Optimizing Clopidogrel Intermediate Production: A Technical Breakthrough in o-Chlorophenylglycine Synthesis
The pharmaceutical industry continuously seeks robust manufacturing routes for critical cardiovascular medications, particularly for the antiplatelet agent Clopidogrel. Patent CN114685300A introduces a significant advancement in the preparation of o-chlorophenylglycine, a pivotal chiral building block and key intermediate in the synthesis of Clopidogrel. This intellectual property addresses longstanding challenges associated with traditional Strecker-type syntheses, specifically targeting the elimination of colored by-products and the optimization of atom economy. By refining reaction parameters such as temperature control and solvent composition, the disclosed method achieves a streamlined pathway that enhances both yield and chemical purity. For global procurement and R&D teams, this represents a shift towards more sustainable and cost-effective manufacturing protocols that minimize waste generation while maximizing output efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of o-chlorophenylglycine has been plagued by the formation of stubborn impurities known colloquially as "red oil." Prior art, including patents such as CN109721502A and CN105237421A, typically involves the reaction of o-chlorobenzaldehyde with cyanide sources under conditions that often lead to the polymerization of intermediate alpha-aminonitriles. This side reaction not only consumes valuable raw materials, thereby depressing overall yield, but also results in deeply colored products that necessitate rigorous and costly decolorization steps. Traditional processes frequently rely on activated carbon treatment or oxidative bleaching with hydrogen peroxide to meet purity specifications, adding complexity to the downstream processing and increasing the environmental footprint through additional solid waste or chemical usage.
The Novel Approach
The methodology outlined in CN114685300A fundamentally re-engineers the process flow to preemptively avoid these impurities rather than removing them post-formation. The core innovation lies in the precise regulation of the initial condensation reaction temperature, maintained strictly between 20°C and 40°C, coupled with a homogeneous solvent system comprising water and alcohols like methanol or ethanol. This specific environment facilitates the efficient conversion of o-chlorobenzaldehyde into o-chlorobenzenehydantoin without triggering the polymerization pathways responsible for discoloration. Consequently, the intermediate obtained is a white crystalline solid that requires no decolorization, directly translating to a simplified workflow and higher throughput. 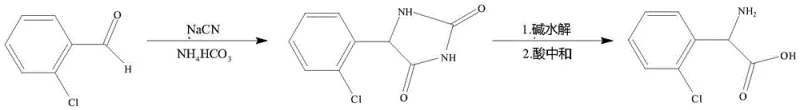
Mechanistic Insights into Side Reaction Suppression
To fully appreciate the technical merit of this invention, one must understand the mechanistic origin of the process failures in older methods. The formation of "red oil" is attributed to the instability of the alpha-aminonitrile intermediate generated during the initial nucleophilic addition of cyanide to the imine species. Under uncontrolled thermal conditions or in heterogeneous systems, these intermediates undergo oligomerization or polymerization, creating complex, colored macromolecules that are difficult to separate. The visual representation of this degradation pathway highlights the vulnerability of the alpha-aminonitrile structure when exposed to suboptimal reaction environments, serving as a cautionary model for process chemists aiming to optimize yield. 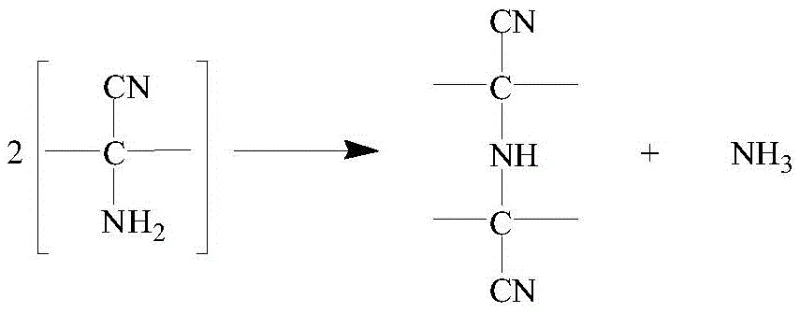
The patented process mitigates this risk through a combination of thermodynamic and kinetic controls. By utilizing a water-alcohol mixed solvent, the system ensures that all reactants—o-chlorobenzaldehyde, ammonium bicarbonate, and sodium cyanide—are fully dissolved, creating a true homogeneous phase that eliminates mass transfer limitations often exacerbated by phase transfer catalysts. Furthermore, the strict temperature cap of 40°C ensures that the activation energy required for the polymerization side reaction is not reached, while still allowing the desired cyclization to the hydantoin ring to proceed efficiently. This selective suppression of side pathways is further reinforced by the immediate acidification and recrystallization of the hydantoin intermediate, which physically separates any trace impurities before they can propagate into the final hydrolysis step.
How to Synthesize o-Chlorophenylglycine Efficiently
The synthesis protocol described in the patent offers a reproducible framework for manufacturing high-purity o-chlorophenylglycine suitable for GMP environments. The procedure begins with the careful mixing of stoichiometric amounts of o-chlorobenzaldehyde, ammonium bicarbonate, and sodium cyanide in a defined water-alcohol solvent matrix. Following the heat preservation reaction period, the mixture is acidified to precipitate the hydantoin intermediate, which is then subjected to a critical recrystallization step to ensure maximum purity prior to hydrolysis. The detailed standardized synthesis steps, including specific molar ratios and safety precautions for handling cyanide salts, are provided in the guide below.
- React o-chlorobenzaldehyde with ammonium bicarbonate and sodium cyanide in a water-alcohol mixed solvent at 20-40°C to form o-chlorobenzenehydantoin, avoiding side reactions.
- Purify the intermediate hydantoin via recrystallization in methanol or ethanol to remove impurities before hydrolysis.
- Hydrolyze the purified hydantoin using 30-50% sodium or potassium hydroxide solution at 120-150°C, followed by acidification to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain directors and procurement managers, the adoption of this optimized synthetic route offers tangible strategic benefits beyond mere technical elegance. The elimination of the decolorization unit operation significantly reduces the consumption of auxiliary materials such as activated carbon or bleaching agents, directly lowering the variable cost per kilogram of the active pharmaceutical ingredient (API) intermediate. Furthermore, the removal of these purification steps shortens the overall cycle time of the batch process, enhancing facility throughput and allowing for more flexible production scheduling to meet fluctuating market demands for Clopidogrel generics.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by removing the need for expensive phase transfer catalysts and decolorization agents. By operating in a homogeneous aqueous-alcoholic system, the method simplifies the reaction setup and reduces the complexity of waste treatment, as there are no heavy metal catalysts or large volumes of spent activated carbon to dispose of. This streamlined approach minimizes utility consumption and labor hours associated with filtration and bleaching operations, resulting in a leaner and more economically viable production model.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, specifically the tolerance for standard alcohol solvents and the avoidance of sensitive catalytic systems, ensures consistent batch-to-batch quality. This reliability is crucial for maintaining uninterrupted supply lines to downstream API manufacturers. The high yield and purity achieved without complex purification trains mean that production campaigns are less likely to fail due to off-specification batches, thereby securing the continuity of supply for critical cardiovascular medications.
- Scalability and Environmental Compliance: The method is inherently designed for large-scale industrial production, utilizing common solvents and reagents that are readily available in bulk quantities. The high atom economy and the absence of toxic heavy metal catalysts align with modern green chemistry principles, facilitating easier regulatory approval and reducing the environmental compliance burden. The simplified wastewater profile, devoid of complex organic polymers from side reactions, lowers the cost and difficulty of effluent treatment, making the process highly scalable from pilot plant to multi-ton commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and process descriptions found within the patent documentation, providing clarity on the practical aspects of adopting this technology for commercial production.
Q: How does this new method prevent the formation of 'red oil' impurities?
A: The method strictly controls the reaction temperature between 20-40°C during the initial condensation step. This specific thermal window prevents the polymerization of the alpha-aminonitrile intermediate, which is the primary cause of the colored 'red oil' by-products seen in conventional processes.
Q: Is a phase transfer catalyst required for this synthesis?
A: No, this optimized process utilizes a specific ratio of water and alcohol solvents (such as methanol or ethanol) that creates a homogeneous reaction system. This eliminates the need for expensive phase transfer catalysts, simplifying the workup and reducing raw material costs.
Q: What is the purity profile of the final o-chlorophenylglycine product?
A: By incorporating an intermediate recrystallization step for the o-chlorobenzenehydantoin, the process effectively removes impurities before the final hydrolysis. This results in a final product with HPLC purity exceeding 99% without the need for post-reaction decolorization treatments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable o-Chlorophenylglycine Supplier
As the global demand for generic Clopidogrel continues to rise, securing a stable source of high-quality intermediates is paramount for pharmaceutical manufacturers. NINGBO INNO PHARMCHEM leverages extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver o-chlorophenylglycine that meets stringent purity specifications. Our rigorous QC labs ensure that every batch adheres to the highest standards of quality, utilizing advanced analytical techniques to verify the absence of critical impurities such as the "red oil" by-products associated with inferior synthesis methods.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this greener, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific volume requirements and quality targets.
