Advanced 2,3'-Bisfuran Synthesis: Enabling Scalable Production of High-Performance Fluorescent Materials
The pharmaceutical and electronic chemical industries are constantly seeking advanced heterocyclic building blocks that offer superior optical properties and structural versatility. Patent CN115073401B introduces a groundbreaking methodology for the preparation of 2,3'-bisfuran compounds and their subsequent oligomerization into 2,3'-oligofuran derivatives. This technology addresses a critical gap in the market, as previous synthetic routes predominantly focused on symmetrical 2,2'- or 3,3'-oligofurans, leaving the asymmetric 2,3'-configurations largely unexplored despite their significant potential in optical applications. The disclosed invention provides a robust platform for generating novel fluorescent materials that exhibit excellent performance in fields ranging from hydrological detection to biological tracking. By leveraging a transition metal-catalyzed cycloisomerization strategy, this patent enables the efficient construction of complex furan architectures that were previously difficult to access with high purity and yield.
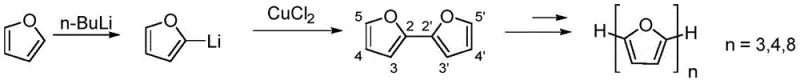
For R&D directors and procurement specialists, understanding the shift from legacy methods to this novel approach is essential for evaluating supply chain resilience and cost structures. Conventional methods for synthesizing oligomeric furans often relied on Ullmann-type couplings or similar cross-coupling reactions that required stoichiometric amounts of copper or harsh reaction conditions. These traditional pathways frequently suffered from limited functional group tolerance and produced symmetrical structures that lacked the specific electronic tuning capabilities offered by asymmetric 2,3'-linkages. The limitations of these older technologies often resulted in extended development timelines and higher purification costs, creating bottlenecks in the manufacturing of high-purity OLED material precursors and specialty chemical intermediates. In contrast, the new methodology streamlines the synthesis, offering a more direct route to diverse structural variants.
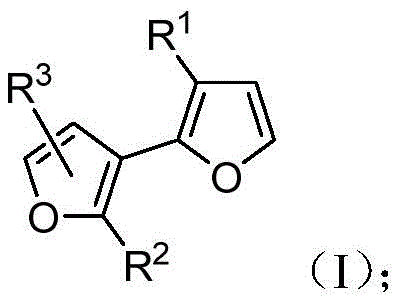
The novel approach detailed in the patent utilizes a platinum-catalyzed cycloisomerization of alkynylaldehyde precursors to construct the 2,3'-bisfuran core with remarkable efficiency. This transformation proceeds under mild thermal conditions, typically between 40°C and 60°C, which significantly reduces the energy footprint compared to high-temperature alternatives. The reaction demonstrates broad substrate scope, accommodating various aryl and alkyl substituents, which allows for the fine-tuning of electronic and steric properties essential for specific electronic chemical manufacturing requirements. Furthermore, the inclusion of protonating additives such as isopropanol enhances the reaction yield and selectivity, ensuring that the final product meets stringent purity specifications without the need for extensive downstream processing. This level of control is vital for producing reliable agrochemical intermediate or pharmaceutical building blocks where impurity profiles must be tightly managed.
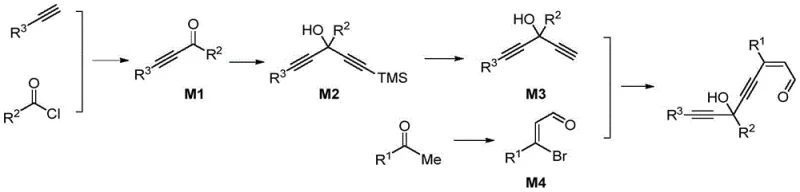
Mechanistically, the synthesis begins with the preparation of a specialized alkynylaldehyde intermediate, which serves as the key precursor for the cyclization event. As illustrated in the reaction schemes, this precursor is assembled through a sequence involving Sonogashira coupling and nucleophilic addition, allowing for the precise installation of diverse substituents at the R1, R2, and R3 positions. The subsequent platinum-catalyzed step involves the activation of the alkyne moiety, facilitating an intramolecular attack by the oxygen atom to close the furan ring. This cascade process is highly atom-economical and minimizes the generation of waste byproducts, aligning with modern green chemistry principles. The ability to introduce trialkylsilyl groups or various aryl rings at specific positions provides chemists with a powerful toolkit for designing molecules with tailored fluorescence emission wavelengths and quantum yields.
How to Synthesize 2,3'-Bisfuran Compound Efficiently
The standardized synthesis protocol involves the careful preparation of the alkynylaldehyde precursor followed by the critical cyclization step under inert atmosphere. Detailed operational parameters regarding solvent selection, catalyst loading, and temperature control are crucial for reproducing the high yields reported in the patent examples. For a comprehensive breakdown of the specific reagents, molar ratios, and workup procedures required to achieve optimal results, please refer to the standardized guide below which outlines the exact experimental conditions validated for commercial feasibility.
From a commercial perspective, the adoption of this synthesis route offers substantial advantages for procurement and supply chain teams managing the sourcing of complex heterocyclic intermediates. The elimination of harsh reaction conditions translates directly into enhanced supply chain reliability, as the process can be safely scaled from laboratory benchtop to multi-ton production without requiring specialized high-pressure equipment. The use of commercially available starting materials, such as substituted aromatic ketones and acid chlorides, ensures that raw material availability remains stable, reducing the risk of production delays caused by scarce reagents. Additionally, the short synthetic route minimizes the number of unit operations, which inherently lowers the operational expenditure associated with labor, energy, and solvent consumption during cost reduction in electronic chemical manufacturing.
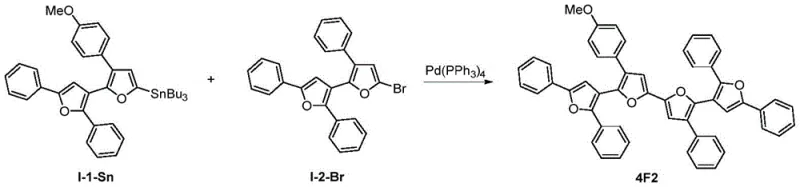
Scalability and environmental compliance are further strengthened by the method's compatibility with standard purification techniques like column chromatography and crystallization. The high selectivity of the platinum catalyst reduces the formation of regioisomers, simplifying the isolation of the target 2,3'-bisfuran compound and reducing solvent waste associated with repeated purification cycles. This efficiency supports the commercial scale-up of complex polymer additives and fluorescent probes, enabling manufacturers to meet increasing demand for high-performance optical materials. By reducing lead time for high-purity 2,3'-oligofurans, companies can accelerate their time-to-market for new products in the display and sensor industries, securing a competitive edge in rapidly evolving technology sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology in industrial settings. These answers are derived directly from the technical disclosures and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this synthesis pathway for their own production lines.
Q: What are the advantages of the new 2,3'-bisfuran synthesis over conventional Ullmann reactions?
A: The new method utilizes mild platinum-catalyzed cycloisomerization conditions (40-60°C) compared to the harsh conditions of traditional Ullmann couplings, resulting in significantly reduced energy consumption and improved safety profiles for commercial manufacturing.
Q: Can these 2,3'-oligofuran compounds be used in biological tracking?
A: Yes, the patent explicitly highlights the potential application of these compounds as fluorescent materials in biological tracking, hydrological detection, and non-destructive flaw detection due to their excellent fluorescence properties and large Stokes shifts.
Q: Is the synthesis route scalable for industrial production?
A: The patent describes the method as having simple operation, mild conditions, and a short route, indicating strong potential for industrialization and commercial scale-up of complex heterocyclic intermediates without requiring extreme pressure or temperature.
- Prepare the alkynylaldehyde precursor (Formula II) via Sonogashira coupling and nucleophilic addition reactions.
- Perform platinum-catalyzed cycloisomerization in toluene with isopropanol additive at 60°C to form the 2,3'-bisfuran core.
- Optional: Conduct Stille coupling or oxidative coupling to polymerize the bisfuran monomers into 2,3'-oligofurans for optical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3'-Bisfuran Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the 2,3'-bisfuran synthesis technology in advancing the field of optoelectronic materials and pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless and efficient. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2,3'-bisfuran compound or oligofuran derivative meets the highest industry standards for consistency and quality.
We invite you to collaborate with our technical team to explore how this innovative chemistry can optimize your product portfolio and reduce overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can support your supply chain goals and drive value for your organization.
