Revolutionizing Ketene Ketal Production: A One-Step Copper-Catalyzed Strategy for Pharmaceutical Intermediates
Introduction to Advanced Ketene Ketal Synthesis
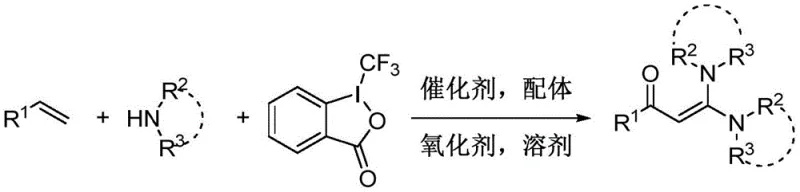
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more efficient, sustainable, and cost-effective methodologies. A significant breakthrough in this domain is documented in patent CN107628960B, which discloses a novel one-step method for synthesizing ketene ketals directly from olefins. This technology represents a paradigm shift from traditional multi-step protocols, offering a streamlined pathway to access highly valuable enaminone scaffolds that are pivotal in medicinal chemistry and organic synthesis. By leveraging a copper-catalyzed oxidative three-component reaction involving olefins, amines, and Togni reagent, this process achieves high atom economy and operational simplicity. For R&D directors and procurement specialists alike, understanding the nuances of this patented approach is crucial for optimizing supply chains and reducing the cost of goods sold (COGS) for complex pharmaceutical intermediates.
Ketene ketals, characterized by their unique electronic structure featuring both electron-withdrawing carbonyl and electron-donating amino groups, serve as versatile building blocks for constructing heterocyclic systems with potent biological activities. The ability to synthesize these compounds in a single pot from readily available starting materials eliminates the need for hazardous reagents and harsh conditions often associated with legacy methods. This patent not only addresses the synthetic challenges but also aligns with modern green chemistry principles by utilizing mild reaction temperatures and commercially accessible catalysts. As we delve deeper into the technical specifics, it becomes evident how this innovation can be leveraged to enhance production capabilities and ensure a reliable supply of high-purity intermediates for the global pharmaceutical market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ketene ketals has been fraught with significant challenges that hinder industrial scalability and economic efficiency. Traditional routes often rely on the reaction of α-carbonyl dithioketals with amines, a process that necessitates the preparation of sulfur-containing precursors which can be malodorous, toxic, and difficult to handle on a large scale. Another common approach involves the reaction of diaminoethylene with acid chlorides; however, this method suffers from limited substrate scope and requires strictly anhydrous conditions to prevent hydrolysis of the sensitive acid chloride intermediates. Furthermore, existing methodologies frequently employ harsh reaction conditions, including strong bases or elevated temperatures, which can lead to the decomposition of sensitive functional groups and result in lower overall yields. These limitations translate directly into higher production costs, increased waste generation, and prolonged lead times, creating bottlenecks for procurement teams striving to maintain lean inventory levels.
The Novel Approach
In stark contrast, the methodology outlined in patent CN107628960B introduces a transformative strategy that utilizes simple olefins and amines as fundamental raw materials. This one-step oxidative amination process bypasses the need for pre-functionalized sulfur or acid chloride intermediates, thereby drastically simplifying the synthetic route. The reaction employs Togni reagent (1-trifluoromethyl-1,2-benziodoxol-3(1H)-one) as a key oxidant and trifluoromethyl source, working in tandem with a copper catalyst to activate the olefinic double bond. This approach operates under mild conditions, typically between 30°C and 80°C, and demonstrates exceptional tolerance for a wide array of functional groups, including halogens, nitriles, and heterocycles. By shifting to this direct functionalization strategy, manufacturers can achieve substantial cost reduction in pharma intermediate manufacturing through reduced raw material complexity and simplified downstream processing.
Mechanistic Insights into Copper-Catalyzed Oxidative Functionalization
The core of this innovative synthesis lies in the intricate interplay between the copper catalyst, the ligand, and the hypervalent iodine oxidant. The reaction mechanism initiates with the coordination of the copper species to the olefin and the amine, facilitating the activation of the carbon-carbon double bond. The Togni reagent acts as a powerful oxidant, enabling the oxidative cleavage or functionalization necessary to install the carbonyl moiety while simultaneously incorporating the amine nucleophiles. The presence of nitrogen- or phosphorus-containing ligands, such as 1,10-phenanthroline derivatives or triphenylphosphine, is critical for stabilizing the copper center and modulating its redox potential, ensuring high catalytic turnover and selectivity. This catalytic cycle effectively transforms simple alkenes into highly polarized enaminone systems without the formation of excessive byproducts, showcasing a high level of chemoselectivity that is essential for producing high-purity API intermediates.
Furthermore, the mechanistic pathway ensures robust impurity control, a key concern for quality assurance teams. The mild oxidative conditions prevent the over-oxidation of sensitive substrates, which is a common pitfall in traditional strong oxidant protocols. The use of specific ligand-copper complexes helps to direct the regioselectivity of the amine addition, minimizing the formation of regioisomers that would otherwise complicate purification. The reaction tolerates a broad spectrum of substituents on the aromatic ring of the olefin, including electron-withdrawing groups like cyano and chloro, as well as electron-donating groups like methyl and tert-butyl. This versatility implies that a single standardized protocol can be adapted for a diverse library of analogs, streamlining the R&D process for new drug candidates and reducing the time required for process optimization.
How to Synthesize Ketene Ketals Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and purity. The general procedure involves dissolving the olefin, amine, Togni reagent, and an oxidant (such as di-tert-butyl peroxide or oxygen) in a polar aprotic solvent like acetonitrile or dimethyl sulfoxide. Following this, the copper catalyst and ligand are introduced, and the mixture is heated with stirring. The detailed standardized synthesis steps, including specific molar ratios and workup procedures validated by the patent data, are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Dissolve the olefin substrate, amine nucleophile, Togni reagent, and oxidant in a suitable organic solvent such as acetonitrile or DMF.
- Add the copper catalyst (e.g., copper sulfide or copper acetate) and the appropriate ligand (e.g., phenanthroline derivatives) to the reaction mixture.
- Heat the mixture to 30-80°C with stirring for 1-10 hours, then cool, extract with ethyl acetate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this one-step olefin functionalization technology offers compelling advantages for supply chain resilience and cost management. By utilizing olefins and amines as starting materials, which are commodity chemicals available in bulk quantities from multiple global suppliers, the dependency on niche, high-cost precursors is significantly reduced. This shift mitigates supply risk and enhances the stability of the raw material supply chain, ensuring continuous production even during market fluctuations. Moreover, the elimination of sulfur-based reagents and acid chlorides simplifies waste treatment protocols, leading to substantial cost savings in environmental compliance and disposal fees. The operational simplicity of the process, requiring only standard heating and stirring equipment, facilitates easy technology transfer from lab to plant, accelerating time-to-market for new products.
- Cost Reduction in Manufacturing: The transition to this catalytic method eliminates the need for expensive, multi-step precursor synthesis, directly lowering the cost of goods. By avoiding the use of hazardous acid chlorides and malodorous dithioketals, facilities can reduce expenditures on specialized containment systems and personal protective equipment. The high atom economy of the three-component reaction ensures that a greater proportion of raw materials end up in the final product, minimizing waste and maximizing resource efficiency. Additionally, the mild reaction conditions reduce energy consumption compared to high-temperature or high-pressure alternatives, contributing to a lower overall carbon footprint and operational expenditure.
- Enhanced Supply Chain Reliability: Sourcing olefins and simple amines is far more straightforward than procuring specialized alpha-carbonyl dithioketals, which may have limited suppliers and long lead times. This abundance of feedstock allows procurement managers to negotiate better pricing and secure long-term contracts, insulating the production schedule from raw material shortages. The robustness of the reaction across various substrates means that alternative starting materials can be quickly substituted if a specific olefin becomes unavailable, providing a flexible contingency plan for supply chain disruptions. This flexibility is critical for maintaining uninterrupted delivery schedules to downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently with standard organic solvents and common copper salts that are easily sourced in metric ton quantities. The absence of heavy metal contaminants beyond trace copper, which can be removed via standard aqueous workup and chromatography, simplifies the purification process and ensures the final product meets stringent purity specifications. The use of less toxic reagents aligns with increasingly strict environmental regulations, reducing the regulatory burden and potential liabilities associated with hazardous chemical handling. This compliance advantage positions manufacturers as preferred partners for eco-conscious pharmaceutical companies seeking sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on reaction parameters and substrate compatibility. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing workflows.
Q: What are the optimal reaction conditions for this ketene ketal synthesis?
A: The reaction typically proceeds best at temperatures between 30°C and 80°C for 1 to 10 hours, using solvents like acetonitrile or DMSO with copper salts and phenanthroline ligands.
Q: Can this method tolerate diverse functional groups on the olefin substrate?
A: Yes, the protocol demonstrates excellent functional group compatibility, successfully accommodating substituents such as cyano, chloro, methyl, and thiazole groups on the aromatic ring.
Q: Is the purification process complex for large-scale production?
A: The workup involves standard aqueous washing and ethyl acetate extraction followed by column chromatography, which is a scalable and routine purification technique in fine chemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ketene Ketal Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN107628960B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of ketene ketal or related intermediate meets the exacting standards required for drug development and manufacturing. We are committed to leveraging cutting-edge chemistry to drive innovation and efficiency for our global partners.
We invite you to collaborate with us to explore how this efficient synthesis route can optimize your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how switching to this methodology can impact your bottom line. Please contact us today to request specific COA data for our available intermediates and to discuss route feasibility assessments for your target molecules. Let us be your trusted partner in navigating the complexities of fine chemical synthesis and securing a competitive edge in the marketplace.
