Advanced Synthetic Strategy for Azaoxindole Intermediates Enabling Commercial Scale-Up
Advanced Synthetic Strategy for Azaoxindole Intermediates Enabling Commercial Scale-Up
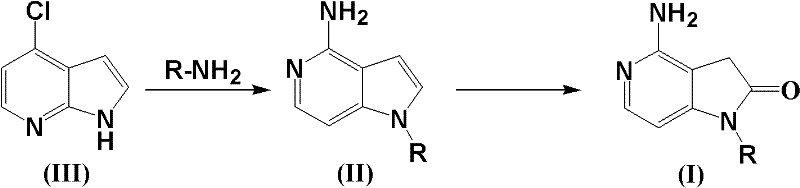
The landscape of oncology drug development is continuously evolving, driven by the urgent need for more effective tyrosine kinase inhibitors that offer higher selectivity and reduced toxicity profiles. A pivotal breakthrough in this domain is documented in patent CN102140097A, which introduces a novel class of azaindole ketone compounds and their efficient synthetic methodologies. These structures serve as critical scaffolds for mimicking the ATP-binding domains of growth factor receptors, similar to the mechanism observed in established drugs like Sunitinib. For R&D directors and procurement specialists alike, understanding the nuances of this synthesis is paramount, as it addresses a significant gap in the availability of 4-amino substituted azaindole ketones. The patent outlines a streamlined pathway that transforms readily available chlorinated precursors into high-value intermediates through a clever sequence of rearrangement, oxidation, and reduction. This technical insight report delves deep into the mechanistic advantages and commercial viability of this process, positioning it as a cornerstone for reliable pharmaceutical intermediate supplier strategies in the competitive oncology market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of azaindole derivatives, particularly those substituted at the 4-position with amino groups, has presented formidable challenges to organic chemists. While literature precedents exist for 4, 6, or 7-azaindole ketones, the specific construction of the 4-amino variant has remained largely unexplored and technically demanding. Conventional routes often suffer from苛刻 reaction conditions, requiring extreme temperatures or pressures that compromise safety and scalability. Furthermore, traditional methods frequently rely on expensive transition metal catalysts or hazardous reagents that introduce complex impurity profiles, necessitating rigorous and costly purification steps. The lack of a generalized, mild method for introducing nitrogen atoms into the relative reactivity positions of the indole skeleton has hindered the rapid exploration of structure-activity relationships (SAR) for new antineoplastic agents. Consequently, the supply chain for these specific intermediates has been fragmented, leading to extended lead times and inconsistent quality for downstream drug manufacturers seeking to optimize their pipelines.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by utilizing 7-chloroazaoxindole as a versatile starting material, effectively bypassing the limitations of previous syntheses. This innovative route leverages a thermal rearrangement and substitution reaction that proceeds efficiently even under solvent-free conditions, drastically reducing the environmental footprint and operational complexity. As illustrated in the specific embodiment below, the reaction of 7-chloroazaoxindole with benzylamine yields the intermediate 4-amino-1-benzyl-1H-pyrrolo[3,2-c]pyridine with impressive efficiency. The subsequent transformation involves a mild oxidation-reduction sequence that constructs the final ketone functionality without degrading the sensitive amino group. This approach not only simplifies the operational workflow but also enhances the overall atom economy of the process. By avoiding the need for protecting groups and utilizing benign reagents, this novel approach offers a robust platform for the commercial scale-up of complex pharmaceutical intermediates, ensuring a steady supply of high-purity materials for clinical and commercial production.
![Specific synthetic embodiment showing the conversion of 7-chloroazaoxindole to 4-amino-1-benzyl-1H-pyrrolo[3,2-c]pyridine-2(3H)-one](/insights/img/azaoxindole-synthesis-pharma-intermediate-supplier-20260306191402-04.png)
Mechanistic Insights into Thermal Rearrangement and Redox Cyclization
The core of this synthetic innovation lies in the initial thermal rearrangement of the 7-chloroazaoxindole scaffold. When subjected to temperatures between 180-200°C in the presence of a primary or secondary amine, the chlorine atom acts as a leaving group, facilitating a nucleophilic aromatic substitution that is concomitant with a skeletal rearrangement. This step is critical as it establishes the nitrogen substitution pattern required for biological activity. The absence of solvent in this stage is particularly noteworthy; it forces the reactants into close proximity, driving the equilibrium towards the desired product while eliminating the need for solvent recovery systems. Following this, the intermediate undergoes a sophisticated oxidation-reduction cascade. The use of pyridinium tribromide as a mild oxidant allows for the controlled activation of the pyrrole ring without over-oxidation, which is a common pitfall in heterocyclic chemistry. This is immediately followed by reduction using zinc powder, which quenches the reactive intermediates and stabilizes the final azaoxindole structure. This redox pair ensures that the reaction proceeds with high chemoselectivity, preserving other functional groups that might be present on the amine substituent.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed cross-coupling reactions. Traditional palladium or copper-catalyzed methods often leave behind trace metal residues that are difficult to remove and strictly regulated in final drug substances. In contrast, the reagents used here—pyridinium tribromide and zinc—are either organic salts or base metals that can be easily removed via aqueous workup and filtration. The patent specifies that the reaction mixture can be simply filtered to remove excess zinc, followed by standard extraction protocols. This inherent cleanliness of the reaction profile translates directly to lower processing costs and higher final purity. For R&D teams, this means that the impurity spectrum is predictable and manageable, reducing the risk of late-stage failures due to genotoxic impurities or heavy metal contamination. The ability to tune the R-group on the amine allows for the generation of diverse libraries of analogues, all accessible through this single, robust mechanistic pathway.
How to Synthesize 4-Aminoazaindole Ketones Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the thermal parameters of the initial rearrangement step and the stoichiometry of the redox reagents. The process begins by mixing the 7-chloroazaoxindole starting material with the chosen amine, ensuring a slight excess of the amine to drive the substitution to completion. The mixture is then heated, typically in an oil bath, to maintain the requisite temperature range of 180-200°C for a period of 10 to 24 hours. Once the intermediate is formed and isolated, it is dissolved in a mixture of tert-butanol and water, creating a biphasic system that supports the subsequent redox chemistry. The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined in the guide below.
- React 7-chloroazaoxindole with a corresponding amine under solvent-free conditions at 180-200°C to effect rearrangement and substitution.
- Dissolve the intermediate in tert-butanol and water, then add an oxidant such as pyridinium tribromide at low temperature (-10 to 10°C).
- Add a reducing agent like zinc powder to the mixture to complete the cyclization and obtain the final azaoxindole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the manufacturing process, which directly correlates to cost reduction in pharma manufacturing. By eliminating the need for expensive noble metal catalysts and complex solvent systems in the initial step, the raw material costs are significantly lowered. Furthermore, the solvent-free nature of the first reaction step reduces the volume of waste solvents generated, thereby lowering disposal costs and environmental compliance burdens. The use of common, commodity chemicals like zinc powder and pyridinium tribromide ensures that the supply chain for reagents is stable and resilient against market fluctuations. This reliability is crucial for maintaining continuous production schedules and meeting the stringent delivery timelines required by global pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for expensive scavenging resins or complex purification columns designed to reduce metal content to ppm levels. Additionally, the solvent-free initial step significantly cuts down on energy consumption associated with heating large volumes of solvent and the subsequent distillation for recovery. The overall process intensity is reduced, allowing for higher throughput in existing reactor vessels without the need for capital-intensive equipment upgrades. These factors combine to create a leaner, more cost-effective production model that maximizes margin potential for high-volume intermediates.
- Enhanced Supply Chain Reliability: The starting material, 7-chloroazaoxindole, is derived from abundant feedstocks, ensuring a secure upstream supply base. Unlike specialized reagents that may have single-source suppliers, the reagents used in the oxidation-reduction steps are widely available from multiple global vendors. This diversification of the supply base mitigates the risk of shortages and price spikes. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, further enhancing the stability of the supply chain and reducing the rate of batch failures.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-ton commercial production. The workup procedures involve simple filtration and liquid-liquid extraction, operations that are easily automated and scaled in industrial settings. From an environmental standpoint, the avoidance of halogenated solvents in the main reaction steps and the use of water as a co-solvent align with green chemistry principles. This facilitates easier regulatory approval and reduces the carbon footprint of the manufacturing process, a key metric for modern sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these azaoxindole intermediates. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this technology into your current development pipeline.
Q: What is the key starting material for this azaoxindole synthesis?
A: The process utilizes 7-chloroazaoxindole as the primary raw material, which undergoes a rearrangement reaction with various amines to form the core scaffold.
Q: How does this method improve upon traditional heterocycle synthesis?
A: Unlike conventional methods that may require harsh conditions or complex multi-step protections, this route employs a mild oxidation-reduction sequence using pyridinium tribromide and zinc powder, significantly simplifying the workflow.
Q: What yields can be expected from this synthetic route?
A: Experimental data indicates robust performance, with specific embodiments demonstrating isolated yields ranging from 74% to 82% for the final purified products after chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azaoxindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of oncology drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your demands whether you are in the early discovery phase or preparing for market launch. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of azaoxindole intermediate meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of heterocyclic chemistry, delivering materials that accelerate your timeline to clinic.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce overall project costs.
