Advanced Chiral Phosphine-Phosphoramidite Ligands for Scalable Asymmetric Hydrogenation
The landscape of asymmetric synthesis is continuously evolving, driven by the demand for higher purity and efficiency in the production of chiral active pharmaceutical ingredients (APIs). Patent CN112824421A introduces a significant advancement in this field through the development of a novel chiral phosphine-phosphoramidite ligand based on a chiral beta-aminophosphine skeleton. This technology addresses critical bottlenecks in catalytic asymmetric hydrogenation, offering a versatile platform for synthesizing optical homochiral drugs, pesticides, and fine chemicals. The core innovation lies in the streamlined preparation method which utilizes asymmetric hydrogenation of (Z)-(alpha-aryl-beta-phosphoryl) enamides followed by hydrolysis and reduction. This approach not only simplifies the synthetic route but also ensures high stereocontrol, making it a compelling solution for industrial applications where consistency and yield are paramount. For R&D directors and procurement specialists, understanding the mechanistic underpinnings of this patent is essential for evaluating its potential integration into existing manufacturing workflows.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing chiral phosphine ligands often involve complex multi-step sequences that suffer from low overall yields and poor atom economy. Many conventional C2 symmetric ligands require rigorous exclusion of air and moisture throughout the synthesis and application phases, which drastically increases operational costs and infrastructure requirements. Furthermore, the reliance on expensive precious metal precursors without efficient recycling mechanisms leads to significant metal contamination in the final product, necessitating costly purification steps to meet stringent pharmaceutical standards. The limited substrate scope of older ligand generations often forces process chemists to develop custom solutions for each new molecule, resulting in prolonged development timelines and inconsistent batch-to-batch quality. These inefficiencies create substantial barriers to scaling up production for commercial quantities of chiral intermediates.
The Novel Approach
The methodology disclosed in CN112824421A overcomes these historical challenges by introducing a robust and modular synthetic strategy. By utilizing a chiral beta-aminophosphine intermediate derived from asymmetric hydrogenation, the process ensures high fidelity in stereochemistry transfer from the very beginning of the synthesis. The subsequent condensation with chlorophosphites is conducted under mild conditions, typically between 18-25°C, which minimizes energy consumption and reduces the risk of thermal degradation of sensitive functional groups. This novel approach significantly enhances the tolerance of the resulting catalysts to air and humidity, thereby simplifying handling procedures and reducing the need for specialized glovebox equipment. The versatility of the ligand structure allows for easy modification of the aryl and aliphatic groups, enabling fine-tuning of electronic and steric properties to match specific substrate requirements without redesigning the entire catalytic system.
Mechanistic Insights into Rh-Catalyzed Asymmetric Hydrogenation
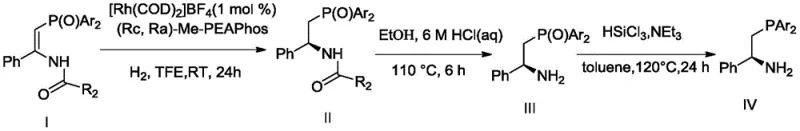
The heart of this technology is the rhodium-catalyzed asymmetric hydrogenation step, which establishes the chiral center with exceptional precision. The reaction employs a metal precursor such as [Rh(COD)2]BF4 combined with the chiral ligand to form an active catalytic species capable of activating molecular hydrogen. The mechanism involves the coordination of the substrate, specifically the (Z)-(alpha-aryl-beta-phosphoryl) enamide, to the rhodium center, followed by migratory insertion and reductive elimination. This cycle proceeds with high turnover frequencies, evidenced by TON values reaching up to 10000, indicating that a single metal center can process thousands of substrate molecules before deactivation. The high enantioselectivity, reported up to 99% ee, is attributed to the rigid chiral environment created by the phosphine-phosphoramidite backbone, which effectively differentiates between the prochiral faces of the substrate during the hydrogen addition step.

Following the hydrogenation, the intermediate undergoes hydrolysis and reduction to yield the chiral phosphine-amine compound, which retains the spatial configuration established in the previous step. This retention of configuration is critical for ensuring that the final ligand imparts the desired stereochemistry in downstream applications. The use of silanes like HSiCl3 in the presence of triethylamine facilitates the reduction of the phosphoryl group to the phosphine moiety without racemization. This careful control over the oxidation state of phosphorus is a key differentiator, as many alternative reduction methods can lead to loss of optical purity. The resulting ligands are then purified via column chromatography and recrystallization, ensuring that the final product meets the high-purity specifications required for GMP manufacturing environments.
How to Synthesize Chiral Phosphine-Phosphoramidite Ligand Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these high-value ligands at scale. The process begins with the preparation of the chiral rhodium catalyst solution, which is stirred under nitrogen protection to ensure complete complexation before substrate addition. The hydrogenation is typically carried out in an autoclave at pressures around 60 bar, although the system demonstrates flexibility across a wide pressure range. After the reaction, the solvent is removed, and the intermediate is subjected to acidic hydrolysis followed by basic workup to isolate the amine.
- Prepare chiral beta-aminophosphine intermediate via asymmetric hydrogenation of (Z)-(alpha-aryl-beta-phosphoryl) enamide using Rh catalyst.
- Perform hydrolysis and reduction on the chiral amide to obtain the chiral phosphine-amine compound with retained configuration.
- Condense the chiral phosphine-amine with chlorophosphite in toluene with triethylamine to yield the final phosphine-phosphoramidite ligand.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this ligand technology offers transformative benefits for supply chain stability and cost management. The high catalytic activity and turnover number mean that significantly less precious metal is required per kilogram of product, directly reducing the raw material cost burden associated with rhodium or iridium catalysts. Additionally, the improved stability of the ligands reduces waste generated from decomposition during storage and transport, leading to more predictable inventory management and reduced losses. The ability to operate under milder conditions also translates to lower energy costs and reduced wear on reactor equipment, contributing to a more sustainable and economically viable manufacturing process. These factors collectively enhance the competitiveness of the final API or intermediate in the global market.
- Cost Reduction in Manufacturing: The elimination of complex protection-deprotection sequences and the high efficiency of the catalyst significantly lower the overall processing costs. By achieving high conversion rates and selectivity, the need for extensive downstream purification is minimized, which reduces solvent usage and waste disposal expenses. The robustness of the ligand allows for potential recycling of the metal catalyst, further driving down the cost of goods sold. This economic efficiency is crucial for maintaining margins in the highly competitive fine chemical sector.
- Enhanced Supply Chain Reliability: The synthetic route relies on readily available starting materials and standard reagents, mitigating the risk of supply disruptions associated with exotic or proprietary precursors. The tolerance of the catalyst to air and moisture simplifies logistics, as strict inert atmosphere shipping is not always required, thereby expanding the pool of qualified logistics providers. This resilience ensures consistent delivery schedules and reduces the likelihood of production stoppages due to material shortages. Procurement managers can rely on a more stable supply base for critical chiral building blocks.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations that are easily transferred from laboratory to pilot and commercial scales. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, reducing the compliance burden on manufacturing sites. The high atom economy of the hydrogenation step ensures that most of the reactant mass ends up in the product, minimizing the environmental footprint. This alignment with green chemistry principles enhances the corporate sustainability profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ligand technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy and reliability. Understanding these details helps stakeholders make informed decisions about process adoption and vendor selection.
Q: What is the enantioselectivity performance of this ligand system?
A: According to patent CN112824421A, the catalyst system achieves up to 99% enantioselectivity (ee) across various substrates including C=C, C=N, and C=O bonds.
Q: How stable is the catalyst under reaction conditions?
A: The catalyst demonstrates high stability with a Turnover Number (TON) reaching up to 10000, and it maintains activity under both normal and high hydrogen pressure conditions.
Q: What are the storage requirements for these ligands?
A: The ligands exhibit good tolerance to air and humidity, simplifying storage and handling requirements compared to highly air-sensitive traditional phosphine ligands.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Phosphine-Phosphoramidite Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced catalytic technologies play in the development of next-generation pharmaceuticals and fine chemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to plant is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of ligand or intermediate meets the highest industry standards. Our commitment to quality and consistency makes us a trusted partner for multinational corporations seeking reliable sources for complex chiral molecules.
We invite you to collaborate with us to leverage this patented technology for your specific applications. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your current manufacturing processes. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our solutions can optimize your supply chain and reduce overall production costs. Let us help you achieve your commercial goals with precision and reliability.
