Advanced One-Step Synthesis of Chiral N-methyl-1-(α-aminobenzyl)-2-naphthol for Asymmetric Catalysis
The landscape of asymmetric catalysis relies heavily on the availability of robust, high-purity chiral ligands that can induce stereocontrol in complex organic transformations. Patent CN101591250A introduces a significant methodological breakthrough in the preparation of chiral amino alcohols, specifically focusing on the synthesis and resolution of N-methyl-1-(α-aminobenzyl)-2-naphthol derivatives. This technology addresses the critical bottleneck of multi-step synthetic routes by proposing a highly efficient one-step Mannich reaction to generate the racemic precursor, followed by a precise chiral resolution strategy using L-(+)-tartaric acid. For R&D directors and procurement specialists in the fine chemical sector, this represents a pivotal shift towards more atom-economical and operationally simple manufacturing processes. The ability to access both (S) and (R) enantiomers of this potent ligand through a streamlined workflow not only enhances the feasibility of large-scale production but also ensures a consistent supply of high-performance catalysts essential for modern drug discovery and agrochemical synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex chiral beta-amino alcohol ligands has been plagued by inefficient, multi-step pathways that suffer from low overall yields and high operational costs. Conventional approaches often require the construction of the carbon skeleton through sequential reactions, involving protection and deprotection strategies that generate substantial chemical waste. As illustrated in the background art of the patent, existing ligands like 1-(α-aminobenzyl)-2-naphthol typically require a three-step synthesis followed by an equally cumbersome three-step fractionation process to achieve optical purity. 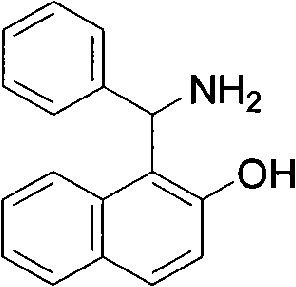 This reliance on lengthy synthetic sequences increases the exposure of intermediates to potential degradation, complicates purification protocols, and drastically inflates the cost of goods sold (COGS). Furthermore, the use of diverse auxiliary reagents across multiple stages creates significant supply chain vulnerabilities, as the availability of each specific reagent becomes a critical path item. For industrial manufacturers, these traditional methods represent a substantial barrier to scaling up production to meet the demands of the global pharmaceutical market.
This reliance on lengthy synthetic sequences increases the exposure of intermediates to potential degradation, complicates purification protocols, and drastically inflates the cost of goods sold (COGS). Furthermore, the use of diverse auxiliary reagents across multiple stages creates significant supply chain vulnerabilities, as the availability of each specific reagent becomes a critical path item. For industrial manufacturers, these traditional methods represent a substantial barrier to scaling up production to meet the demands of the global pharmaceutical market.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN101591250A offers a radically simplified route that collapses the synthesis of the racemic core into a single, elegant step. By leveraging the reactivity of 2-naphthol, benzaldehyde, and methylamine in a direct Mannich condensation, the process eliminates the need for intermediate isolation and complex skeletal construction. The resulting racemic compound, Formula (I), serves as a versatile platform for subsequent chiral resolution. 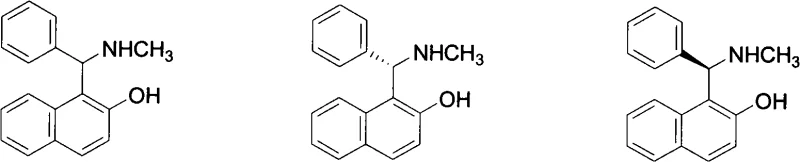 This novel approach not only reduces the total number of reaction steps from six (in conventional methods) to just three (one synthesis plus two resolution steps) but also significantly minimizes the variety of solvents and reagents required. The structural modification of adding a methyl group to the nitrogen atom, as seen in the new compounds compared to the prior art, alters the electronic environment of the catalyst, potentially enhancing its performance in specific asymmetric transformations. This strategic molecular design, combined with process intensification, positions this method as a superior choice for cost reduction in pharmaceutical intermediate manufacturing.
This novel approach not only reduces the total number of reaction steps from six (in conventional methods) to just three (one synthesis plus two resolution steps) but also significantly minimizes the variety of solvents and reagents required. The structural modification of adding a methyl group to the nitrogen atom, as seen in the new compounds compared to the prior art, alters the electronic environment of the catalyst, potentially enhancing its performance in specific asymmetric transformations. This strategic molecular design, combined with process intensification, positions this method as a superior choice for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Mannich Condensation and Chiral Resolution
The core of this synthesis lies in the mechanistic elegance of the Mannich reaction, which facilitates the formation of the carbon-nitrogen bond in a single pot. The reaction proceeds through the in situ generation of an iminium ion intermediate from the condensation of methylamine and benzaldehyde. This electrophilic species is then attacked by the electron-rich aromatic ring of 2-naphthol at the ortho-position relative to the hydroxyl group, driven by the strong activating effect of the phenolic moiety. 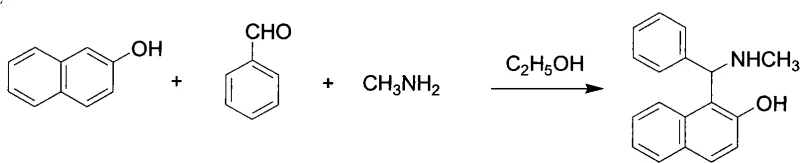 The use of dehydrated ethanol as the solvent is critical, as it solubilizes the reactants while allowing the product to crystallize out upon standing, driving the equilibrium forward. This one-pot methodology avoids the handling of unstable imine intermediates and ensures high atom economy. Following the synthesis, the separation of enantiomers is achieved through diastereomeric salt formation. L-(+)-Tartaric acid acts as a chiral resolving agent, forming a less soluble salt with the (S)-enantiomer of the amine in acetone. This precipitation allows for the physical removal of the (S)-isomer, while the (R)-isomer remains dissolved in the mother liquor, enabling the recovery of both valuable enantiomers from a single batch.
The use of dehydrated ethanol as the solvent is critical, as it solubilizes the reactants while allowing the product to crystallize out upon standing, driving the equilibrium forward. This one-pot methodology avoids the handling of unstable imine intermediates and ensures high atom economy. Following the synthesis, the separation of enantiomers is achieved through diastereomeric salt formation. L-(+)-Tartaric acid acts as a chiral resolving agent, forming a less soluble salt with the (S)-enantiomer of the amine in acetone. This precipitation allows for the physical removal of the (S)-isomer, while the (R)-isomer remains dissolved in the mother liquor, enabling the recovery of both valuable enantiomers from a single batch.
Impurity control in this process is inherently managed by the crystallization dynamics of both the initial Mannich product and the subsequent tartrate salts. The initial white needle-shaped crystals of the racemate can be further purified via recrystallization in toluene, removing unreacted starting materials and side products. During the resolution phase, the specificity of the L-(+)-tartaric acid interaction ensures that only the target stereoisomer precipitates, leaving impurities and the opposite enantiomer in the solution phase. 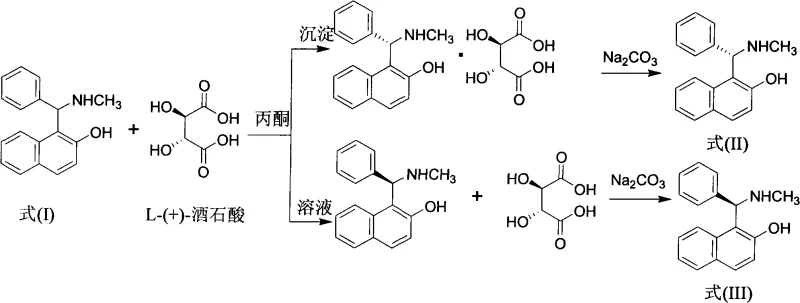 The final liberation of the free base using sodium carbonate and ethyl acetate extraction provides a clean workup procedure that avoids the use of harsh mineral acids or bases that could degrade the sensitive amino alcohol structure. This meticulous control over the solid-state chemistry ensures that the final ligands meet the stringent purity specifications required for high-value asymmetric catalysis applications.
The final liberation of the free base using sodium carbonate and ethyl acetate extraction provides a clean workup procedure that avoids the use of harsh mineral acids or bases that could degrade the sensitive amino alcohol structure. This meticulous control over the solid-state chemistry ensures that the final ligands meet the stringent purity specifications required for high-value asymmetric catalysis applications.
How to Synthesize N-methyl-1-(α-aminobenzyl)-2-naphthol Efficiently
The operational simplicity of this patent makes it highly attractive for pilot plant and commercial scale-up. The process begins with the generation of methylamine gas or the use of aqueous methylamine solutions, which are reacted with 2-naphthol and benzaldehyde in ethanol under mild heating. The reaction mixture is then allowed to stand in the dark, facilitating the slow growth of high-quality crystals. Detailed standardized operating procedures regarding temperature profiles, stirring rates, and crystallization times are essential for reproducibility.
- Conduct a one-step Mannich reaction by reacting 2-Naphthol, Benzaldehyde, and Methylamine in dehydrated ethanol to form the racemic intermediate.
- Perform chiral resolution by treating the racemic mixture with L-(+)-tartaric acid in acetone to precipitate the (S)-enantiomer salt.
- Isolate the (R)-enantiomer from the mother liquor and liberate both free bases using sodium carbonate extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates directly into enhanced operational resilience and significant cost optimization. By collapsing a multi-step synthesis into a single reaction vessel, the process drastically reduces the consumption of solvents, energy, and labor hours associated with intermediate transfers and purifications. This streamlining of the manufacturing workflow eliminates several unit operations, thereby reducing the capital expenditure required for reactor capacity and minimizing the footprint of the production facility. The reliance on commodity chemicals such as 2-naphthol, benzaldehyde, and methylamine ensures a stable and secure raw material supply chain, mitigating the risks associated with sourcing exotic or expensive precursors.
- Cost Reduction in Manufacturing: The elimination of multiple synthetic steps and the reduction in auxiliary reagents lead to a substantial decrease in the overall cost of production. By avoiding the need for complex protection group chemistry and multiple isolation stages, the process minimizes material loss and waste disposal costs. The use of common solvents like ethanol, acetone, and ethyl acetate further contributes to cost efficiency, as these are readily available and inexpensive compared to specialized chlorinated or fluorinated solvents often used in fine chemical synthesis. Additionally, the high yield of the resolution step ensures that the maximum amount of value-added product is recovered from the raw materials input.
- Enhanced Supply Chain Reliability: The simplified process flow reduces the lead time for manufacturing batches, allowing for faster response to market demand fluctuations. Since the synthesis does not rely on sensitive catalysts or cryogenic conditions, it can be executed in standard stainless steel reactors found in most multipurpose chemical plants. This versatility means that production can be easily outsourced or transferred between different manufacturing sites without the need for specialized equipment modifications. The robustness of the crystallization-based purification also ensures consistent product quality, reducing the risk of batch failures and supply disruptions.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory experiments to ton-scale commercial production. The reduction in the number of reaction steps correlates with a lower E-factor (environmental factor), meaning less waste is generated per kilogram of product. The avoidance of heavy metal catalysts and toxic reagents aligns with increasingly strict environmental regulations and green chemistry principles. This environmental compatibility not only simplifies regulatory compliance but also enhances the corporate sustainability profile of the manufacturing organization, which is a key consideration for downstream pharmaceutical customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral ligand synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for technical assessment.
Q: What is the primary advantage of the Mannich reaction route described in CN101591250A?
A: The primary advantage is the drastic simplification of the synthetic route. Unlike conventional methods requiring multiple steps to construct the carbon skeleton, this patent utilizes a direct one-step condensation of 2-naphthol, benzaldehyde, and methylamine, significantly reducing processing time and auxiliary reagent consumption.
Q: How is the chiral purity achieved in this manufacturing process?
A: Chiral purity is achieved through classical resolution using L-(+)-tartaric acid. The racemic amine forms a diastereomeric salt with the chiral acid, where the (S)-enantiomer salt precipitates out of acetone solution due to lower solubility, allowing for physical separation from the (R)-enantiomer which remains in the mother liquor.
Q: What are the typical applications for these chiral amino alcohols?
A: These compounds serve as highly effective chiral ligands in asymmetric catalysis. They are specifically applicable in promoting enantioselective transformations such as nucleophilic additions to carbonyls, asymmetric Diels-Alder reactions, Aldol reactions, and epoxidation reactions, which are critical for synthesizing complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-methyl-1-(α-aminobenzyl)-2-naphthol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance chiral ligands play in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering these complex chiral amino alcohols with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify enantiomeric excess and chemical purity. Our infrastructure is designed to handle the specific solvent systems and crystallization requirements outlined in CN101591250A, guaranteeing a consistent supply of material that meets the exacting standards of the global pharmaceutical industry.
We invite R&D directors and procurement leaders to collaborate with us to leverage this efficient synthetic route for their specific catalytic applications. By partnering with us, you gain access to a Customized Cost-Saving Analysis that demonstrates how switching to this streamlined manufacturing process can optimize your budget without compromising on quality. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your project needs, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
