Advanced Thiophene-Alkyne Liquid Crystal Compounds for Next-Gen Display Manufacturing
The rapid evolution of the flat panel display industry demands liquid crystal materials that can simultaneously deliver high optical anisotropy and low rotational viscosity, a challenging balance that traditional compounds often fail to achieve. Patent CN114350378B introduces a groundbreaking class of thiophene-containing alkyne liquid crystal compounds designed specifically to address these critical performance bottlenecks in modern electro-optical devices. By integrating a rigid thiophene ring with a conjugated alkyne bridge, these novel structures provide a unique electronic environment that significantly shortens response times while maintaining excellent chemical and optical stability. The general formula (I) disclosed in the patent encompasses a versatile library of derivatives where substituents R1 and R2 can be finely tuned to optimize phase transition temperatures and dielectric constants for specific display applications ranging from TN to STN panels. 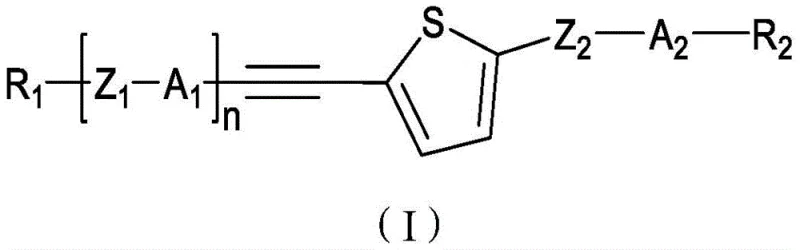 This technological advancement represents a significant leap forward for manufacturers seeking reliable electronic chemical suppliers capable of delivering next-generation materials that meet the rigorous specifications of high-resolution displays.
This technological advancement represents a significant leap forward for manufacturers seeking reliable electronic chemical suppliers capable of delivering next-generation materials that meet the rigorous specifications of high-resolution displays.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of nematic liquid crystals has been constrained by the inherent trade-off between optical anisotropy (Δn) and rotational viscosity (γ1), where increasing the conjugation length to boost Δn typically results in prohibitively high viscosity that slows down pixel switching speeds. Conventional biphenyl or cyclohexyl-based liquid crystals often require complex mixtures to achieve acceptable performance, which complicates the formulation process and can lead to compatibility issues such as crystallization at low temperatures or insufficient clearing points. Furthermore, traditional synthetic routes for high-Δn materials frequently involve multi-step sequences with harsh reaction conditions, leading to lower overall yields and the generation of difficult-to-remove impurities that degrade the voltage holding ratio of the final display device. These limitations necessitate a fundamental restructuring of the molecular core to decouple the relationship between birefringence and viscosity, a challenge that older chemical architectures simply cannot overcome without compromising other essential physical properties required for commercial viability.
The Novel Approach
The innovative strategy outlined in the patent leverages the unique electronic properties of the thiophene heterocycle combined with a linear alkyne linker to create a highly conjugated yet sterically efficient molecular framework. This specific structural motif allows for extensive electron delocalization along the molecular long axis, which dramatically enhances the refractive index difference (Δn) without introducing the bulky steric hindrance that typically increases rotational drag. The synthesis relies on a robust palladium-catalyzed cross-coupling reaction that efficiently joins the alkyne precursor with the brominated thiophene derivative, offering a direct and atom-economical pathway to the target compounds. 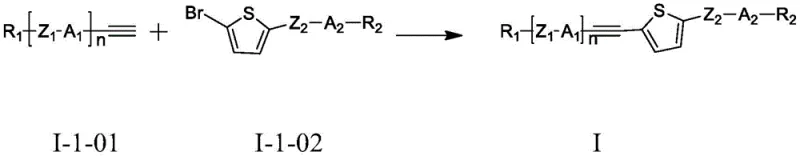 By utilizing this streamlined approach, manufacturers can access a diverse range of derivatives, such as the specific embodiment BYLC-01 shown below, which demonstrates exceptional purity and performance metrics suitable for immediate integration into advanced liquid crystal mixtures.
By utilizing this streamlined approach, manufacturers can access a diverse range of derivatives, such as the specific embodiment BYLC-01 shown below, which demonstrates exceptional purity and performance metrics suitable for immediate integration into advanced liquid crystal mixtures. 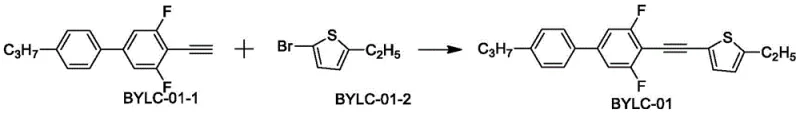 This novel methodology not only simplifies the production workflow but also ensures that the resulting materials possess the precise molecular alignment characteristics necessary for high-speed electro-optical switching.
This novel methodology not only simplifies the production workflow but also ensures that the resulting materials possess the precise molecular alignment characteristics necessary for high-speed electro-optical switching.
Mechanistic Insights into Palladium-Catalyzed Sonogashira Coupling
The core chemical transformation driving the production of these high-performance liquid crystals is the Sonogashira coupling reaction, a powerful tool in organic synthesis that facilitates the formation of carbon-carbon bonds between terminal alkynes and aryl or vinyl halides. In the context of this patent, the mechanism involves the oxidative addition of a zero-valent palladium catalyst, specifically tetrakis(triphenylphosphine)palladium(0), to the carbon-bromine bond of the thiophene precursor, generating a reactive organopalladium intermediate. This species subsequently undergoes transmetallation with a copper-acetylide complex formed in situ from the terminal alkyne and the amine base, ultimately leading to reductive elimination that forge the critical carbon-carbon triple bond linking the aromatic core to the thiophene ring. The presence of triethylamine serves a dual purpose by acting as both a solvent modifier and a base to neutralize the hydrogen bromide byproduct, thereby driving the equilibrium towards the desired product and preventing catalyst deactivation. Understanding this catalytic cycle is crucial for R&D teams aiming to optimize reaction parameters, as slight variations in ligand electronics or base strength can significantly influence the rate of coupling and the profile of side products generated during the process.
Controlling the impurity profile in the synthesis of electronic-grade liquid crystals is paramount, as even trace amounts of residual palladium or halogenated byproducts can severely impact the electrical reliability of the final display panel. The patent describes a rigorous purification protocol involving aqueous workup to remove inorganic salts, followed by solvent extraction and multiple recrystallization steps using ethanol and n-heptane to achieve gas chromatographic purities exceeding 99.8%. This high level of purity is essential for minimizing ion contamination, which is a primary cause of image sticking and flicker in active matrix displays. Furthermore, the selection of specific substituents on the phenyl and thiophene rings, such as fluorine or alkyl chains, is strategically designed to enhance the chemical stability of the molecule against UV radiation and thermal stress, ensuring long-term operational durability. By meticulously managing the reaction stoichiometry and purification conditions, producers can consistently deliver materials that meet the stringent quality standards required by top-tier display manufacturers, thereby reducing the risk of batch-to-batch variability in the final liquid crystal mixture.
How to Synthesize Thiophene Alkyne Liquid Crystal Efficiently
The synthesis of these specialized compounds follows a well-defined protocol that balances reaction efficiency with product quality, making it suitable for both laboratory optimization and large-scale manufacturing environments. The process begins with the careful preparation of anhydrous reaction conditions under a nitrogen atmosphere to prevent oxidation of the sensitive palladium catalyst and the alkyne starting materials. Operators must precisely control the reflux temperature and reaction time to ensure complete conversion of the starting materials while minimizing the formation of homocoupling byproducts that can complicate downstream purification. For a comprehensive guide on the exact molar ratios, temperature profiles, and safety precautions required for this synthesis, please refer to the standardized operating procedures detailed below.
- Prepare the reaction vessel with terminal alkyne precursor and bromo-thiophene derivative in toluene solvent under inert nitrogen atmosphere.
- Add triethylamine as the base and introduce the tetrakis(triphenylphosphine)palladium(0) catalyst to initiate the coupling reaction.
- Heat the mixture to reflux for approximately 6 hours, followed by aqueous workup, solvent extraction, and double recrystallization to achieve >99.8% purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this thiophene-alkyne technology offers substantial strategic benefits by simplifying the supply chain for high-performance display materials. The reliance on commercially available starting materials, such as brominated thiophenes and substituted phenylacetylenes, ensures a stable and resilient supply base that is less susceptible to the volatility often associated with exotic or proprietary intermediates. This accessibility translates directly into cost reduction in electronic chemical manufacturing, as the raw material costs are predictable and the synthesis route avoids the need for expensive cryogenic conditions or specialized high-pressure equipment. Furthermore, the high yields reported in the patent examples indicate a highly efficient process that maximizes material throughput, effectively lowering the cost per kilogram of the final active ingredient and providing a competitive edge in price-sensitive markets. By partnering with a supplier who has mastered this specific chemistry, procurement managers can secure a reliable source of advanced liquid crystal intermediates that offer superior performance without the premium pricing typically associated with cutting-edge display technologies.
- Cost Reduction in Manufacturing: The streamlined single-step coupling reaction significantly reduces processing time and energy consumption compared to multi-step traditional syntheses, leading to lower utility costs and higher reactor utilization rates. The elimination of complex protection and deprotection steps further simplifies the workflow, reducing labor costs and the consumption of auxiliary chemicals, which collectively contribute to a more economical production model. Additionally, the ability to recycle the solvent system and recover valuable palladium catalyst residues adds another layer of financial efficiency, ensuring that the overall cost structure remains optimized for high-volume commercial production.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route means that production schedules are less likely to be disrupted by technical failures or yield fluctuations, guaranteeing consistent delivery timelines for downstream formulators. The use of standard chemical reagents that are widely stocked by global chemical distributors mitigates the risk of supply shortages, allowing for better inventory management and long-term planning. This reliability is critical for display manufacturers who operate on tight production cycles and cannot afford delays in the arrival of key raw materials, making this technology a dependable choice for securing the continuity of the supply chain.
- Scalability and Environmental Compliance: The reaction conditions described, involving reflux in toluene with triethylamine, are inherently scalable from gram to ton quantities without requiring significant process re-engineering, facilitating a smooth transition from pilot plant to full commercial scale. Moreover, the process generates minimal hazardous waste, as the primary byproducts are inorganic salts that can be easily treated, aligning with increasingly strict environmental regulations and sustainability goals. This environmental compatibility not only reduces disposal costs but also enhances the corporate social responsibility profile of the supply chain, appealing to end-users who prioritize eco-friendly manufacturing practices in their sourcing decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of these thiophene-containing alkyne liquid crystal compounds. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for making informed decisions about material selection and process integration.
Q: What are the key performance advantages of these thiophene-alkyne liquid crystals?
A: These compounds exhibit significantly higher optical anisotropy (Δn) and lower rotational viscosity (γ1) compared to traditional biphenyl structures, directly translating to faster response times in display panels without sacrificing voltage holding ratios.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the patented method utilizes standard Sonogashira coupling conditions with readily available reagents like toluene and triethylamine, making it highly amenable to scale-up from kilogram to multi-ton commercial batches with consistent quality control.
Q: How does the thiophene ring influence the dielectric properties?
A: The incorporation of the sulfur-containing thiophene ring enhances the polarizability of the molecular core, which contributes to a favorable dielectric anisotropy (Δε), allowing for lower driving voltages in TN and STN liquid crystal display applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiophene Alkyne Liquid Crystal Supplier
As a leader in the fine chemical industry, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure necessary to bring these advanced liquid crystal intermediates from the laboratory to the global market. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demanding volume requirements of major display panel manufacturers while maintaining stringent purity specifications. We utilize state-of-the-art rigorous QC labs equipped with advanced analytical instrumentation to verify every batch against the high standards set forth in patent CN114350378B, guaranteeing that our customers receive materials with consistent optical and electrical properties. Our commitment to quality assurance extends beyond simple testing, encompassing a holistic approach to process control that minimizes variability and maximizes yield.
We invite potential partners to engage with our technical procurement team to discuss how our thiophene-alkyne solutions can enhance your product portfolio and drive innovation in your display applications. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into how switching to our optimized synthesis route can improve your bottom line without compromising on performance. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific project needs, allowing you to move forward with confidence in selecting a supplier who truly understands the complexities of high-performance electronic materials.
