Advanced Iridium Catalysis for High-Purity Alpha,Beta-Unsaturated Alcohol Manufacturing
Introduction to Breakthrough Selective Hydrogenation Technology
The landscape of pharmaceutical intermediate synthesis is constantly evolving, driven by the need for greener, more efficient, and highly selective chemical transformations. A pivotal advancement in this domain is detailed in Chinese Patent CN110002952B, which discloses a sophisticated preparation method for α,β-unsaturated alcohols and/or α,β-saturated alcohols. This technology addresses a longstanding challenge in organic synthesis: the chemoselective reduction of α,β-unsaturated aldehydes. Traditionally, distinguishing between the reduction of the carbon-carbon double bond and the carbon-oxygen double bond has required complex catalytic systems or extreme reaction conditions. However, this patented methodology leverages a specialized iridium-based catalytic system to achieve high selectivity under remarkably mild conditions. By utilizing transfer hydrogenation in an air atmosphere at temperatures ranging from 25 to 100°C, the process eliminates the safety hazards associated with high-pressure hydrogen gas. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this innovation represents a significant leap forward in process reliability and cost-efficiency, offering a robust pathway to valuable alcohol derivatives ubiquitous in bioactive compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of α,β-unsaturated aldehydes has been plagued by thermodynamic and kinetic hurdles that complicate industrial application. The hydrogenation of C=C double bonds is thermodynamically more favorable than the selective reduction of C=O bonds, often leading to mixtures of saturated alcohols when the desired product is the unsaturated alcohol. Conventional methods frequently rely on precious metal catalysts like Ruthenium or Platinum-Iron nanoparticles, which often exhibit low turnover frequencies (TOF) or necessitate hazardous operating environments. For instance, prior art involving Ruthenium-catalyzed transfer hydrogenation in aqueous phases has demonstrated TOF values as low as 3.5, rendering the process economically unviable for large-scale manufacturing. Furthermore, alternative approaches using supported Platinum-Iron nanoparticles have required hydrogen pressures as high as 20 bar, demanding expensive, high-pressure resistant reactors and stringent safety protocols. These limitations not only inflate capital expenditure but also introduce significant operational risks and complexity into the supply chain, making cost reduction in fine chemical manufacturing difficult to achieve with legacy technologies.
The Novel Approach
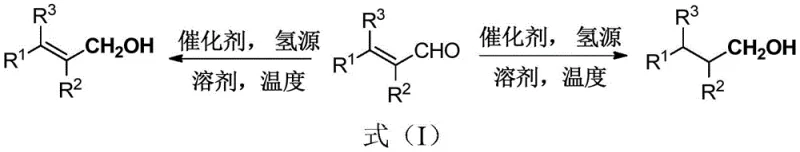
In stark contrast to these cumbersome traditional methods, the novel approach outlined in the patent utilizes a dinitrogen iridium chelate catalyst that operates with exceptional efficiency and selectivity. This method employs transfer hydrogenation using benign hydrogen sources such as formic acid, sodium formate, or triethylamine, thereby removing the need for gaseous hydrogen infrastructure entirely. The reaction proceeds smoothly in common solvents like methanol, ethanol, or water, and critically, the selectivity is tunable simply by adjusting the pH of the reaction medium. Acidic conditions (pH 1-5) drive the selective formation of α,β-unsaturated alcohols, while neutral to basic conditions (pH 7-10) yield fully saturated alcohols. This level of control allows manufacturers to produce distinct high-purity API intermediates from the same starting materials merely by tweaking process parameters. The simplicity of the workup, involving standard ethyl acetate extraction and column chromatography, further underscores the practical superiority of this route, making it an ideal candidate for the commercial scale-up of complex organic syntheses.
Mechanistic Insights into Iridium-Catalyzed Transfer Hydrogenation
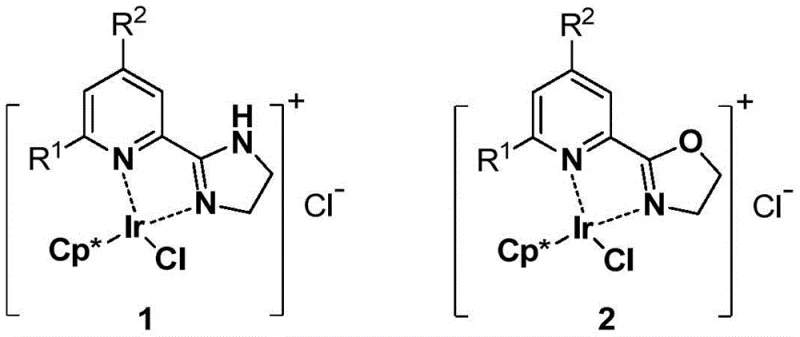
The core of this technological breakthrough lies in the unique electronic and steric properties of the dinitrogen iridium chelate catalysts, specifically structures designated as 1 and 2 in the patent documentation. These catalysts feature a Cp* (pentamethylcyclopentadienyl) ligand coordinated to the iridium center, stabilized by a bidentate nitrogen-containing ligand system. This specific coordination environment modulates the electron density at the metal center, facilitating the activation of the hydrogen source (e.g., formate) and the subsequent hydride transfer to the substrate. The mechanism likely involves the formation of an iridium-hydride species which selectively attacks the electrophilic carbonyl carbon under acidic conditions, preserving the conjugated alkene system. Conversely, under basic conditions, the catalyst may facilitate the reduction of the alkene moiety either through a different mechanistic pathway or by altering the substrate-catalyst interaction dynamics. Understanding this mechanistic nuance is vital for R&D teams aiming to optimize reaction parameters for specific substrates, ensuring that the delicate balance between activity and selectivity is maintained throughout the batch.
Furthermore, the impurity profile of the final product is tightly controlled by the high regioselectivity of this catalytic system. In many conventional reductions, over-reduction or isomerization of the double bond can lead to difficult-to-separate impurities that compromise the purity specifications required for pharmaceutical applications. The iridium-catalyzed process described herein demonstrates a remarkable ability to suppress these side reactions, achieving product ratios of fully reduced to semi-reduced products of greater than 99:1 or vice versa, depending on the pH setting. This high fidelity in chemical transformation minimizes the burden on downstream purification processes. For quality assurance teams, this means a more consistent product with a cleaner impurity spectrum, reducing the risk of batch rejection and ensuring that the stringent purity specifications demanded by global regulatory bodies are consistently met without excessive reprocessing.
How to Synthesize Alpha,Beta-Unsaturated Alcohols Efficiently
Implementing this synthesis route requires careful attention to the stoichiometric ratios and environmental conditions specified in the patent to maximize yield and selectivity. The process begins with the precise weighing of the α,β-unsaturated aldehyde substrate and the dinitrogen iridium chelate catalyst, typically used in molar ratios ranging from 0.0001:1 to 0.1:1 relative to the substrate. The choice of hydrogen source is critical; formic acid is preferred for acidic conditions to generate unsaturated alcohols, while sodium formate is often utilized for basic conditions to produce saturated alcohols. The reaction mixture is stirred in a conventional vessel under an air atmosphere, highlighting the robustness of the catalyst against oxygen, which is a significant advantage over air-sensitive alternatives. Detailed standardized synthetic steps see the guide below.
- Prepare the reaction mixture by combining the alpha,beta-unsaturated aldehyde substrate, dinitrogen iridium chelate catalyst, solvent (methanol, ethanol, or water), and hydrogen source (formic acid or sodium formate).
- Adjust the pH of the solution to control selectivity: pH 1-5 for alpha,beta-unsaturated alcohols or pH 7-10 for alpha,beta-saturated alcohols, then stir at 25-100°C.
- Upon completion, cool the reaction, extract with ethyl acetate, remove solvent under reduced pressure, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iridium-catalyzed technology offers transformative benefits that extend far beyond simple yield improvements. The elimination of high-pressure hydrogen gas removes a major bottleneck in facility utilization, allowing production to occur in standard glass-lined or stainless steel reactors without the need for specialized autoclaves. This flexibility drastically simplifies scheduling and increases plant throughput, directly addressing the industry-wide challenge of reducing lead time for high-purity pharmaceutical intermediates. Moreover, the use of inexpensive and readily available hydrogen sources like formic acid and sodium formate replaces costly gaseous hydrogen supplies, leading to substantial cost savings in raw material procurement. The mild reaction temperatures (25-100°C) also translate to significantly lower energy consumption compared to high-temperature/high-pressure processes, aligning with corporate sustainability goals and reducing the overall carbon footprint of the manufacturing operation.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the removal of expensive infrastructure requirements. By avoiding the need for 20-bar pressure reactors and complex gas handling systems, capital expenditure for new production lines is significantly reduced. Additionally, the high selectivity of the reaction minimizes the formation of byproducts, which reduces the volume of solvents and adsorbents required for purification. This efficiency gain lowers the cost of goods sold (COGS) and improves margin potential for high-volume commodities. The catalyst loading is also remarkably low, often in the range of 0.01 mol% to 0.1 mol%, which mitigates the cost impact of using a precious metal like iridium, ensuring that the overall process remains economically competitive against cheaper but less efficient base metal alternatives.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of stable, shelf-stable reagents that do not require cryogenic storage or specialized transport logistics. Formic acid and sodium formate are commodity chemicals with robust global supply networks, insulating the manufacturing process from the volatility often seen in specialty gas markets. The ability to run the reaction under air atmosphere further de-risk the operation, as it removes the dependency on inert gas blankets (nitrogen or argon) for the reaction phase, although standard safety practices still apply. This operational simplicity ensures consistent batch-to-batch reproducibility, a critical factor for maintaining long-term contracts with multinational pharmaceutical clients who demand unwavering supply continuity.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this technology is exceptionally well-positioned for green chemistry initiatives. The solvent systems employed, such as ethanol-water mixtures, are far more environmentally benign than the chlorinated solvents often required in traditional reductions. The aqueous compatibility of the catalyst allows for easier product isolation and potentially reduces the generation of hazardous organic waste streams. As regulatory scrutiny on pharmaceutical manufacturing waste intensifies, adopting a process that inherently generates less waste and uses safer solvents provides a strategic compliance advantage. The straightforward scale-up from gram-scale laboratory optimization to multi-ton commercial production is facilitated by the lack of exothermic runaway risks associated with high-pressure hydrogenation, making it a safe and scalable solution for growing market demands.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and technical disclosures within the patent literature to provide clarity on process capabilities. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios. The responses cover critical aspects ranging from selectivity control to catalyst recovery, ensuring a comprehensive understanding of the operational landscape.
Q: How is chemoselectivity achieved between C=O and C=C bond reduction?
A: Selectivity is precisely regulated by the pH value of the reaction system. Acidic conditions (pH 1-5) favor the selective reduction of the carbonyl group to yield alpha,beta-unsaturated alcohols, while neutral to basic conditions (pH 7-10) promote total reduction to saturated alcohols.
Q: What are the advantages of this iridium catalyst over traditional ruthenium systems?
A: Unlike traditional ruthenium catalysts which often suffer from low turnover frequencies or require harsh conditions, this dinitrogen iridium chelate operates efficiently under air atmosphere at mild temperatures (25-100°C) without the need for high-pressure hydrogen gas.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes conventional reaction vessels rather than specialized high-pressure autoclaves, significantly simplifying safety protocols and equipment requirements, which facilitates straightforward commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha,Beta-Unsaturated Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN110002952B for the production of high-value pharmaceutical intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready supply is seamless. Our state-of-the-art facilities are equipped to handle sensitive organometallic catalysis with the utmost precision, adhering to stringent purity specifications and rigorous QC labs to guarantee product quality. We understand that in the fast-paced pharmaceutical industry, time-to-market is critical, and our optimized processes are designed to deliver high-purity intermediates with the speed and reliability your projects demand.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific synthesis needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating exactly how this iridium-catalyzed route can optimize your budget. Please contact our technical procurement team today to request specific COA data for similar alcohol derivatives and to discuss route feasibility assessments for your upcoming projects. Let us be your partner in turning complex chemical challenges into commercial successes.
