of development.
We invite you to collaborate with our technical procurement team to explore how this innovative synthesis route can be integrated into your pipeline. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your target molecules. We encourage you to reach out for specific COA data and route feasibility assessments tailored to your project needs. Whether you require small quantities for preclinical studies or large-scale volumes for commercial launch, our team is ready to provide the expertise and capacity necessary to bring your deuterated drug candidates to market efficiently.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
