Advanced Synthesis of Perfluorobisphenol A: Enabling High-Performance Fluorine Material Manufacturing
Advanced Synthesis of Perfluorobisphenol A: Enabling High-Performance Fluorine Material Manufacturing
The rapid evolution of the advanced materials sector demands precursors that offer superior thermal stability and chemical resistance, driving the industry toward perfluorinated alternatives to traditional bisphenols. Patent CN113004144B, published in late 2022, introduces a groundbreaking synthesis method for Perfluorobisphenol A that addresses critical limitations in current manufacturing protocols. This technology leverages a strategic two-step pathway involving active ester intermediates to achieve yields approaching 72% with exceptional purity profiles. For R&D directors and procurement specialists in the fluoropolymer industry, this represents a pivotal shift away from hazardous high-pressure processes toward a more controllable, mild reaction environment. The ability to produce high-purity Perfluorobisphenol A reliably is essential for next-generation polycarbonates, epoxy resins, and optical materials used in aerospace and microelectronics. By utilizing 2,3,5,6-tetrafluoro-4-(1,1,1,3,3,3-hexafluoro-2-hydroxypropan-2-yl)phenol as a starting scaffold, the inventors have unlocked a route that minimizes side reactions and simplifies downstream processing, ensuring a consistent supply of this critical monomer for high-value applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of perfluorinated bisphenols has been plagued by severe operational challenges that hinder commercial scalability and increase total cost of ownership. The traditional approach typically involves the direct condensation of hexafluoroacetone with phenol derivatives catalyzed by aggressive superacids such as hydrogen fluoride or trifluoromethanesulfonic acid. These reactions necessitate extreme conditions, including high temperatures and elevated pressures, which impose rigorous demands on reactor metallurgy and safety systems. The corrosive nature of hydrogen fluoride creates significant maintenance burdens and poses acute safety risks to personnel, while the harsh reaction environment often leads to uncontrolled polymerization or degradation of the sensitive fluorinated backbone. Furthermore, the success rate of these conventional methods is notoriously low, resulting in inconsistent batch quality and complex purification requirements to remove residual acid catalysts and oligomeric byproducts. For supply chain managers, these factors translate into volatile lead times and unpredictable production costs, making it difficult to secure a reliable source of high-grade perfluorobisphenol A for precision manufacturing needs.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a sophisticated activation strategy that circumvents the need for extreme conditions by employing an active ester intermediate. As illustrated in the reaction scheme below, the process begins with the conversion of the phenolic hydroxyl group into a highly reactive leaving group, such as a triflate or trifluoroacetate, under mild basic conditions. This activation step primes the molecule for a subsequent coupling reaction with tetrafluorophenol, facilitated by a range of accessible Lewis or Brønsted acids rather than corrosive superacids. 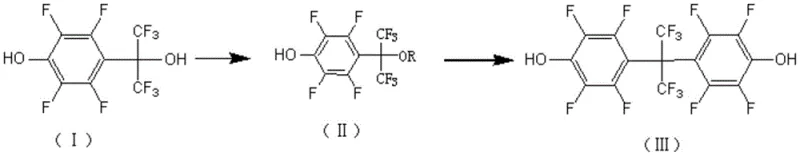 The transition from a direct condensation to this stepwise coupling mechanism allows the reaction to proceed at moderate temperatures ranging from 80°C to 180°C, drastically reducing energy consumption and equipment stress. The use of common solvents like dichloroethane or dichlorobenzene further enhances the practicality of the process, enabling standard glass-lined or stainless steel reactors to be used without specialized fluoropolymer linings. This novel approach not only improves the overall yield to approximately 72% but also ensures that the final product exhibits a purity exceeding 99%, effectively eliminating the need for costly and time-consuming recrystallization cycles that are typical of older methods.
The transition from a direct condensation to this stepwise coupling mechanism allows the reaction to proceed at moderate temperatures ranging from 80°C to 180°C, drastically reducing energy consumption and equipment stress. The use of common solvents like dichloroethane or dichlorobenzene further enhances the practicality of the process, enabling standard glass-lined or stainless steel reactors to be used without specialized fluoropolymer linings. This novel approach not only improves the overall yield to approximately 72% but also ensures that the final product exhibits a purity exceeding 99%, effectively eliminating the need for costly and time-consuming recrystallization cycles that are typical of older methods.
Mechanistic Insights into Acid-Catalyzed Coupling of Fluorinated Esters
The core innovation of this synthesis lies in the mechanistic pathway of the second step, where the activated ester intermediate undergoes an electrophilic aromatic substitution or a Friedel-Crafts type alkylation with tetrafluorophenol. In the presence of acid catalysts such as titanium tetrachloride, aluminum trichloride, or trifluoroacetic acid, the leaving group on the intermediate is protonated or coordinated, facilitating its departure and generating a stabilized carbocation species at the benzylic position. This electron-deficient center is then attacked by the electron-rich aromatic ring of the tetrafluorophenol, despite the deactivating influence of the fluorine substituents, due to the high reactivity of the carbocation intermediate. The strong electron-withdrawing nature of the multiple fluorine atoms on both aromatic rings requires careful tuning of the acid strength to balance reactivity with selectivity, preventing over-reaction or decomposition of the sensitive hexafluoroisopropylidene bridge. By controlling the stoichiometry and temperature, the reaction selectively forms the desired carbon-carbon bond between the two fluorinated aromatic systems, preserving the integrity of the phenolic hydroxyl groups essential for subsequent polymerization.
Impurity control is inherently built into this mechanism through the specificity of the activation step and the mildness of the coupling conditions. In traditional high-pressure syntheses, the formation of regioisomers and polymeric tars is common due to the indiscriminate nature of superacid catalysis at high thermal energy. However, by pre-forming the active ester in Step 1 using bases like triethylamine or pyridine at 40-60°C, the system ensures that only the desired hydroxyl group is activated, minimizing side reactions at other sites on the molecule. During the coupling phase, the use of inert atmospheres prevents oxidative degradation of the phenolic moieties, which is a frequent source of colored impurities in fluorinated phenol chemistry. The resulting crude product typically requires only a simple aqueous wash to remove acid residues and inorganic salts, followed by concentration, to yield a material of sufficient purity for most applications, with recrystallization serving merely as a final polishing step to achieve the reported 99.0144% purity specification.
How to Synthesize Perfluorobisphenol A Efficiently
To implement this synthesis effectively in a pilot or production setting, operators must adhere to strict control parameters regarding reagent addition rates and temperature gradients to maximize the formation of the active ester. The process begins with the dissolution of the starting tetrafluoro-phenol derivative in a polar aprotic solvent like tetrahydrofuran, followed by the controlled addition of the base and the activating agent, ensuring the exotherm is managed to prevent thermal runaway. Once the intermediate is isolated or used in situ, the coupling reaction requires precise monitoring of the acid catalyst concentration to drive the equilibrium toward the bisphenol product without degrading the fluorinated backbone. Detailed standardized operating procedures covering reagent ratios, specifically the mass ratio of intermediate to tetrafluorophenol to acid (approximately 2.5-2.8 : 1 : 1.5-1.9), are critical for reproducibility.
- Perform esterification of 2,3,5,6-tetrafluoro-4-(1,1,1,3,3,3-hexafluoro-2-hydroxypropan-2-yl)phenol with trifluoromethanesulfonic anhydride or trifluoroacetyl chloride using a base catalyst like triethylamine at 40-60°C.
- Couple the resulting active ester intermediate with tetrafluorophenol under inert atmosphere using an acid catalyst such as titanium tetrachloride or trifluoroacetic acid at elevated temperatures (80-180°C).
- Execute post-reaction workup by washing with water to neutrality, concentrating the solution, and recrystallizing from toluene to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers profound strategic advantages that extend beyond mere technical feasibility to impact the bottom line directly. The elimination of hazardous reagents like anhydrous hydrogen fluoride removes the need for specialized storage facilities and expensive corrosion-resistant infrastructure, leading to substantial capital expenditure savings during plant setup or retrofitting. Furthermore, the mild reaction conditions significantly extend the lifespan of standard processing equipment, reducing maintenance downtime and replacement costs associated with acid corrosion, thereby enhancing overall asset utilization rates. The simplicity of the post-treatment workflow, which relies on basic aqueous washing rather than complex distillation or chromatographic separation, translates into reduced utility consumption and faster batch cycle times. These operational efficiencies collectively contribute to a more robust and cost-effective supply chain, mitigating the risks of production delays caused by equipment failure or regulatory compliance issues related to hazardous material handling.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive and dangerous superacids with commercially available, lower-cost acid catalysts such as sulfuric acid or p-toluenesulfonic acid. By avoiding the high-pressure reactors required for hexafluoroacetone condensation, manufacturers can utilize standard vessel designs, significantly lowering the barrier to entry and operational overhead. The high yield of approximately 72% ensures that raw material utilization is optimized, minimizing waste generation and the associated costs of disposal and environmental compliance. Additionally, the ability to achieve high purity without extensive purification steps reduces solvent usage and energy demand for heating and cooling, further driving down the variable cost per kilogram of the final fluorine material.
- Enhanced Supply Chain Reliability: The reliance on easily sourced raw materials, such as tetrafluorophenol and common acylating agents, insulates the production process from the volatility often seen in the supply of specialized fluorinated gases like hexafluoroacetone. The robustness of the reaction conditions means that production schedules are less susceptible to disruptions caused by equipment maintenance or safety shutdowns, ensuring a steady flow of product to downstream customers. This reliability is crucial for industries like semiconductor manufacturing and aerospace, where interruptions in the supply of high-performance polymers can halt entire production lines. The scalability of the method allows suppliers to respond quickly to surges in demand without the long lead times associated with commissioning high-pressure infrastructure.
- Scalability and Environmental Compliance: From an environmental perspective, the reduction in hazardous waste streams aligns with increasingly stringent global regulations on fluorinated compound manufacturing. The aqueous workup generates waste streams that are easier to neutralize and treat compared to the fluoride-rich effluents from traditional HF-based processes, simplifying wastewater treatment requirements. The process is inherently safer, reducing the risk of catastrophic accidents and the associated liability and insurance costs. This improved environmental profile not only facilitates regulatory approval in key markets but also enhances the brand reputation of manufacturers as sustainable producers of advanced materials, appealing to eco-conscious end-users in the electronics and automotive sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology for Perfluorobisphenol A. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational parameters. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the purity levels achievable with this new Perfluorobisphenol A synthesis method?
A: The patented method consistently achieves product purity exceeding 99.0%, with specific examples demonstrating 99.0144% purity via gas chromatography analysis, making it suitable for high-end electronic and optical applications.
Q: How does this route improve safety compared to traditional hexafluoroacetone methods?
A: Unlike conventional methods requiring hazardous hydrogen fluoride or trifluoromethanesulfonic acid under high temperature and pressure, this novel approach utilizes milder acid catalysts and moderate temperatures, significantly reducing equipment corrosion risks and operational hazards.
Q: Is this synthesis method scalable for industrial production of fluorine materials?
A: Yes, the process is designed for batch production with easily obtained raw materials and simple post-treatment procedures, allowing for straightforward scale-up from laboratory to commercial manufacturing without complex purification bottlenecks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Perfluorobisphenol A Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity fluorinated monomers play in the development of next-generation advanced materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are equipped with rigorous QC labs and state-of-the-art analytical instrumentation to guarantee stringent purity specifications for every batch of Perfluorobisphenol A we deliver. Our commitment to quality assurance means that we can consistently meet the demanding requirements of clients in the semiconductor, aerospace, and specialty polymer industries, providing a stable foundation for their own R&D and manufacturing activities.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this superior manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will enhance your supply chain resilience and product performance.
