Scalable Production of High-Purity Strobilurin Intermediates via Novel Methylation
Introduction to Advanced Agrochemical Intermediate Synthesis
The global demand for high-efficiency, low-toxicity agricultural fungicides continues to drive innovation in intermediate synthesis, particularly for the strobilurin class which includes market leaders like Kresoxim-methyl and Trifloxystrobin. As detailed in the recent patent CN115417793A, a significant technological breakthrough has been achieved in the preparation of the critical precursor, (E)-2-methyl-α-methoxyiminophenylacetic acid methyl ester. This compound serves as the foundational building block for these high-value active ingredients, and its efficient production is paramount for maintaining a robust supply chain in the agrochemical sector. The patent outlines a novel methodology that addresses long-standing challenges regarding stereoselectivity and process complexity, offering a pathway that is not only chemically elegant but also commercially viable for large-scale manufacturing.
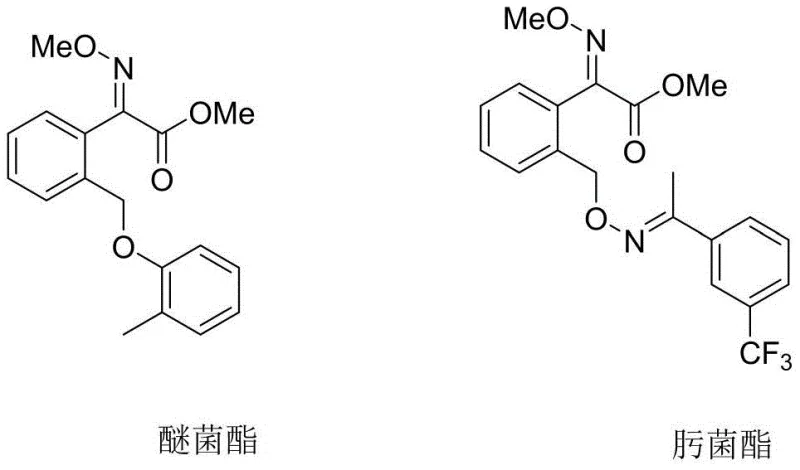
Understanding the structural relationship between the intermediate and the final active pharmaceutical ingredients (APIs) is crucial for R&D directors evaluating process feasibility. The intermediate features a specific E-configured oxime ether moiety attached to a methylated phenylacetic acid backbone, a structural motif essential for biological activity against fungal pathogens. Traditional manufacturing approaches have often struggled to maintain this specific stereochemistry while keeping costs low, leading to yield losses and extensive purification requirements. The new method described in the patent leverages a telescoped strategy that minimizes unit operations, thereby reducing the overall environmental footprint and enhancing the economic attractiveness of the final fungicide products. This advancement represents a strategic opportunity for chemical manufacturers to optimize their production portfolios and offer more competitive pricing to downstream formulators.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
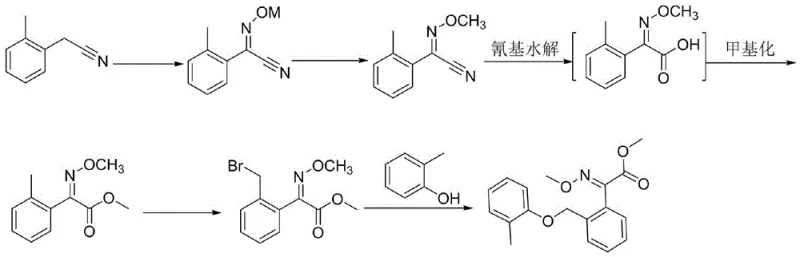
Prior art methodologies, such as those disclosed in patents CN 110396054 A and CN 108863845 B, typically rely on multi-step sequences that are inherently inefficient and prone to generating impurities. For instance, conventional routes often involve the initial formation of an oxime nitrile, followed by a separate hydrolysis step to generate the carboxylic acid, and finally a distinct esterification step to produce the methyl ester. In some variations, this requires the use of hazardous gaseous reagents like hydrogen chloride in methanol, which necessitates specialized corrosion-resistant equipment and poses significant safety risks during scale-up. Furthermore, these traditional pathways frequently suffer from poor stereocontrol, resulting in mixtures of E and Z isomers that are difficult and costly to separate, ultimately dragging down the overall process yield and increasing the cost of goods sold.
The Novel Approach
In stark contrast, the innovative process defined in CN115417793A streamlines the synthesis by integrating the final functionalization steps into a highly efficient one-pot transformation. Instead of isolating the acid intermediate and subjecting it to harsh esterification conditions, the new method utilizes a direct double alkylation strategy on the oxime acid precursor. By employing dimethyl sulfate in the presence of a suitable base within a polar aprotic solvent like DMF, both the oxime hydroxyl group and the carboxylic acid group are methylated simultaneously. This approach not only eliminates the need for intermediate isolation and drying but also operates under remarkably mild conditions, typically between 10°C and 30°C. The result is a drastic reduction in processing time and solvent consumption, coupled with the ability to consistently achieve high stereopurity without the need for complex chromatographic separations.
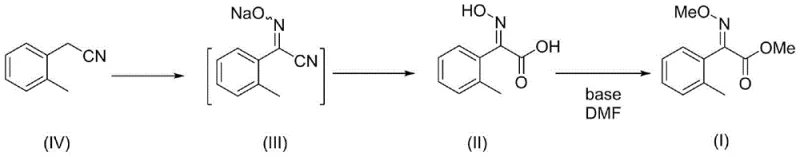
Mechanistic Insights into One-Pot Double Alkylation
The core chemical innovation lies in the simultaneous O-alkylation of two distinct nucleophilic sites within the same molecule under basic conditions. In the final step (S3), the precursor compound (II), which contains both an oxime hydroxyl group and a carboxylic acid group, is treated with a strong base such as sodium hydroxide or potassium carbonate in DMF. This deprotonation generates a dianionic or mixed anionic species where both the oximate oxygen and the carboxylate oxygen possess high nucleophilicity. Upon the addition of dimethyl sulfate, a potent methylating agent, an SN2 reaction occurs at both sites concurrently. The choice of DMF as the solvent is critical, as it effectively solvates the cationic counter-ions, thereby enhancing the nucleophilicity of the anionic centers and facilitating rapid reaction kinetics even at ambient temperatures. This mechanistic efficiency ensures that the reaction proceeds to completion with minimal side reactions, preserving the integrity of the sensitive oxime ether linkage.
Furthermore, the stereospecificity of the final product is meticulously controlled in the preceding hydrolysis step (S2), which acts as a kinetic and thermodynamic filter for the desired E-isomer. During the alkaline hydrolysis of the nitrile intermediate (III), the reaction mixture is subsequently subjected to an in situ acid adjustment using mineral acids like hydrochloric or sulfuric acid at low temperatures. This acidic environment promotes the isomerization of any formed Z-oxime acid into the thermodynamically more stable E-configuration. By locking in the E-geometry before the methylation step, the process ensures that the final ester product retains a purity of greater than 98.5% E-isomer. This level of control is vital for agrochemical efficacy, as the Z-isomer often exhibits significantly lower biological activity, and its presence would otherwise necessitate expensive recrystallization or purification steps that erode profit margins.
How to Synthesize (E)-2-Methyl-α-methoxyiminophenylacetic Acid Methyl Ester Efficiently
The synthesis protocol outlined in the patent provides a robust framework for laboratory and pilot-scale production, emphasizing operational simplicity and high reproducibility. The process begins with the nitrosation of o-tolylacetonitrile using an alkyl nitrite in an alcoholic solvent, followed by a telescoped hydrolysis and isomerization sequence to generate the high-purity E-acid intermediate. The final transformation involves the strategic addition of base and methylating agent to effect the dual functionalization. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized guide below which encapsulates the critical process controls necessary for successful implementation.
- React o-tolylacetonitrile with an alkyl nitrite and base in alcohol to form the oxime nitrile intermediate.
- Hydrolyze the nitrile under alkaline conditions followed by in situ acid adjustment to secure the high-purity E-isomer acid.
- Perform a one-pot double alkylation using dimethyl sulfate and base in DMF to simultaneously methylate the oxime and carboxyl groups.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthesis route offers compelling advantages that directly impact the bottom line and operational resilience. The reduction in the number of discrete chemical steps translates to fewer unit operations, which significantly lowers the consumption of utilities such as steam for heating and cooling water for temperature control. Moreover, the elimination of intermediate isolation steps means that solvents do not need to be recovered and recycled between every stage, reducing the load on solvent recovery plants and minimizing waste generation. This streamlined workflow allows for faster batch turnover times, enabling manufacturing facilities to respond more agilely to market fluctuations and urgent customer demands without the need for excessive inventory buffering.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the consolidation of reaction steps and the avoidance of hazardous reagents. By replacing the traditional gas-phase esterification involving hydrogen chloride with a liquid-phase methylation using dimethyl sulfate, the process eliminates the need for specialized gas scrubbing systems and corrosion-resistant reactors, leading to substantial capital expenditure savings. Additionally, the high yield and stereoselectivity reduce the loss of valuable raw materials, ensuring that a greater proportion of the input mass is converted into saleable product, thereby optimizing the cost of goods sold and improving overall margin potential for the manufacturer.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as dimethyl sulfate, common bases like sodium hydroxide, and standard solvents like DMF ensures a stable and secure supply of raw materials. Unlike specialized reagents that may be subject to geopolitical supply constraints or long lead times, these inputs are readily available from multiple global suppliers, mitigating the risk of production stoppages due to material shortages. The robustness of the reaction conditions, which tolerate a range of temperatures and concentrations without significant degradation in quality, further enhances supply chain continuity by reducing the likelihood of batch failures and off-spec production runs.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of gaseous byproducts make this process inherently safer and easier to scale from kilogram to multi-ton production volumes. The simplified workup procedure, which often involves straightforward filtration and crystallization, reduces the complexity of waste streams, facilitating easier compliance with increasingly stringent environmental regulations. By minimizing the generation of saline waste and organic effluents associated with multiple extraction and washing steps, manufacturers can lower their environmental compliance costs and enhance their sustainability profiles, which is becoming a critical factor in supplier selection for major agrochemical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and process descriptions provided in the patent literature, aiming to clarify the practical implications for industrial adoption. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing infrastructure.
Q: How does the new method improve stereoselectivity compared to prior art?
A: The process incorporates a specific in situ acid adjustment step during the hydrolysis phase (Step S2), which effectively converts the Z-isomer to the thermodynamically stable E-isomer, achieving purity greater than 98.5% before the final methylation.
Q: What are the safety advantages of this route over traditional methods?
A: Unlike older methods that require handling hazardous hydrogen chloride gas for esterification, this novel route utilizes dimethyl sulfate in a controlled liquid-phase reaction at mild temperatures (10-30°C), significantly reducing operational risks and equipment corrosion.
Q: Can this process be scaled for industrial production?
A: Yes, the method is designed for industrial scalability by telescoping multiple reactions, eliminating intermediate isolation steps, and using common solvents like DMF and alcohols, which simplifies waste treatment and reactor turnover.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (E)-2-Methyl-α-methoxyiminophenylacetic Acid Methyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of next-generation agrochemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of (E)-2-methyl-α-methoxyiminophenylacetic acid methyl ester performs consistently in your downstream synthesis of Kresoxim-methyl or Trifloxystrobin.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can drive value and reliability for your organization.
