Scalable Green Manufacturing of 2,3-Dihydrofuran Scaffolds via Aqueous Halocyclization
Scalable Green Manufacturing of 2,3-Dihydrofuran Scaffolds via Aqueous Halocyclization
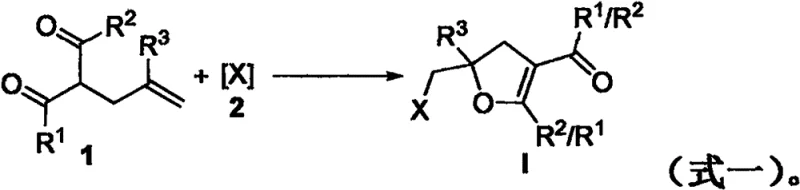
The landscape of fine chemical manufacturing is rapidly shifting towards sustainable methodologies that minimize environmental footprint while maximizing operational efficiency. A pivotal advancement in this domain is documented in patent CN112159375B, which discloses a novel, green preparation method for 2,3-dihydrofuran compounds. This technology leverages a catalyst-free halocyclization strategy performed in an aqueous medium at room temperature, representing a significant departure from traditional synthetic routes that often demand harsh conditions. For R&D directors and procurement specialists alike, this innovation offers a compelling value proposition: the ability to access complex heterocyclic scaffolds through a process that is not only chemically elegant but also economically and environmentally superior. By eliminating the need for expensive additives and utilizing water as a primary solvent, this method addresses critical pain points in modern pharmaceutical intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the electrophilic halocyclization of alkenes to form cyclic ethers like 2,3-dihydrofurans has been plagued by significant operational and environmental drawbacks. Conventional protocols frequently necessitate the use of stoichiometric amounts of strong bases or oxidants, which generate substantial quantities of chemical waste and complicate downstream processing. Furthermore, many established methods rely on toxic organic solvents such as dichloromethane or acetonitrile, posing serious health and safety risks to personnel and requiring costly waste disposal infrastructure. The requirement for elevated reaction temperatures or strict inert atmospheres further exacerbates energy consumption and equipment costs. From a supply chain perspective, the reliance on sensitive catalysts that require rigorous removal steps to meet ppm-level impurity specifications adds unnecessary lead time and expense to the manufacturing timeline, making these traditional routes less attractive for large-scale commercialization.
The Novel Approach
In stark contrast, the methodology outlined in the patent introduces a streamlined, additive-free protocol that operates under remarkably mild conditions. The core innovation lies in the utilization of a DMSO/Water mixed solvent system, where water acts as the dominant green medium. This approach allows the reaction to proceed efficiently at room temperature while openly exposed to the atmosphere, completely removing the need for inert gas blanketing or specialized pressure vessels. The versatility of the system is demonstrated by its compatibility with various halogenating reagents, including N-iodosuccinimide (NIS) and N-bromosuccinimide (NBS), delivering high yields without the burden of catalyst residues. This shift towards aqueous chemistry not only aligns with green chemistry principles but also drastically simplifies the work-up procedure, as the product can often be isolated through straightforward extraction and chromatography.
Mechanistic Insights into Electrophilic Halocyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for diverse substrates. The reaction proceeds via an electrophilic activation pathway rather than a radical mechanism. The halogenating reagent generates an electrophilic halogen species that attacks the electron-rich alkene moiety of the 1,3-dicarbonyl substrate. This initial interaction forms a transient halonium ion intermediate, which is subsequently intercepted by the neighboring carbonyl oxygen atom acting as an intramolecular nucleophile. This cyclization event constructs the dihydrofuran ring with high regioselectivity. Crucially, control experiments utilizing radical scavengers such as TEMPO and BHT showed no significant inhibition of the reaction, confirming that free radical pathways are not operative. This mechanistic clarity assures chemists that the reaction is robust and predictable, minimizing the risk of unpredictable side reactions often associated with radical processes.
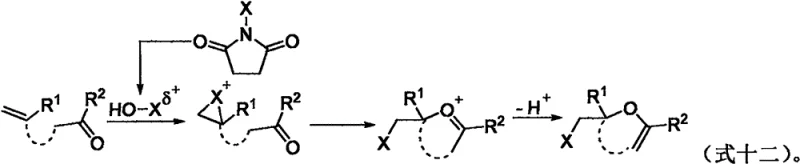
Furthermore, the tolerance of the reaction to various functional groups on the aromatic rings of the substrate underscores its utility for synthesizing complex pharmaceutical intermediates. The electronic nature of the substituents influences the nucleophilicity of the alkene and the stability of the intermediate, yet the aqueous DMSO system appears to buffer these effects effectively. The final deprotonation step restores the conjugated system, yielding the stable 2,3-dihydrofuran product. This deep mechanistic understanding allows process chemists to confidently scale the reaction, knowing that the fundamental driving forces are based on well-established principles of electrophilic addition, thereby reducing the technical risk associated with technology transfer from the laboratory to the pilot plant.
How to Synthesize 2,3-Dihydrofuran Derivatives Efficiently
Implementing this green synthesis route requires careful attention to solvent ratios and reagent stoichiometry to maximize yield and purity. The patented procedure outlines a straightforward operational sequence that begins with the charging of the 1,3-dicarbonyl substrate and the halogenating agent into a reaction vessel. The choice of solvent is critical, with a specific mixture of DMSO and water (optimized at 0.5 mL DMSO to 1.5 mL water) providing the ideal balance of solubility and reactivity. Once the reagents are combined, the mixture is stirred at ambient temperature, and the progress is monitored via thin-layer chromatography (TLC) or gas chromatography (GC). Upon completion, typically achieved within 8 hours, the product is isolated through a standard work-up involving ethyl acetate extraction and column chromatography. For detailed standard operating procedures and specific parameter adjustments for different substrates, please refer to the standardized synthesis guide below.
- Charge a Schlenk reaction flask with the allyl-substituted 1,3-dicarbonyl substrate and the selected halogenating reagent (such as NIS or NBS).
- Add the green solvent system, specifically a mixture of DMSO and water (optimized ratio 0.5 mL DMSO to 1.5 mL water), to the reaction vessel.
- Stir the reaction mixture at room temperature in an open atmosphere until TLC monitoring indicates complete consumption of the starting material, typically requiring approximately 8 hours.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this aqueous halocyclization technology translates into tangible strategic benefits that extend beyond simple yield metrics. The elimination of transition metal catalysts removes the need for expensive scavenging resins and complex filtration steps, directly lowering the cost of goods sold (COGS). Additionally, the use of water as a primary solvent significantly reduces the volume of hazardous organic waste generated, leading to substantial savings in waste disposal fees and environmental compliance costs. The ability to run reactions open to air at room temperature minimizes energy consumption associated with heating, cooling, and inert gas purging, further enhancing the overall economic efficiency of the manufacturing process. These factors collectively contribute to a more resilient and cost-effective supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The catalyst-free nature of this process eliminates the procurement costs associated with precious metal catalysts and the operational costs of their removal. By relying on inexpensive halogenating reagents and a water-based solvent system, the raw material input costs are significantly optimized. Moreover, the simplified work-up procedure reduces labor hours and solvent usage during purification, driving down the overall manufacturing expenditure without compromising on the quality of the final API intermediate.
- Enhanced Supply Chain Reliability: Operating under ambient conditions with readily available reagents mitigates the risk of supply disruptions often caused by the scarcity of specialized catalysts or dry solvents. The robustness of the reaction against atmospheric moisture and oxygen means that production can continue with minimal downtime for equipment maintenance or atmosphere control. This reliability ensures consistent delivery schedules for downstream customers, strengthening the partnership between the supplier and the pharmaceutical manufacturer.
- Scalability and Environmental Compliance: The use of water as a reaction medium inherently improves the safety profile of the process, making it easier to scale from kilogram to multi-ton production without encountering the thermal runaway risks associated with volatile organic solvents. This green chemistry approach aligns perfectly with increasingly stringent global environmental regulations, future-proofing the supply chain against potential regulatory hurdles and enhancing the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green halocyclization technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a transparent view of the process capabilities. Understanding these details helps stakeholders make informed decisions about integrating this methodology into their existing production workflows. For further technical clarification or custom feasibility studies, our team is available to provide detailed route assessments.
Q: Does this halocyclization process require expensive transition metal catalysts?
A: No, the patented method operates without any additive or catalyst system. It relies on the intrinsic electrophilicity of the halogenating reagent in a polar aqueous medium, which significantly simplifies the purification process and reduces raw material costs.
Q: What is the environmental impact of the solvent system used in this synthesis?
A: The process utilizes a DMSO/Water mixed solvent system. Water is the primary component, making the process inherently greener and safer compared to traditional methods that rely on volatile organic compounds (VOCs) or toxic chlorinated solvents.
Q: Can this reaction be performed under standard atmospheric conditions?
A: Yes, one of the key advantages of this technology is that the reaction proceeds efficiently at room temperature while open to the air. This eliminates the need for inert gas protection (such as nitrogen or argon), lowering operational complexity and energy consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydrofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green synthetic methodologies like the aqueous halocyclization described in CN112159375B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required by top-tier pharmaceutical companies. We are committed to leveraging such advanced, eco-friendly technologies to deliver high-purity 2,3-dihydrofuran intermediates that meet the exacting standards of the global market.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this efficient synthetic route. Our technical procurement team is ready to conduct a Customized Cost-Saving Analysis tailored to your specific project needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your drug development timeline while maintaining the highest standards of quality and sustainability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
