Optimizing 1-Amino-2,3-Propanediol Production for High-Purity Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for critical intermediates that balance high purity with operational safety. A pivotal advancement in this domain is detailed in patent CN101560160A, which discloses a novel catalytic synthesis method for 1-amino-2,3-propanediol (1-APD). This compound serves as an essential building block for non-ionic contrast agents like Iohexol, as well as various agrochemical and pharmaceutical applications. The patented technology addresses long-standing challenges in amination reactions, specifically the trade-off between conversion rates and selectivity. By employing a unique staged temperature-rise protocol combined with specific amine or phenol derivative catalysts, the process achieves a remarkable yield of up to 81.5% and a purity exceeding 99.5%. For R&D directors and procurement specialists, understanding the nuances of this catalytic system is vital for securing a reliable pharmaceutical intermediate supplier capable of delivering consistent quality at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
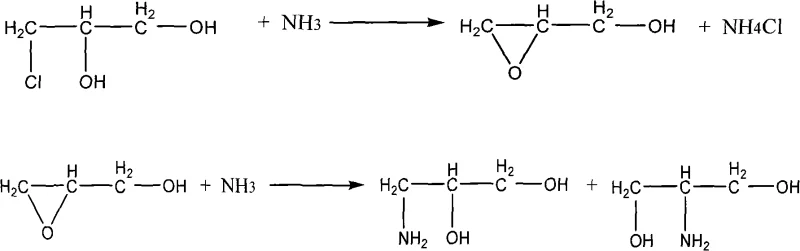
Historically, the synthesis of 1-amino-2,3-propanediol via the ammonolysis of 3-chloro-1,2-propanediol has been plagued by significant inefficiencies and safety concerns. Traditional protocols, such as those cited in prior art like USP4B60697, often necessitate harsh reaction conditions, including temperatures around 85°C and pressures as high as 4 MPa. These aggressive parameters not only impose severe stress on reactor equipment but also drive unwanted side reactions. Under such high-energy conditions, the nucleophilic attack by ammonia becomes less selective, leading to the formation of substantial quantities of isomeric by-products like 2-amino-1,3-propanediol (2-APD), as well as secondary and tertiary amines. Furthermore, intermolecular condensation polymers often form, complicating downstream purification. The resulting crude mixtures typically require extensive and energy-intensive distillation processes to achieve acceptable purity, yet even then, the final product purity often hovers around 99%, with unspecified levels of the difficult-to-separate 2-APD isomer. This lack of selectivity directly impacts the cost of goods sold and creates bottlenecks in the supply chain for high-value contrast media.
The Novel Approach
In stark contrast, the methodology outlined in CN101560160A introduces a paradigm shift through the implementation of a controlled, staged temperature-rise reaction coupled with specialized catalysis. Instead of a single high-temperature spike, the reaction initiates at a mild 40-45°C and gradually progresses to 65°C over distinct time intervals. This kinetic control allows the primary amination reaction to proceed with high specificity before the energy threshold for significant by-product formation is reached. The introduction of catalysts such as diphenylamine or hydroquinone further modulates the reaction pathway, effectively suppressing the formation of the 2-APD isomer to levels below 0.3%. This approach not only enhances the yield to over 80% but also drastically simplifies the purification workflow. By minimizing the generation of polymeric and isomeric impurities at the source, the process reduces the load on vacuum distillation units, thereby lowering energy consumption and improving the overall throughput of high-purity API intermediate manufacturing.
Mechanistic Insights into Catalytic Amination and Selectivity Control
The core innovation of this synthesis lies in the synergistic effect between the staged thermal profile and the specific choice of organic catalysts. The reaction begins with the nucleophilic substitution of the chlorine atom in 3-chloro-1,2-propanediol by ammonia. In conventional systems, the high reactivity of the epoxide intermediate (formed via intramolecular cyclization under basic conditions) often leads to ring opening at the wrong position, generating the undesired 2-amino isomer. However, the presence of amine derivatives like diphenylamine or phenolic compounds like resorcinol appears to stabilize the transition state or modify the local pH environment within the reaction matrix. This stabilization favors the attack at the primary carbon (C3) rather than the secondary carbon (C2), ensuring the formation of the 1-amino structure. The gradual increase in temperature from 40°C to 65°C ensures that the activation energy for the desired substitution is met without triggering the higher-energy pathways associated with polymerization or excessive isomerization. This precise control is critical for maintaining the integrity of the diol functionality while introducing the amine group.
Furthermore, the mechanism inherently supports superior impurity control, which is a primary concern for R&D teams focusing on regulatory compliance. The suppression of 2-amino-1,3-propanediol is particularly valuable because this isomer has physical properties very similar to the target product, making separation via standard distillation extremely difficult and costly. By keeping the 2-APD content below 0.3%, the process eliminates the need for complex chromatographic separations or multiple recrystallization steps. Additionally, the catalyst system minimizes the formation of heavy ends and tars, which typically foul distillation columns and reduce campaign lengths. The ability to recover the catalyst post-reaction—achieving recovery rates of 75-80%—further indicates that the catalyst remains chemically stable throughout the process and does not degrade into the product stream. This stability ensures that the final commercial scale-up of complex pharmaceutical intermediates remains consistent batch after batch, with minimal risk of catalyst-derived contamination.
How to Synthesize 1-Amino-2,3-Propanediol Efficiently
The practical execution of this synthesis requires careful attention to the molar ratios and pressure controls defined in the patent. The process utilizes a molar excess of ammonia (1:8 to 1:15 ratio relative to the chlorohydrin) to drive the equilibrium forward and minimize dialkylation. The reaction is conducted in a high-pressure reactor capable of maintaining low positive pressures (0.15-0.30 MPa), which is significantly safer than traditional high-pressure ammonolysis. Following the reaction, the workup involves a streamlined sequence of ammonia recovery, pH adjustment to >12.0, dehydration, and decolorization using activated carbon. The detailed standardized operating procedures for implementing this catalytic route in a pilot or production setting are outlined below.
- Mix 3-chloro-1,2-propanediol with 25-30% ammonia water at a molar ratio of 1: 8 to 1:15 in a high-pressure reactor.
- Add 0.1-2.0% molar equivalent of catalyst (diphenylamine or hydroquinone) and initiate staged heating starting at 40-45°C.
- Progressively increase temperature to 55°C and finally 65°C while maintaining pressure below 0.30 MPa to maximize selectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic synthesis method offers profound strategic advantages beyond mere technical metrics. The shift from harsh, non-selective conditions to a mild, catalyzed process translates directly into operational resilience and cost optimization. By eliminating the need for extreme pressures and temperatures, the process reduces the wear and tear on critical reactor assets, extending their lifecycle and reducing maintenance downtime. Moreover, the high selectivity of the reaction means that raw material utilization is maximized; less 3-chloro-1,2-propanediol is wasted on forming useless by-products, effectively lowering the raw material cost per kilogram of finished good. The ability to recover and reuse the catalyst further compounds these savings, creating a closed-loop system that minimizes both material costs and hazardous waste disposal fees. These factors collectively contribute to a more stable and predictable pricing model for buyers seeking a reliable pharmaceutical intermediate supplier.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the downstream purification train. Because the reaction selectively produces the target 1-isomer with minimal 2-isomer contamination, the energy-intensive fractional distillation steps required in older methods can be shortened or optimized. The removal of salts and decolorization steps prior to distillation protects the column efficiency, allowing for faster cycle times. Additionally, the recovery of the expensive organic catalysts (diphenylamine/hydroquinone) means that the effective cost of the catalytic system is amortized over multiple batches. This elimination of waste and optimization of energy usage results in substantial cost savings in pharmaceutical intermediate manufacturing without compromising on the stringent quality standards required for injectable contrast agents.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by processes that are sensitive to minor fluctuations in conditions or raw material quality. The robust nature of this staged-temperature protocol makes it highly tolerant to standard industrial variances, ensuring consistent output. The use of readily available starting materials like 3-chloro-1,2-propanediol and aqueous ammonia, combined with common organic catalysts, mitigates the risk of raw material shortages. Furthermore, the milder reaction conditions reduce the safety risks associated with high-pressure operations, lowering the likelihood of unplanned shutdowns due to safety incidents. This operational stability allows suppliers to commit to tighter delivery windows, effectively reducing lead time for high-purity pharmaceutical intermediates and providing buyers with greater confidence in their inventory planning.
- Scalability and Environmental Compliance: As regulatory scrutiny on chemical manufacturing intensifies, the environmental profile of a synthesis route becomes a key differentiator. This method generates significantly less hazardous waste compared to traditional high-pressure ammonolysis, primarily due to the high catalyst recovery rate and reduced formation of tarry by-products. The lower energy demand for heating and distillation also reduces the carbon footprint of the manufacturing process. From a scalability perspective, the reaction does not require exotic high-pressure vessels rated for 4 MPa; standard reactors rated for roughly 0.3-0.5 MPa are sufficient. This lowers the capital expenditure barrier for scaling production from pilot plants to multi-ton commercial facilities, facilitating the commercial scale-up of complex pharmaceutical intermediates while adhering to modern green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 1-amino-2,3-propanediol. These insights are derived directly from the technical specifications and experimental data provided in the underlying patent literature, ensuring that stakeholders have accurate information for decision-making.
Q: What is the primary advantage of the staged temperature method for 1-APD synthesis?
A: The staged temperature approach significantly improves reaction selectivity, reducing the formation of difficult-to-remove by-products like 2-amino-1,3-propanediol and achieving purity levels above 99.5%.
Q: Can the catalyst used in this process be recovered?
A: Yes, the amine or phenol derivative catalysts can be recovered and reused with a recovery rate of approximately 75-80%, contributing to substantial cost efficiency and waste reduction.
Q: How does this method compare to traditional high-pressure amination?
A: Unlike traditional methods requiring pressures up to 4 MPa and temperatures of 85°C, this catalytic method operates at mild pressures (0.15-0.30 MPa) and lower temperatures, enhancing safety and equipment longevity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Amino-2,3-Propanediol Supplier
The technological advancements described in patent CN101560160A represent a significant leap forward in the efficient production of critical pharmaceutical building blocks. At NINGBO INNO PHARMCHEM, we recognize the value of such innovative processes in delivering superior products to the global market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with the necessary pressure-rated reactors and advanced distillation columns to implement this staged-temperature catalytic synthesis with precision. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of 1-amino-2,3-propanediol meets the exacting standards required for Iohexol synthesis and other high-value applications.
We invite procurement leaders and R&D directors to collaborate with us to leverage this optimized synthetic route for your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project benefits from the highest quality intermediates delivered with speed and reliability.
