Revolutionizing Acyl Arylamine Production: A Direct One-Step Catalytic Strategy for Global Supply Chains
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the need for more sustainable and efficient synthetic routes, particularly for high-volume intermediates like acyl arylamines. Patent CN1332936C introduces a groundbreaking methodology that challenges the conventional multi-step dogma by enabling the direct conversion of nitroaromatics into acyl arylamines in a single operational step. This technological leap bypasses the traditional requirement to first reduce nitro groups to anilines before acylation, thereby collapsing two distinct unit operations into one streamlined process. For R&D directors and process chemists, this represents a paradigm shift in how we approach the synthesis of critical scaffolds found in herbicides like propanil and pharmaceutical agents such as ciprofloxacin precursors. The core innovation lies in the synergistic use of red phosphorus and iodine catalysts, which facilitate both the reduction of the nitro group and the subsequent acylation in situ, offering a robust alternative to legacy methods that rely on hazardous acylating agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the industrial production of acyl arylamines has been burdened by a fragmented synthetic sequence that inherently accumulates cost and environmental liability. The standard protocol necessitates the initial reduction of nitroaromatics to aromatic amines, a step often plagued by the use of stoichiometric metal reducers like iron or zinc, which generate massive quantities of solid waste sludge, or catalytic hydrogenation which demands high-pressure equipment and expensive noble metal catalysts. Following isolation of the amine, a second reaction with acid chlorides or anhydrides is required, typically involving aggressive reagents such as thionyl chloride or phosgene derivatives that release corrosive hydrogen chloride and sulfur dioxide gases. This two-step approach not only doubles the processing time and solvent consumption but also introduces significant safety hazards related to the handling of moisture-sensitive acyl chlorides and the management of acidic off-gases, creating a complex supply chain bottleneck for procurement teams seeking reliable sources.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a direct reductive acylation strategy that merges the reduction and coupling events into a single pot, fundamentally altering the economic and safety profile of the manufacture. By employing nitroaromatics and carboxylic acids directly in the presence of a red phosphorus and iodine system, the process eliminates the need for pre-formed acid chlorides and external acid-binding agents entirely. This approach allows for the use of inexpensive, stable carboxylic acids as acyl donors, removing the volatility and toxicity associated with thionyl chloride while simultaneously avoiding the isolation of potentially unstable intermediate amines. The reaction operates effectively across a broad temperature range of 20 to 200°C, with optimal results often achieved between 80 and 150°C, providing process engineers with significant flexibility in reactor design and thermal management without the need for specialized high-pressure infrastructure.
Mechanistic Insights into Red Phosphorus/Iodine Catalyzed Reductive Acylation
The mechanistic elegance of this transformation relies on the in situ generation of reactive phosphorus species that serve dual functions as both reducing agents and activators for the carboxylic acid. It is hypothesized that the interaction between red phosphorus and iodine generates phosphorus triiodide or similar reactive intermediates which activate the carboxylic acid towards nucleophilic attack while concurrently reducing the nitro functionality through a series of electron transfer steps. This concerted mechanism ensures that the newly formed amine species is immediately trapped by the activated acyl species, preventing side reactions such as over-reduction to hydrazines or azo compounds that often plague traditional nitro reductions. The stoichiometry is carefully balanced, with the patent specifying a molar ratio of nitroaromatic to carboxylic acid of 1:1 to 1:10, preferably 1:4 to 1:5, ensuring that the acid acts as both reactant and solvent medium to drive the equilibrium towards the desired amide product.
Furthermore, the impurity profile of this reaction is remarkably clean compared to classical methods, primarily because it avoids the formation of sulfonated by-products or chlorinated impurities often associated with thionyl chloride usage. The selectivity is further enhanced by the catalytic nature of the iodine component, which is used in minute quantities ranging from 0.01 to 0.1 equivalents relative to the substrate. Experimental data indicates that varying the iodine source, whether elemental iodine, potassium iodide, or sodium iodide, maintains high conversion rates above 99% with selectivity exceeding 95% for model substrates like 3,4-dichloronitrobenzene. This high level of control over the reaction pathway minimizes the burden on downstream purification units, allowing for simpler workup procedures such as aqueous washing and recrystallization rather than energy-intensive distillation or chromatography.
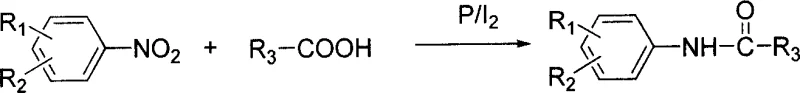
How to Synthesize 3,4-Dichloro-N-propionylanilide Efficiently
The practical implementation of this chemistry is demonstrated through the efficient synthesis of 3,4-dichloro-N-propionylanilide, a key intermediate in agrochemical manufacturing, which serves as a benchmark for the technology's scalability. The protocol involves charging a reaction vessel with 3,4-dichloronitrobenzene and propionic acid in a molar ratio of 1:4, followed by the addition of red phosphorus and a catalytic amount of iodine. The mixture is heated to 110°C and stirred for approximately 6 hours, after which the reaction mass is cooled and poured into ice water for quenching. The crude product is then extracted using toluene, washed with bicarbonate solution to remove residual acid, dried, and concentrated to yield the target compound with a purity of 96.5% and an isolated yield of 95.4%.
- Combine nitroaromatic substrate with excess carboxylic acid (molar ratio 1: 4 to 1:5) in a reaction vessel.
- Add red phosphorus (1.5 to 2 equivalents) and a catalytic amount of iodine or iodide (0.01 to 0.1 equivalents) to the mixture.
- Heat the reaction mixture to 80-150°C for 1 to 20 hours, followed by standard workup including extraction and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this direct synthesis route offers profound strategic advantages that extend far beyond simple yield improvements, fundamentally reshaping the cost structure of acyl arylamine production. By eliminating the requirement for thionyl chloride and other chlorinating agents, manufacturers can drastically reduce their exposure to volatile raw material markets and the stringent regulatory compliance costs associated with handling hazardous gases. The removal of the intermediate amine isolation step translates directly into reduced cycle times and lower utility consumption, as there is no longer a need for separate drying, filtration, and storage of the amine intermediate, thereby freeing up reactor capacity and increasing overall plant throughput significantly.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the substitution of expensive and hazardous acyl chlorides with commodity carboxylic acids, which are widely available and significantly cheaper on a per-mole basis. Additionally, the process operates without the need for stoichiometric acid-binding agents like triethylamine or pyridine, which are costly to purchase and dispose of, leading to substantial savings in raw material expenditure. The simplified workup procedure reduces solvent usage and waste treatment costs, as the absence of inorganic salt by-products from neutralization steps minimizes the volume of aqueous waste requiring treatment, resulting in a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: From a supply chain perspective, relying on stable carboxylic acids instead of moisture-sensitive acid chlorides mitigates the risk of raw material degradation during storage and transport, ensuring consistent quality and availability. The robustness of the reaction conditions, which tolerate a wide range of temperatures and do not require anhydrous environments, reduces the likelihood of batch failures due to minor deviations in operating parameters. This resilience enhances the predictability of delivery schedules, allowing supply chain planners to maintain lower safety stock levels while still meeting customer demand, ultimately improving cash flow and inventory turnover rates for the entire value chain.
- Scalability and Environmental Compliance: The environmental footprint of this process is markedly smaller, aligning with increasingly strict global regulations on emissions and waste disposal. By avoiding the generation of sulfur dioxide and hydrogen chloride gases, facilities can operate with simpler scrubbing systems and avoid the capital expenditure associated with specialized corrosion-resistant equipment. The high atom economy of the direct reductive acylation means that a greater proportion of the input mass ends up in the final product, reducing the E-factor of the process and supporting corporate sustainability goals, which is a critical factor for multinational corporations evaluating potential suppliers for long-term partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology, derived directly from the experimental data and scope of the patent documentation. These insights are intended to clarify the operational boundaries and potential applications of the red phosphorus/iodine catalytic system for stakeholders evaluating its integration into existing production lines.
Q: What are the primary advantages of this direct synthesis method over traditional routes?
A: This method eliminates the need for separate nitro reduction and subsequent acylation steps, avoiding hazardous reagents like thionyl chloride and reducing overall process time and waste generation significantly.
Q: Can this technology be applied to the synthesis of cyclic imides?
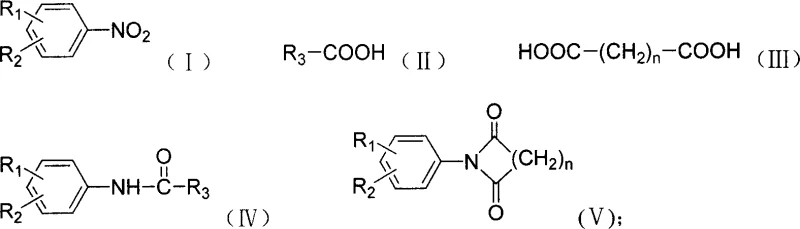
A: Yes, the patent explicitly demonstrates that reacting nitroaromatics with dicarboxylic acids under these conditions successfully yields aryl cyclic imides, expanding its utility beyond simple acyl arylamines.
Q: Is the use of solvents mandatory for this reaction protocol?
A: No, the reaction can be conducted under solvent-free conditions, although inert solvents like toluene or xylene can be used if necessary for heat transfer or mixing efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acyl Arylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic methodologies requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN1332936C are fully realized in large-scale operations. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of acyl arylamine meets the exacting standards required by the global pharmaceutical and agrochemical industries.
We invite you to engage with our technical procurement team to discuss how this innovative technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits for your specific supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and competitiveness in your manufacturing operations.
