Advanced Chiral N-Heterocyclic Carbene Catalysts for High-Purity Pharmaceutical Intermediates
The landscape of asymmetric catalysis is undergoing a significant transformation with the disclosure of patent CN116082272A, which introduces a novel class of chiral N-heterocyclic carbene (NHC) catalysts designed to overcome longstanding limitations in organic synthesis. This technological breakthrough addresses critical inefficiencies in the preparation of complex pharmaceutical intermediates, specifically targeting the low yields and poor stereocontrol associated with traditional amide distal C(sp3)-H activation reactions. By integrating advanced transition metal catalysis with tailored steric environments, this new methodology achieves yields approaching quantitative levels while maintaining rigorous control over diastereomeric ratios. For research and development teams focused on process optimization, this represents a pivotal shift from empirical trial-and-error approaches to rational catalyst design that prioritizes both efficiency and selectivity. The implications for industrial manufacturing are profound, as the ability to access high-purity chiral building blocks with reduced waste generation directly correlates to improved process economics and environmental sustainability. As we delve into the technical specifics, it becomes evident that this innovation offers a robust platform for the scalable production of high-value fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing similar chiral scaffolds often relied on heterogeneous catalysts such as montmorillonite under solvent-free conditions at elevated temperatures, typically around 140 °C. While these methods demonstrated feasibility, they suffered from significant drawbacks including limited substrate scope, restricted primarily to basic alkynes, and modest yields that rarely exceeded 85%. Furthermore, the diastereoselectivity (dr value) achieved with these conventional systems was often insufficient for demanding pharmaceutical applications, typically hovering around 10:1, necessitating costly and time-consuming purification steps to isolate the desired enantiomer. The reliance on harsh thermal conditions also raised concerns regarding energy consumption and the potential for thermal degradation of sensitive functional groups, limiting the versatility of the synthetic route. These inherent limitations created a bottleneck in the supply chain for complex intermediates, forcing manufacturers to accept lower throughput and higher operational costs to meet purity specifications.
The Novel Approach
In stark contrast, the novel approach detailed in the patent employs a sophisticated multi-step sequence utilizing homogeneous transition metal catalysis to achieve superior outcomes. The process initiates with the precise formation of alpha-bromocycloheptanone, followed by a nickel-catalyzed hydroboration using NiCl2(dppp) and DIBAL-H, which allows for the functionalization of a much broader range of alkyne substrates, including substituted variants. Subsequent coupling reactions mediated by PEPSI-IPr and KOH in THF at moderate temperatures (70 °C) ensure high conversion rates without the thermal stress associated with older methods. The culmination of this sequence involves an asymmetric hydrogenation step utilizing a rhodium-DuanPhos complex under 80 atm of hydrogen pressure, which establishes the critical chiral centers with exceptional fidelity. This comprehensive redesign of the synthetic pathway not only elevates yields to nearly 99% but also enhances the dr value to between 10:1 and 14:1, driven by the increased steric hindrance of the substituted carbene ligands.
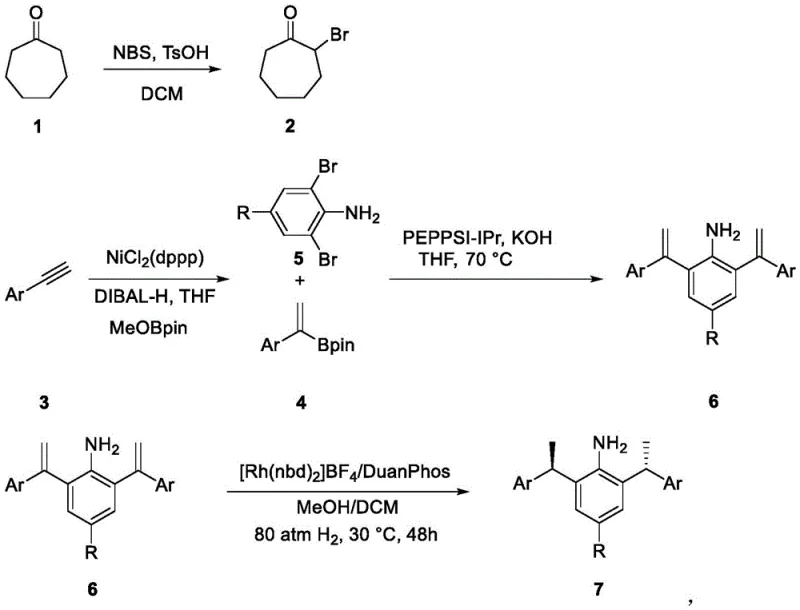
Mechanistic Insights into Ni/Rh Dual-Catalyzed Asymmetric Synthesis
The mechanistic superiority of this new catalyst system lies in the intricate interplay between the electronic properties of the N-heterocyclic carbene ligand and the steric bulk introduced by the substituent groups. The substitution pattern on the carbene backbone creates a chiral pocket that effectively discriminates between competing transition states during the radical decarboxylation alkylation and C-H activation steps. This steric differentiation is crucial for suppressing the formation of unwanted diastereomers, thereby streamlining the downstream purification process and reducing the overall material loss. The use of DuanPhos in the hydrogenation step further reinforces this stereocontrol through its rigid bite angle and chiral backbone, ensuring that the reduction of the olefinic precursors proceeds with high enantioselectivity. For R&D directors, understanding this mechanism provides a blueprint for further optimizing reaction conditions, such as fine-tuning the solvent polarity or adjusting the hydrogen pressure to maximize turnover numbers. The robustness of the catalytic cycle is evidenced by its tolerance to various functional groups, allowing for the late-stage functionalization of complex molecules without the need for extensive protecting group strategies.
Furthermore, the control over impurity profiles is significantly enhanced by the specificity of the nickel and rhodium catalysts, which minimize side reactions such as polymerization or over-reduction that are common in less selective systems. The patent data indicates that the reaction conditions are mild enough to preserve sensitive moieties while being vigorous enough to drive the reaction to completion, striking an optimal balance for process chemistry. This level of control is essential for meeting the stringent regulatory requirements of the pharmaceutical industry, where even trace impurities can disqualify a batch. By leveraging the unique electronic characteristics of the NHC ligand, the catalyst facilitates single-electron transfer processes that are otherwise difficult to achieve with traditional organocatalysts. This opens up new retrosynthetic disconnections for medicinal chemists, enabling the construction of molecular architectures that were previously inaccessible or economically unviable.
How to Synthesize Chiral N-Heterocyclic Carbene Catalyst Efficiently
The synthesis of these high-performance catalysts follows a logical progression of bond-forming events that can be adapted for kilogram-scale production with appropriate engineering controls. The initial bromination step requires careful temperature management to prevent poly-bromination, while the subsequent nickel-catalyzed step demands strict exclusion of moisture and oxygen to maintain catalyst activity. Detailed standard operating procedures for each transformation, including workup and purification protocols, are essential for ensuring batch-to-batch consistency and maximizing overall yield. The final cyclization and salt formation steps involve precise stoichiometric control of reagents like carbon disulfide and perchlorate salts to ensure the formation of the stable cationic catalyst species.
- Preparation of alpha-bromocycloheptanone via NBS bromination of cycloheptanone using TsOH catalyst in DCM.
- Nickel-catalyzed hydroboration of phenylacetylene derivatives using NiCl2(dppp) and DIBAL-H to generate vinyl boronate intermediates.
- Suzuki-Miyaura coupling with dibromoaniline substrates followed by asymmetric hydrogenation using Rh-DuanPhos complex to establish chirality.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel catalytic technology translates into tangible strategic benefits that extend beyond simple yield improvements. The ability to utilize a wider range of starting materials, including substituted alkynes, reduces dependency on单一 sources of raw materials and mitigates supply risk associated with commodity chemicals. Moreover, the significant increase in reaction yield implies a drastic reduction in the volume of solvent and reagents required per unit of product, leading to substantial cost savings in raw material procurement and waste disposal. The enhanced stereoselectivity minimizes the need for expensive chiral chromatography or recrystallization steps, shortening the overall production cycle time and freeing up manufacturing capacity for other projects. These efficiencies collectively contribute to a more resilient and cost-effective supply chain capable of responding rapidly to market demands.
- Cost Reduction in Manufacturing: The elimination of harsh thermal conditions and the move towards higher-yielding catalytic cycles significantly lowers energy consumption and raw material usage. By achieving near-quantitative yields, the process minimizes the generation of by-products that require costly separation and disposal, directly impacting the bottom line. The use of commercially available ligands and standard transition metals ensures that the catalyst cost remains manageable, avoiding the prohibitive expenses often associated with exotic noble metal complexes. This economic efficiency makes the technology viable for the production of both high-value active pharmaceutical ingredients and lower-margin fine chemical intermediates.
- Enhanced Supply Chain Reliability: The broad substrate scope of this catalytic system allows for greater flexibility in sourcing raw materials, reducing the risk of bottlenecks caused by shortages of specific reagents. The robustness of the reaction conditions ensures consistent output quality even when minor variations in feedstock occur, providing stability to the production schedule. Additionally, the scalability of the process from laboratory to pilot plant has been demonstrated, assuring partners that supply volumes can be ramped up quickly to meet commercial deadlines without compromising product integrity. This reliability is critical for maintaining just-in-time inventory levels and meeting the rigorous delivery schedules of global pharmaceutical clients.
- Scalability and Environmental Compliance: The process operates under relatively mild conditions compared to traditional methods, reducing the safety hazards associated with high-pressure and high-temperature operations. The improved atom economy resulting from higher selectivity means less chemical waste is generated, simplifying compliance with increasingly stringent environmental regulations. The ability to recycle solvents and recover catalyst residues further enhances the sustainability profile of the manufacturing process. These factors make the technology attractive for companies aiming to reduce their carbon footprint and align with green chemistry principles while maintaining high production standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel chiral N-heterocyclic carbene catalyst technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of its practical utility. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this chemistry into their existing manufacturing portfolios.
Q: What are the key advantages of this novel chiral NHC catalyst over prior art?
A: The novel catalyst features substituted groups that provide greater steric hindrance, resulting in significantly improved yields (up to 99%) and superior diastereoselectivity (dr values up to 14:1) compared to basic body catalysts.
Q: Can this catalyst be used for substrates other than basic alkynes?
A: Yes, unlike previous methods limited to basic alkynes using montmorillonite, this innovative process utilizes a Nickel/PEPSI-IPr system that is applicable to both basic and substituted alkynes, broadening its synthetic utility.
Q: What specific reactions does this catalyst mediate effectively?
A: This catalyst is highly effective in mediating radical decarboxylation alkylation reactions and amide distal C(sp3)-H activated tandem cyclization reactions, enabling the synthesis of complex chiral scaffolds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral N-Heterocyclic Carbene Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this novel chiral N-heterocyclic carbene catalyst technology in advancing the synthesis of complex pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the highest industry standards for enantiomeric excess and chemical purity. We are committed to leveraging our technical expertise to optimize this catalytic process for your specific application, delivering cost-effective solutions that accelerate your drug development timelines.
We invite you to engage with our technical procurement team to discuss how this innovative catalyst can enhance your current synthetic routes and reduce overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into the potential economic benefits of adopting this technology for your specific product portfolio. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a partnership built on transparency, technical excellence, and mutual success.
