Scalable Synthesis of 3-Substituted Benzofluorenone Derivatives for Advanced Material Applications
Introduction to Advanced Fused Ring Synthesis
The development of efficient synthetic routes for complex polycyclic aromatic hydrocarbons is a cornerstone of modern material science and pharmaceutical research. Patent CN113336784B, published in late 2022, introduces a groundbreaking methodology for constructing 3-substituted benzofluorenone and naphthofluorenone derivatives. These molecular scaffolds are increasingly recognized for their exceptional optoelectronic properties, making them prime candidates for next-generation organic light-emitting diodes (OLEDs) and as critical intermediates in the synthesis of bioactive agents targeting nicotinic acetylcholine receptors. The innovation lies in a streamlined catalytic cycle that bypasses traditional limitations, offering a reliable pathway for producing high-purity electronic chemical intermediates with significant potential for commercial scalability.
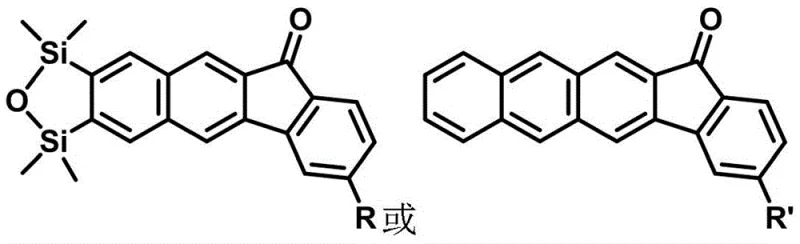
The core innovation described in this patent centers on the utilization of oxabenzobicycloalkenes and oxanaphthobicycloalkenes as versatile building blocks. Unlike conventional methods that often rely on harsh conditions or multiple protection-deprotection sequences, this approach leverages the unique reactivity of the oxabicycloalkene moiety. The resulting compounds exhibit tunable fluorescence quantum yields and robust thermal stability, characteristics that are essential for reliable organic luminescent material supplier operations. By establishing a direct link between simple precursors and highly functionalized fused rings, this technology addresses the growing demand for cost reduction in electronic chemical manufacturing while maintaining rigorous quality standards required by top-tier multinational corporations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzofluorenone skeletons has been plagued by inefficiencies inherent to classical electrophilic aromatic substitution and multi-step functionalization strategies. Traditional routes often necessitate the use of pre-halogenated starting materials, which increases raw material costs and generates significant stoichiometric waste. Furthermore, these legacy processes frequently require aggressive Lewis acids or extreme temperatures that can compromise the integrity of sensitive functional groups, leading to complex impurity profiles that are difficult to remediate. For procurement managers, these factors translate into volatile supply chains and unpredictable lead times, as the purification of crude reaction mixtures often becomes a bottleneck. The reliance on such archaic methodologies limits the ability to rapidly scale production to meet the surging demands of the display and pharmaceutical sectors.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent employs a sophisticated nickel-catalyzed C-H activation strategy that fundamentally reshapes the synthetic landscape. By directly activating inert carbon-hydrogen bonds, this process eliminates the need for pre-functionalized halides, thereby streamlining the synthetic sequence and enhancing overall atom economy. The reaction proceeds under relatively mild thermal conditions, typically between 120°C and 130°C in a sealed tube, which significantly reduces energy consumption and operational hazards. This modern approach not only simplifies the workflow but also ensures a cleaner reaction profile, facilitating easier downstream processing. For supply chain heads, this translates to enhanced supply chain reliability, as the robustness of the catalytic system minimizes batch-to-batch variability and ensures consistent output of high-value intermediates.
Mechanistic Insights into Ni-Catalyzed C-H Activation and Cyclization
The mechanistic elegance of this synthesis relies on a synergistic catalytic system comprising nickel trifluoromethanesulfonate, silver acetate, and a specialized bulky imidazole ligand. The cycle initiates with the coordination of the nickel catalyst to the quinoline directing group of the benzamide substrate, facilitating the selective cleavage of the ortho C-H bond. This is followed by the insertion of the oxabicycloalkene olefin into the nickel-carbon bond, a critical step that constructs the new carbon-carbon framework with high regioselectivity. Subsequent intramolecular nucleophilic attack by the oxygen atom closes the ring, forming the tetrahydroepoxy intermediate. This cascade of transformations occurs with remarkable efficiency, demonstrating the power of modern organometallic chemistry to construct complex architectures from simple precursors in a single operational step.
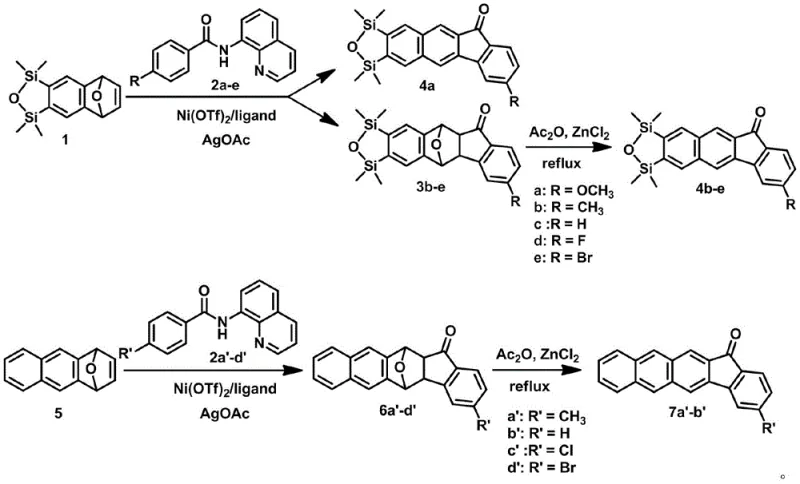
Following the initial cyclization, the intermediate undergoes a deoxyaromatization reaction driven by zinc chloride and acetic anhydride. This second stage is crucial for restoring the aromaticity of the central ring system, yielding the final planar benzofluorenone or naphthofluorenone structure. The removal of the oxygen bridge is achieved under mild heating (80-85°C), preserving the integrity of other substituents such as methoxy, methyl, or halogen groups. From an R&D perspective, this two-stage protocol offers exceptional control over the impurity spectrum. The specificity of the nickel catalyst minimizes side reactions, while the distinct conditions of the aromatization step allow for the selective removal of byproducts. This level of control is vital for achieving the stringent purity specifications demanded by high-performance electronic applications.
How to Synthesize 3-Substituted Benzofluorenone Efficiently
The practical execution of this synthesis involves a carefully orchestrated sequence of reagent addition and thermal management to maximize yield and purity. The process begins with the combination of the oxabicycloalkene and the N-(quinolin-8-yl)benzamide derivative in a toluene solvent system, supplemented by the nickel catalyst, silver oxidant, and ligand. The mixture is heated in a sealed vessel to maintain pressure and prevent solvent loss, ensuring the reaction proceeds to completion over a 20 to 24-hour period. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results effectively.
- React oxabenzobicycloalkene with N-(quinolin-8-yl)-4-substituted benzamide using Ni(OTf)2, AgOAc, and imidazole ligand in toluene at 120-130°C.
- Isolate the cycloaddition intermediate via filtration and column chromatography to obtain the tetrahydroepoxy derivative.
- Perform deoxyaromatization by reacting the intermediate with ZnCl2 and acetic anhydride at 80-85°C to yield the final benzofluorenone product.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this advanced synthetic route offers profound strategic benefits for organizations focused on optimizing their chemical supply chains and reducing overall manufacturing expenditures. The elimination of pre-functionalized halide starting materials represents a significant shift in raw material sourcing, allowing procurement teams to access cheaper and more abundant commodity chemicals. Furthermore, the simplified workup procedures, which involve basic filtration and chromatography rather than complex extraction sequences, reduce the consumption of solvents and consumables. These operational efficiencies culminate in substantial cost savings, making the production of these high-value intermediates economically viable on a commercial scale without compromising on quality or performance metrics.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the direct C-H activation strategy, which removes the necessity for expensive halogenated precursors and the waste associated with their preparation. By utilizing nickel catalysis, the process achieves high turnover numbers, meaning less precious metal is required per unit of product. Additionally, the mild reaction conditions reduce energy overheads related to heating and cooling, while the high yields (often exceeding 75%) minimize material loss. This holistic reduction in input costs allows for a more competitive pricing structure for the final active pharmaceutical ingredients or electronic materials derived from these scaffolds.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of robust and commercially available starting materials such as oxabenzobicycloalkenes and substituted benzamides. Unlike specialized reagents that may have single-source suppliers, these precursors are accessible from multiple global vendors, mitigating the risk of shortages. The resilience of the catalytic system against minor variations in reaction parameters further ensures consistent batch quality, reducing the likelihood of production delays due to failed runs. This reliability is critical for maintaining uninterrupted production schedules in fast-moving industries like consumer electronics and pharmaceuticals.
- Scalability and Environmental Compliance: The transition from laboratory to industrial scale is facilitated by the absence of extreme conditions; the reaction operates at atmospheric pressure equivalents and moderate temperatures, removing the need for specialized high-pressure reactors. The environmental footprint is also favorably impacted by the higher atom economy and reduced solvent usage compared to traditional multi-step syntheses. Waste streams are simpler to manage, primarily consisting of organic residues that can be treated through standard incineration or recovery protocols. This alignment with green chemistry principles supports corporate sustainability goals and simplifies regulatory compliance for environmental discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on performance expectations and operational requirements. Understanding these details is essential for technical directors and procurement specialists evaluating the feasibility of integrating these derivatives into their existing product portfolios or supply networks.
Q: What are the key advantages of this Ni-catalyzed synthesis over traditional methods?
A: This method utilizes direct C-H bond activation, eliminating the need for pre-functionalized halide starting materials. This results in fewer synthetic steps, milder reaction conditions (120-130°C), and improved atom economy compared to classical Friedel-Crafts approaches.
Q: What is the expected purity and yield for these fused ring compounds?
A: The patent data indicates robust yields ranging from 67% to 82% across various substituents. The process includes column chromatography purification steps, ensuring high-purity solids suitable for sensitive electronic or pharmaceutical applications.
Q: Are these compounds suitable for large-scale commercial production?
A: Yes, the reaction conditions are mild and do not require extreme cryogenic temperatures or hazardous high-pressure equipment. The use of stable catalysts and readily available starting materials supports scalable manufacturing from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Substituted Benzofluorenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this nickel-catalyzed technology in advancing the fields of optoelectronics and medicinal chemistry. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from pilot studies to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of benzofluorenone or naphthofluorenone derivative meets the exacting standards required for high-performance applications. We are committed to delivering consistency and quality that empowers your innovation.
We invite you to engage with our technical procurement team to discuss how this novel synthetic route can be tailored to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to validate the performance of these materials in your own systems with confidence. Let us collaborate to bring these advanced materials to market efficiently and effectively.
