Advanced Microwave-Promoted Synthesis of 2-Phenyl-3-Aldehyde Indoles for Pharmaceutical Applications
Introduction to Next-Generation Indole Synthesis
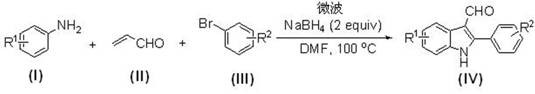
The landscape of pharmaceutical intermediate manufacturing is undergoing a significant transformation, driven by the urgent need for greener, more efficient synthetic routes. A pivotal development in this arena is detailed in patent CN110028437B, which discloses a novel microwave-promoted method for preparing 2-phenyl-3-aldehyde indole compounds. These indole derivatives are critical scaffolds in medicinal chemistry, serving as key precursors for a wide array of bioactive molecules including antibiotics, cardiotonics, and agents for treating cardiovascular and cerebrovascular diseases. Traditional synthetic pathways often rely on multi-step sequences involving expensive transition metal catalysts, which introduce complexities regarding metal residue removal and environmental compliance. In contrast, this innovative approach utilizes a simple, one-pot reaction system comprising aniline derivatives, acrolein, and bromobenzene compounds in the presence of sodium borohydride under microwave irradiation. This technology represents a paradigm shift towards sustainable manufacturing, offering a robust solution for producing high-purity pharmaceutical intermediates with enhanced operational efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indole core, particularly the 2-phenyl-3-aldehyde substitution pattern, has been fraught with synthetic challenges that hinder scalable production. Conventional methodologies predominantly depend on transition metal-catalyzed coupling reactions, such as palladium or copper-mediated processes, which necessitate rigorous exclusion of air and moisture. These methods typically involve multiple discrete steps, leading to cumulative yield losses and increased consumption of solvents and reagents. Furthermore, the reliance on precious metal catalysts introduces a significant burden on downstream processing; removing trace metal impurities to meet regulatory standards for Active Pharmaceutical Ingredients (APIs) requires specialized scavenging resins or complex recrystallization protocols. The harsh reaction conditions often associated with these traditional routes, including high temperatures and strong acidic or basic environments, can also compromise the integrity of sensitive functional groups, limiting the substrate scope and generating complex impurity profiles that are difficult to characterize and control.
The Novel Approach
The methodology outlined in patent CN110028437B offers a transformative alternative by leveraging microwave energy to drive a direct, three-component coupling reaction. This novel approach eliminates the need for transition metals entirely, substituting them with sodium borohydride, a ubiquitous and inexpensive reducing agent that acts effectively under microwave conditions. The reaction proceeds in a single pot using N,N-dimethylformamide (DMF) as the solvent, operating at a moderate temperature of 100 °C. This simplification drastically reduces the operational complexity, as it avoids the stringent inert atmosphere requirements typical of organometallic chemistry. The microwave promotion not only accelerates the reaction kinetics but also ensures uniform heating, which is crucial for maintaining consistent product quality across batches. By streamlining the synthesis into a single step with readily available starting materials, this method significantly enhances the overall atom economy and reduces the environmental footprint, making it an ideal candidate for green chemistry initiatives in the fine chemical sector.
Mechanistic Insights into Microwave-Promoted Cyclization
The mechanistic pathway of this microwave-assisted transformation involves a synergistic interaction between the organic substrates and the microwave field, facilitated by the polar nature of the DMF solvent and the ionic character of the sodium borohydride. Under microwave irradiation at 2450 MHz, the dipolar molecules in the reaction mixture align rapidly with the oscillating electric field, generating heat internally and efficiently. This internal heating mechanism promotes the nucleophilic attack of the aniline nitrogen on the electron-deficient beta-carbon of the acrolein, initiating the cascade. Subsequently, the bromobenzene component participates in the cyclization process, likely through a base-mediated activation that allows for the formation of the new carbon-carbon bonds required to close the indole ring. The presence of sodium borohydride is critical, not only for its potential role in reducing intermediate species but also for maintaining the necessary basicity to drive the condensation forward without degrading the sensitive aldehyde moiety. This delicate balance ensures that the formyl group at the 3-position remains intact, a feature that is often challenging to preserve in harsher oxidative or reductive environments.
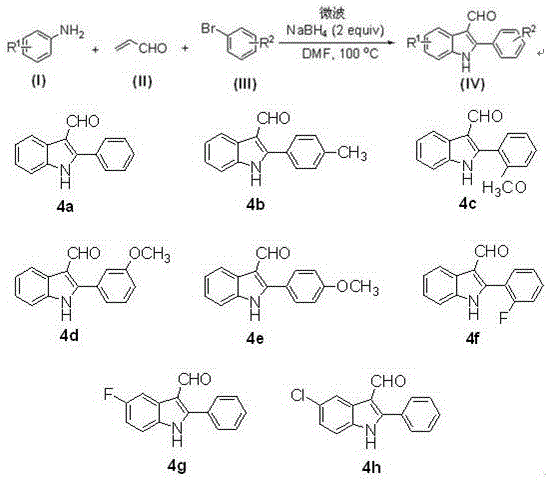
From an impurity control perspective, the simplicity of this catalytic system offers distinct advantages. The absence of transition metals removes a major class of potential contaminants, thereby simplifying the impurity profile primarily to organic byproducts which are easier to separate via standard chromatographic techniques. The reaction conditions are mild enough to prevent the polymerization of acrolein, a common side reaction that can plague indole syntheses involving alpha,beta-unsaturated aldehydes. Furthermore, the broad substrate tolerance demonstrated in the patent data indicates that electronic variations on the aniline and bromobenzene rings—such as halogens, methyl, or methoxy groups—are well-tolerated. This suggests that the transition state of the rate-determining step is not overly sensitive to steric or electronic perturbations, allowing for the synthesis of a diverse library of analogues with consistent quality. Such robustness is essential for process chemists aiming to develop a versatile platform technology for generating structural diversity in drug discovery campaigns.
How to Synthesize 2-Phenyl-3-Aldehyde Indole Efficiently
The practical execution of this synthesis is designed for straightforward implementation in both laboratory and pilot-scale settings. The protocol involves charging a reaction vessel with equimolar amounts of the aniline and bromobenzene derivatives, along with a slight excess of acrolein, in DMF solvent. Sodium borohydride is then added as the promoter, and the mixture is subjected to microwave irradiation. The specific parameters, including a frequency of 2450 MHz and power of 1200 W, ensure rapid energy transfer. Following the reaction period, the workup procedure is remarkably simple, involving aqueous quenching and extraction, followed by purification. For a detailed breakdown of the standardized operating procedures and safety precautions required for this specific microwave protocol, please refer to the technical guide below.
- Combine aniline derivative (5 mmol), bromobenzene derivative (5 mmol), and acrolein (6 mmol) in a reaction vessel with DMF solvent.
- Add sodium borohydride (10 mmol) as the base catalyst and subject the mixture to microwave irradiation at 100 °C for 15 hours.
- Perform workup by extraction with ethyl acetate, dry over sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this microwave-promoted synthesis route offers compelling economic and logistical benefits that extend beyond mere technical feasibility. The shift away from precious metal catalysts to commodity chemicals like sodium borohydride fundamentally alters the cost structure of the manufacturing process. By eliminating the need for expensive palladium or copper salts and their associated ligands, the raw material costs are significantly reduced. Moreover, the simplification of the downstream processing—specifically the removal of heavy metal scavenging steps—translates into substantial savings in both time and auxiliary materials. This streamlined workflow enhances the overall throughput of the production facility, allowing for faster turnaround times on custom synthesis orders and improved responsiveness to market demands for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic impact of replacing transition metal catalysts with sodium borohydride cannot be overstated. Precious metals are subject to volatile market pricing and require complex recovery processes to minimize loss, whereas sodium borohydride is a stable, low-cost bulk chemical. Additionally, the one-pot nature of the reaction reduces solvent consumption and labor hours associated with isolating intermediates between multiple steps. This consolidation of the synthetic sequence leads to a drastic simplification of the bill of materials and a reduction in waste disposal costs, contributing to a leaner and more cost-effective manufacturing model that maximizes margin potential for high-volume production.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available starting materials. Anilines, bromobenzenes, and acrolein are commodity chemicals produced by numerous global suppliers, mitigating the risk of single-source dependency that often plagues specialized catalyst supply chains. The robustness of the reaction conditions, which do not require cryogenic temperatures or ultra-high vacuum systems, means that the process can be executed in a wider range of manufacturing facilities, including those in emerging markets. This flexibility allows for a more diversified sourcing strategy, ensuring continuity of supply even in the face of regional disruptions or logistical bottlenecks, thereby securing the production pipeline for downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns perfectly with modern green chemistry principles. The elimination of toxic heavy metals reduces the hazardous waste load, simplifying effluent treatment and lowering the regulatory burden associated with metal discharge limits. The use of DMF, while requiring careful handling, is a well-understood solvent with established recovery protocols. The energy efficiency of microwave heating, particularly when scaled using continuous flow microwave reactors, offers a pathway to further reduce the carbon footprint of the synthesis. These factors collectively enhance the sustainability profile of the product, making it highly attractive for pharmaceutical companies striving to meet their corporate social responsibility goals and adhere to increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this microwave-assisted technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the process viability. Understanding these nuances is crucial for technical teams assessing the fit of this methodology within their existing manufacturing infrastructure.
Q: What are the advantages of using microwave promotion for indole synthesis?
A: Microwave promotion significantly accelerates reaction kinetics, allowing for milder temperatures (100 °C) compared to traditional thermal heating. It eliminates the need for expensive transition metal catalysts, reducing heavy metal contamination risks in the final API.
Q: Is sodium borohydride safe for large-scale production of these intermediates?
A: Yes, sodium borohydride is a widely available, cost-effective, and relatively safe reducing agent compared to complex organometallic catalysts. Its use simplifies waste treatment and lowers the overall hazard profile of the manufacturing process.
Q: What is the typical purity profile of the resulting indole compounds?
A: The process yields high-purity products (75-95% isolated yield) after standard silica gel chromatography. The absence of transition metals simplifies the purification process, ensuring the impurity profile meets stringent pharmaceutical standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Phenyl-3-Aldehyde Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting innovative synthetic technologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert process chemists has thoroughly evaluated the microwave-promoted route described in CN110028437B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art microwave reactors and rigorous QC labs capable of ensuring stringent purity specifications for every batch of 2-phenyl-3-aldehyde indole derivatives we produce. Our commitment to quality assurance means that we can deliver these critical intermediates with the consistency and reliability required for GMP-grade API synthesis, supporting your drug development timelines from preclinical stages through to commercial launch.
We invite you to collaborate with us to leverage this advanced chemistry for your specific project needs. Whether you require custom kilogram quantities for clinical trials or metric ton volumes for commercial supply, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your molecule. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in microwave-assisted synthesis can optimize your supply chain and reduce your overall cost of goods sold.
