Scalable Synthesis of American White Moth Pheromone Intermediates via Nickel Boride Catalysis
Scalable Synthesis of American White Moth Pheromone Intermediates via Nickel Boride Catalysis
The global demand for environmentally friendly pest control solutions has intensified the search for efficient synthetic routes to insect sex pheromones. Specifically, the American white moth (Hyphantria cunea) represents a significant quarantine pest threatening forestry and agriculture, necessitating robust monitoring and trapping strategies. Patent CN108299342B discloses a groundbreaking methodology for synthesizing (2S,3R)-2,3-epoxy-1-tetradecanol, a pivotal chiral intermediate required for producing components III and V of the moth's sex pheromone blend. This technical disclosure moves beyond theoretical chemistry to address practical manufacturing bottlenecks, offering a pathway that replaces cryogenic dependencies with ambient temperature protocols. For R&D directors and process chemists, this represents a shift from fragile, high-maintenance laboratory procedures to robust, plant-ready operations. The ability to generate high-purity intermediates with superior Z/E selectivity (>85) without the burden of extreme reaction conditions marks a significant evolution in agrochemical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral epoxy-alcohol intermediates for lepidopteran pheromones has relied heavily on the Sharpless asymmetric epoxidation protocol. While academically elegant, this traditional approach imposes severe constraints on industrial scalability. As illustrated in the prior art synthesis routes, the process typically mandates the use of expensive titanium tetraisopropoxide and diethyl tartrate ligands, coupled with tert-butyl hydroperoxide as an oxidant. Crucially, these reactions must be conducted at cryogenic temperatures ranging from -30°C to -25°C to maintain stereocontrol, requiring substantial energy investment in cooling infrastructure. Furthermore, the sensitivity of titanium catalysts to moisture necessitates rigorously anhydrous conditions and inert gas protection throughout the operation, increasing both capital expenditure for specialized equipment and operational complexity. The cumulative effect is a process that is economically burdensome and operationally fragile, creating significant supply chain risks for manufacturers aiming to produce pheromones at a commercial scale.
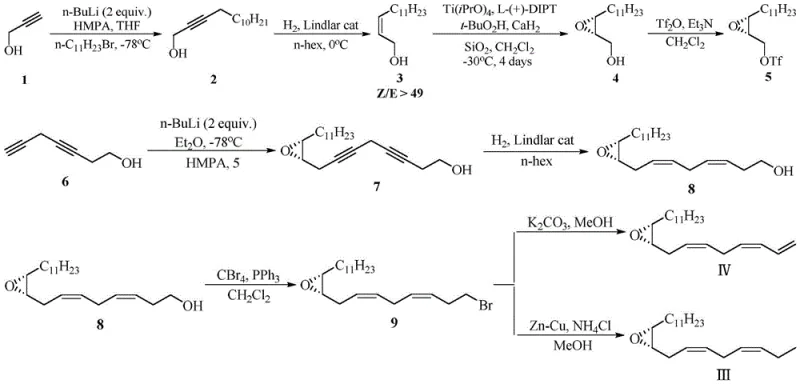
The Novel Approach
In stark contrast, the methodology outlined in CN108299342B introduces a streamlined two-step sequence that fundamentally alters the economic and operational landscape of pheromone intermediate production. The innovation lies in the substitution of precious metal catalysts and cryogenic conditions with an in-situ generated nickel boride (P-2 Nickel) system for the initial hydrogenation step. This modification allows the reduction of the alkyne precursor to proceed efficiently at ambient temperatures (10-30°C) while achieving exceptional Z-selectivity. The subsequent epoxidation step utilizes m-chloroperoxybenzoic acid (m-CPBA) buffered with simple carbonates, eliminating the need for moisture-sensitive titanium complexes. By operating under mild thermal conditions and tolerating the presence of water in the biphasic epoxidation system, this novel approach drastically reduces the technical barriers to entry. For procurement managers, this translates to a supply chain less vulnerable to the logistical challenges of handling hazardous, moisture-sensitive reagents and maintaining sub-zero processing environments.
Mechanistic Insights into Nickel Boride Catalyzed Hydrogenation and Epoxidation
The core of this technological advancement rests on the precise control of stereochemistry during the hydrogenation phase. The process begins with the generation of nickel boride catalyst within the reactor by reacting nickel acetate tetrahydrate with sodium borohydride in the presence of ethylenediamine. This in-situ formation creates a highly active surface capable of facilitating cis-addition of hydrogen across the triple bond of 2-tetradecyn-1-ol. The mechanistic advantage here is twofold: firstly, the nickel boride surface inherently favors the formation of the Z-alkene isomer due to steric constraints during adsorption, yielding a Z/E ratio greater than 85, which is superior to many heterogeneous alternatives. Secondly, the use of ethylenediamine acts as a stabilizer, preventing over-reduction to the saturated alkane. This high fidelity in geometric control is critical because the downstream biological activity of the final pheromone is strictly dependent on the cis-configuration of the double bond relative to the epoxy group.
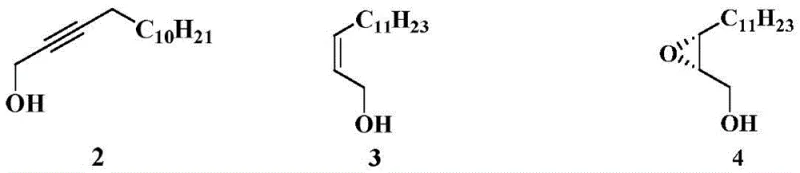
Following the hydrogenation, the stereoselective epoxidation leverages the electron-rich nature of the Z-alkene to react with m-chloroperoxybenzoic acid. The inclusion of a carbonate buffer (such as sodium bicarbonate) serves a dual mechanistic purpose: it neutralizes the m-chlorobenzoic acid byproduct, preventing acid-catalyzed ring opening of the sensitive epoxide, and it maintains a pH environment that favors the desired stereoisomer. Unlike the Sharpless mechanism which relies on a chiral titanium template, this method achieves high diastereoselectivity (dr > 13) and enantiomeric excess (ee = 85%) through the inherent chirality transfer from the preceding steps and the specific transition state stabilized by the buffer system. This robustness against acidic degradation ensures that the yield remains high (up to 85% isolated yield) even during scale-up, providing R&D teams with a reliable platform for generating high-purity agrochemical intermediates without the need for complex chiral auxiliaries.
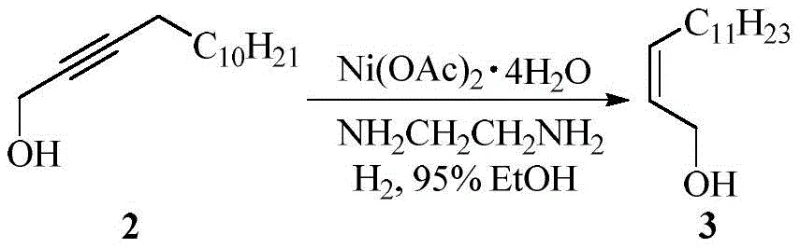
How to Synthesize (2S,3R)-2,3-epoxy-1-tetradecanol Efficiently
Implementing this synthesis route requires careful attention to the preparation of the nickel boride catalyst and the stoichiometry of the epoxidation reagents to maximize yield and purity. The process is designed to be operationally simple, avoiding the need for gloveboxes or specialized low-temperature baths, which facilitates easier technology transfer from the laboratory to the pilot plant. Operators should focus on maintaining the specified molar ratios between nickel acetate and sodium borohydride to ensure complete catalyst formation, as well as controlling the addition rate of the oxidant to manage exothermicity. The following guide outlines the standardized procedural framework derived from the patent examples, serving as a baseline for process optimization and quality control validation.
- Generate nickel boride catalyst in situ from nickel acetate and sodium borohydride, then perform cis-hydrogenation of 2-tetradecyn-1-ol to obtain (Z)-2-tetradecen-1-ol with high Z/E selectivity.
- React the resulting Z-alkene with m-chloroperoxybenzoic acid (m-CPBA) in the presence of a carbonate buffer to achieve stereoselective epoxidation yielding the target chiral epoxy alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the bottom line and supply continuity, the shift from the traditional Sharpless-based protocol to this nickel boride methodology offers profound strategic benefits. The elimination of cryogenic requirements removes a major energy cost center, as maintaining reactors at -30°C demands significant refrigeration capacity and insulation. Furthermore, the replacement of expensive, moisture-sensitive titanium catalysts with commodity chemicals like nickel acetate and sodium borohydride results in substantial raw material cost savings. From a supply chain perspective, the robustness of the reaction conditions means that production is less susceptible to delays caused by equipment failure or stringent environmental controls. The ability to run reactions at ambient temperatures also simplifies the engineering requirements for the manufacturing facility, allowing for faster scale-up and more flexible production scheduling to meet seasonal demand peaks for pest control agents.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of high-cost inputs and energy-intensive unit operations. By avoiding the use of titanium tetraisopropoxide and chiral tartrates, which are costly and require cold chain logistics for storage, the direct material cost is significantly lowered. Additionally, the ambient temperature operation eliminates the continuous electrical load associated with industrial chillers, leading to lower utility bills. The simplified work-up procedure, which does not require rigorous drying of solvents or reagents, further reduces processing time and labor costs, contributing to a more competitive cost structure for the final pheromone intermediate.
- Enhanced Supply Chain Reliability: Operational resilience is markedly improved due to the tolerance of the reaction system to minor variations in moisture and temperature. Traditional methods often suffer from batch failures if water ingress occurs, leading to scrapped batches and supply shortages. In this novel process, the epoxidation step explicitly utilizes an aqueous carbonate solution, meaning that strict anhydrous conditions are unnecessary. This tolerance reduces the risk of batch rejection and ensures a more consistent output of high-purity product. Consequently, procurement managers can rely on more stable lead times and reduced safety stock requirements, knowing that the manufacturing process is inherently more forgiving and robust against typical plant variances.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and waste disposal challenges, but this methodology mitigates those risks effectively. The reagents used, such as nickel salts and peracids, are well-understood in industrial hygiene contexts, and the waste streams are easier to treat compared to those containing heavy metals like titanium or complex organic ligands. The absence of pyrophoric catalysts like Lindlar catalyst (which requires careful quenching) enhances plant safety profiles. Moreover, the high atom economy and selectivity of the reaction minimize the formation of byproducts, reducing the volume of hazardous waste requiring disposal. This alignment with green chemistry principles facilitates smoother regulatory approvals and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of adopting this synthesis route for their specific applications, we have compiled answers to common inquiries regarding the process parameters and quality outcomes. These insights are derived directly from the experimental data provided in the patent documentation, ensuring accuracy and relevance for process development teams. Understanding these nuances is essential for integrating this technology into existing manufacturing workflows and achieving the desired purity specifications for agrochemical registration.
Q: Why is the Z/E ratio critical in the synthesis of American white moth pheromones?
A: The biological activity of the final pheromone components (III and V) is strictly dependent on the stereochemistry of the epoxy group and the geometry of the double bonds. A high Z/E ratio (>85) in the intermediate alkene ensures the correct spatial arrangement for the subsequent epoxidation, directly impacting the enantiomeric excess (ee) and diastereomeric ratio (dr) of the final bioactive molecule.
Q: How does this nickel boride method improve upon the traditional Sharpless epoxidation route?
A: Traditional methods often rely on Sharpless asymmetric epoxidation which requires cryogenic conditions (-30°C), expensive titanium catalysts, and strictly anhydrous environments. The patented nickel boride route operates at mild temperatures (10-30°C), utilizes inexpensive and stable reagents, and tolerates aqueous conditions in the second step, drastically simplifying process control and reducing energy consumption.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is specifically designed for industrial scalability. It eliminates the need for specialized low-temperature reactors and rigorous inert gas protection required by prior art. The use of robust catalysts like in-situ generated nickel boride and common oxidants like m-CPBA allows for safer handling and easier waste management, making it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2S,3R)-2,3-epoxy-1-tetradecanol Supplier
The technical potential of this nickel boride catalyzed route represents a significant opportunity for optimizing the production of pest control intermediates, yet translating patent claims into commercial reality requires experienced partnership. NINGBO INNO PHARMCHEM stands ready to leverage its extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring this efficient synthesis to market. Our facilities are equipped with the necessary hydrogenation and oxidation capabilities to execute this chemistry safely and consistently, adhering to stringent purity specifications and rigorous QC labs to ensure every batch meets the high standards required for pheromone formulation. We understand that the consistency of the Z/E ratio and the enantiomeric excess are non-negotiable for biological efficacy, and our process engineers are dedicated to maintaining these critical quality attributes.
We invite supply chain leaders and R&D directors to engage with us to explore how this optimized route can enhance your product portfolio. By requesting a Customized Cost-Saving Analysis, you can quantify the potential operational efficiencies specific to your volume requirements. Our technical procurement team is prepared to provide specific COA data from pilot runs and detailed route feasibility assessments to support your vendor qualification process. Let us collaborate to secure a sustainable and cost-effective supply of this critical agrochemical intermediate.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
