Advanced Catalytic Synthesis of Chiral 1,4-Diphenyl-2-Hydroxy-1,4-Dibutanone for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing chiral carbon skeletons, which are fundamental building blocks for bioactive molecules. Patent CN112979523A introduces a significant technological advancement in this domain by providing a highly efficient preparation method for chiral 1,4-diphenyl-2-hydroxy-1,4-dibutanone compounds. This innovation centers on the utilization of a novel chiral metal complex to catalyze the asymmetric Mukaiyama aldol reaction between enol silyl ethers and phenylglyoxal monohydrate or its substituted derivatives. The breakthrough lies in the ability to achieve high enantioselectivity under remarkably mild conditions, effectively overcoming the limitations of previous protocols that often demanded harsh reagents and complex operational setups. By leveraging this catalytic system, manufacturers can access critical intermediates for drug development with superior purity profiles and streamlined processing.
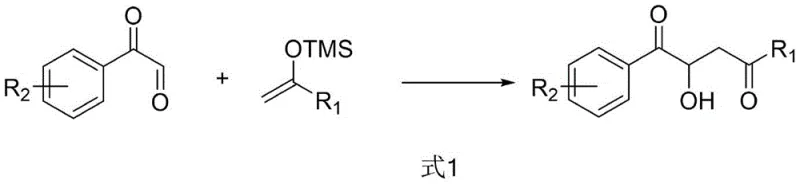
The core of this technology involves the reaction of enol silyl ethers, where the R1 group can be a phenyl or furan structure, with phenylglyoxal monohydrate derivatives containing various substituents such as halogens, methyl, methoxy, nitro, or trifluoromethyl groups. The versatility of the substrate scope allows for the synthesis of a diverse library of chiral beta-hydroxy dicarbonyl compounds, which are valuable precursors in the synthesis of natural products and pharmaceutical agents. The reaction proceeds smoothly in common organic solvents like acetone, acetonitrile, or chloroform, demonstrating the practical adaptability of this method for large-scale manufacturing environments where solvent recovery and safety are paramount concerns.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,4-diphenyl-2-hydroxy-1,4-dibutanone compounds via Mukaiyama aldol reactions has been plagued by significant operational challenges that hinder industrial adoption. Traditional protocols often necessitate the use of strong acids, such as hydrochloric acid, during the conversion of reaction intermediates into the final product, which creates severe corrosion issues for reactor equipment and generates substantial acidic waste streams that require costly neutralization and disposal procedures. Furthermore, many existing catalytic systems require strict inert gas protection throughout the entire reaction process to prevent catalyst deactivation or side reactions, adding layers of complexity and expense to the manufacturing workflow. The need for harsh reaction conditions not only compromises the safety of the operation but also limits the functional group tolerance, thereby restricting the diversity of substrates that can be successfully employed in the synthesis.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN112979523A offers a transformative solution by enabling the direct conversion of reactants into the final chiral product in a single step without the need for intermediate isolation or harsh acidic treatment. The use of the specialized chiral metal complex allows the reaction to proceed efficiently at temperatures ranging from -20°C to 30°C, significantly reducing energy consumption compared to high-temperature processes. This mild condition profile eliminates the requirement for rigorous inert atmosphere maintenance, simplifying the engineering controls needed for production. The result is a greener, more environmentally friendly process that aligns with modern sustainability goals while delivering products with exceptional enantiomeric excess values, often exceeding 90% ee, which minimizes the need for downstream chiral resolution steps.
Mechanistic Insights into Chiral Copper-Catalyzed Asymmetric Mukaiyama Aldol Reaction
The exceptional performance of this synthetic route is attributed to the unique structural features of the chiral metal complex, which acts as a highly selective Lewis acid catalyst. The catalyst is formed by the coordination of a proline-derived chiral ligand with a metal center, typically copper, iron, or zinc. The ligand framework, characterized by a rigid backbone incorporating phenolic and pyrrolidine moieties, creates a well-defined chiral pocket around the metal ion. This steric environment precisely directs the approach of the nucleophilic enol silyl ether to the electrophilic carbonyl group of the phenylglyoxal derivative, ensuring that the bond formation occurs with high facial selectivity. The electron-withdrawing trifluoromethyl group on the salicylaldehyde portion of the ligand further enhances the Lewis acidity of the metal center, accelerating the reaction rate without compromising stereocontrol.
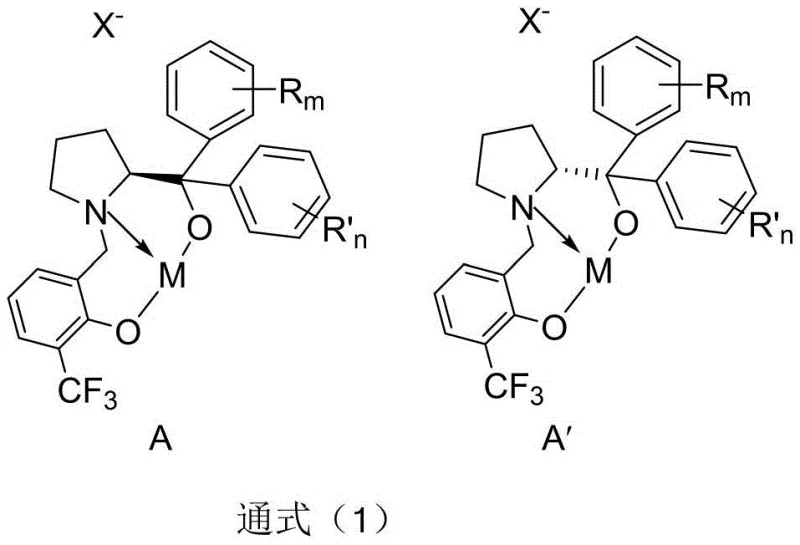
From an impurity control perspective, the mechanism inherently suppresses common side reactions associated with dicarbonyl compounds. In traditional aldol reactions, the presence of multiple reactive carbonyl sites can lead to polymerization or self-condensation byproducts. However, the specific coordination geometry of the chiral metal complex activates the phenylglyoxal substrate in a manner that favors the cross-aldol pathway exclusively. The rapid turnover of the catalyst ensures that the intermediate species are quickly converted to the stable beta-hydroxy ketone product, preventing degradation or rearrangement. This high level of chemoselectivity results in a cleaner crude reaction mixture, which significantly simplifies the purification process and improves the overall yield of the desired optical isomer, a critical factor for meeting the stringent quality standards of the pharmaceutical industry.
How to Synthesize Chiral 1,4-Diphenyl-2-Hydroxy-1,4-Dibutanone Efficiently
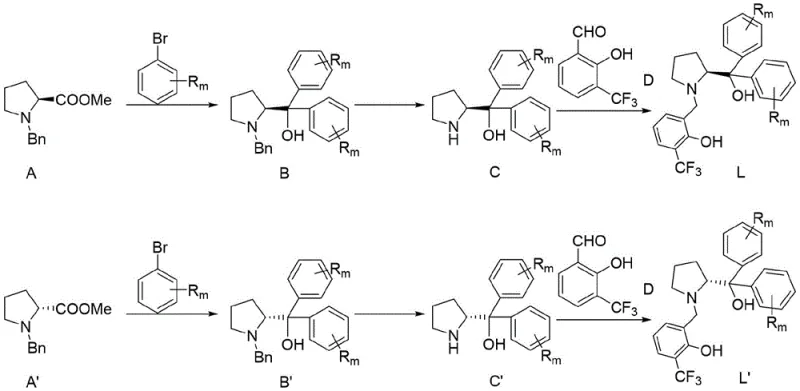
The preparation of the target compounds begins with the synthesis of the chiral ligand, which serves as the foundation for the catalytic activity. The ligand synthesis is a straightforward three-step sequence starting from commercially available proline esters, making the supply chain for the catalyst robust and cost-effective. Once the ligand is prepared, it is complexed with a metal salt in situ to generate the active catalytic species. The subsequent aldol reaction is performed by mixing the catalyst solution with the enol silyl ether and the phenylglyoxal substrate in a suitable solvent. Detailed standardized synthesis steps for the ligand and the final catalytic reaction are provided in the guide below, outlining the precise molar ratios and temperature controls required to replicate the high enantioselectivity reported in the patent literature.
- Perform a Grignard reaction between protected proline methyl ester and brominated aromatic hydrocarbon to obtain amino alcohol compound B.
- Reduce the amino alcohol compound B using hydrogen and a reducing agent to remove protection groups, yielding amino alcohol C.
- React the resulting amino alcohol C with 2-hydroxy-3-trifluoromethylbenzaldehyde in t-butanol to finalize the chiral ligand L.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers tangible strategic benefits that extend beyond mere technical feasibility. The shift towards this catalytic system addresses several critical pain points in the sourcing of complex chiral intermediates, primarily by stabilizing the supply chain through the use of readily available raw materials and simplifying the manufacturing infrastructure. The elimination of hazardous reagents and the reduction in process steps directly translate to lower operational risks and improved throughput capabilities, ensuring a more reliable flow of materials to downstream drug formulation units.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by the simplification of the reaction workflow and the elimination of expensive purification requirements. By avoiding the use of strong acids and strict inert gas conditions, the process significantly reduces the capital expenditure associated with specialized corrosion-resistant reactors and complex gas handling systems. Furthermore, the high enantioselectivity achieved minimizes the loss of material during chiral separation, effectively increasing the yield of the usable product per batch. The use of common solvents like acetone and acetonitrile also facilitates easier solvent recovery and recycling, contributing to substantial long-term savings in raw material costs and waste disposal fees.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system enhances supply security by reducing the dependency on exotic or hard-to-source reagents. The starting materials, such as phenylglyoxal monohydrate and substituted bromobenzenes, are commodity chemicals with stable global supply networks, mitigating the risk of production delays due to raw material shortages. Additionally, the mild reaction conditions allow for greater flexibility in manufacturing scheduling, as the process is less sensitive to minor fluctuations in environmental parameters. This reliability ensures consistent delivery timelines for clients, which is essential for maintaining uninterrupted production schedules in the fast-paced pharmaceutical sector.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the inherent safety and simplicity of the reaction design. The absence of highly exothermic steps or dangerous reagents makes the scale-up predictable and safe, allowing for seamless transition from kilogram to ton-scale manufacturing. From an environmental compliance standpoint, the green chemistry attributes of the method, such as reduced waste generation and lower energy consumption, align perfectly with increasingly stringent global environmental regulations. This proactive approach to sustainability not only future-proofs the manufacturing site against regulatory changes but also enhances the corporate social responsibility profile of the supply chain partners involved.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral synthesis technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation, providing clarity on the operational parameters and potential applications of the method. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing process development pipelines.
Q: What are the optimal reaction conditions for this asymmetric Mukaiyama aldol reaction?
A: The patent specifies a reaction temperature range of -20°C to 30°C, preferably -10°C to 20°C, using solvents like acetone or acetonitrile with a reaction time of 1 to 2.5 hours.
Q: Which metals are suitable for forming the chiral metal complex catalyst?
A: The chiral ligand can coordinate with copper, iron, or zinc salts, with copper trifluoromethanesulfonate showing particularly high catalytic efficiency and enantioselectivity.
Q: How does this method improve upon traditional synthesis routes?
A: Unlike conventional methods requiring harsh acidic conditions and strict inert gas protection, this novel approach operates under mild conditions without strong acids, simplifying workup and enhancing environmental safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 1,4-Diphenyl-2-Hydroxy-1,4-Dibutanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality chiral intermediates in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demanding volume requirements of global pharmaceutical clients. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of chiral 1,4-diphenyl-2-hydroxy-1,4-dibutanone delivered meets the highest international standards for enantiomeric purity and chemical integrity.
We invite you to collaborate with us to leverage this advanced catalytic technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your production volumes, demonstrating how this efficient synthesis route can optimize your budget. Please contact us today to request specific COA data and comprehensive route feasibility assessments, and let us partner with you to accelerate your drug development timeline with reliable, high-performance chemical solutions.
