Revolutionizing Aryl Alpha-Keto Ester Production: A Green Copper-Catalyzed Approach for Commercial Scale-Up
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for sustainable and cost-effective synthetic routes. Patent CN111116368B introduces a groundbreaking methodology for the preparation of aryl α-keto ester compounds, a class of versatile biscarbonyl intermediates critical to the pharmaceutical and agrochemical industries. Traditionally, the synthesis of these valuable building blocks has been plagued by reliance on expensive precious metal catalysts and harsh reaction conditions. This new technology leverages a copper sulfate-catalyzed system in an aqueous medium, operating at a mild temperature of 60°C to achieve high yields. For R&D directors and procurement managers seeking a reliable aryl α-keto ester supplier, this innovation represents a pivotal shift towards greener, more economical production capabilities that do not compromise on purity or structural integrity.
Aryl α-keto esters serve as indispensable precursors in the development of bioactive molecules, including M receptor antagonists like Oxyphenonium Bromide and various enzyme inhibitors used in treating hypertension and inflammation. The ability to access these structures efficiently is paramount for maintaining supply chain continuity in drug development. The patented process utilizes alpha-diazo compounds as starting materials, which are transformed into the target keto esters through a streamlined catalytic cycle. By replacing toxic organic solvents with water and substituting rhodium with copper, this method addresses both economic and environmental pain points simultaneously, offering a robust solution for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl α-keto esters has relied heavily on transition metal catalysis, often utilizing rhodium complexes which are notoriously expensive and subject to volatile market pricing. These conventional protocols typically require refluxing conditions in organic solvents, creating significant safety hazards and increasing the operational expenditure related to solvent recovery and disposal. Furthermore, the use of precious metals introduces stringent regulatory hurdles regarding residual metal limits in final API intermediates, necessitating additional purification steps that lower overall throughput. The complexity of these traditional routes often results in lower yields and limited substrate tolerance, making them less suitable for the diverse library synthesis required in modern medicinal chemistry programs.
The Novel Approach
In stark contrast, the methodology disclosed in CN111116368B employs copper sulfate, an inexpensive and readily available inorganic salt, as the catalyst. This switch from precious metals to base metals fundamentally alters the cost structure of the synthesis, making it highly attractive for large-scale manufacturing. The reaction proceeds in water, a non-toxic, non-flammable, and universally available solvent, which eliminates the generation of hazardous organic waste streams. Operating at a moderate temperature of 60°C reduces energy consumption compared to high-temperature reflux methods. This novel approach not only simplifies the operational workflow but also enhances the safety profile of the manufacturing process, aligning perfectly with the goals of cost reduction in pharmaceutical intermediate manufacturing while maintaining high product quality.
Mechanistic Insights into Copper-Catalyzed Diazo Decomposition
The core of this green synthesis lies in the efficient activation of the alpha-diazo compound by the copper catalyst in an aqueous environment. The reaction initiates with the coordination of the diazo species to the copper center, facilitating the extrusion of nitrogen gas and the formation of a reactive copper-carbenoid intermediate. In the presence of water, this electrophilic species undergoes nucleophilic attack or insertion, ultimately leading to the formation of the carbonyl functionality characteristic of the α-keto ester. The mild conditions prevent the decomposition of sensitive functional groups on the aromatic ring, ensuring high fidelity in the final product structure. Understanding this mechanistic pathway is crucial for optimizing reaction parameters and scaling the process from gram to ton quantities without losing efficiency.
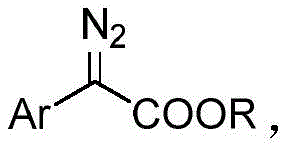
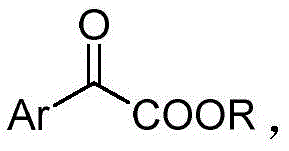
Impurity control is another critical aspect where this method excels. The use of water as a solvent helps to suppress side reactions that are common in organic media, such as dimerization of the carbenoid species or solvent insertion byproducts. The high selectivity of the copper sulfate catalyst ensures that the primary transformation pathway dominates, resulting in a cleaner crude reaction mixture. This inherent purity reduces the burden on downstream purification processes like column chromatography or recrystallization. For quality assurance teams, this means a more consistent impurity profile and easier validation of the manufacturing process, which is essential for meeting the rigorous standards of high-purity OLED material or pharmaceutical intermediate production.
How to Synthesize Ethyl Benzoylformate Efficiently
The practical implementation of this synthesis is straightforward and designed for ease of operation in standard chemical reactors. The process involves simply dispersing the alpha-diazo precursor and the copper sulfate catalyst in water, followed by heating to the specified temperature. The detailed standardized synthesis steps, including precise stoichiometric ratios and workup procedures, are outlined below to guide technical teams in replicating these results. This simplicity allows for rapid technology transfer from the laboratory to pilot plants, minimizing the downtime usually associated with adopting new synthetic routes.
- Prepare the reaction mixture by dispersing the alpha-diazo compound substrate and copper sulfate catalyst (molar ratio 1: 0.2) in water.
- Heat the aqueous suspension to 60°C and maintain stirring for the required reaction time (typically 1-20 hours depending on substrate).
- Extract the product using dichloromethane, dry the organic phase over anhydrous sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed aqueous method offers transformative benefits that extend beyond simple yield improvements. The elimination of expensive rhodium catalysts directly translates to substantial cost savings in raw material procurement, insulating the supply chain from the volatility of precious metal markets. Furthermore, the use of water as a solvent drastically simplifies waste management protocols, reducing the environmental compliance costs associated with hazardous organic solvent disposal. These factors combined create a more resilient and cost-efficient supply chain for aryl α-keto esters.
- Cost Reduction in Manufacturing: The substitution of rhodium with copper sulfate removes one of the most significant cost drivers in traditional synthesis. Since copper is a base metal with stable pricing, manufacturers can achieve significant cost reduction in pharmaceutical intermediate manufacturing without sacrificing reaction efficiency. Additionally, the removal of heavy metal scavenging steps further lowers processing costs, making the final product more competitive in the global market.
- Enhanced Supply Chain Reliability: Relying on abundant and cheap reagents like copper sulfate and water ensures that production is not hindered by the scarcity of specialized catalysts. This availability enhances supply chain reliability, allowing for consistent production schedules and reducing lead time for high-purity pharmaceutical intermediates. The robustness of the reaction conditions also means fewer batch failures due to sensitive reagent degradation.
- Scalability and Environmental Compliance: The aqueous nature of the reaction makes it inherently safer and easier to scale up to industrial volumes. The absence of flammable organic solvents reduces fire risks in large reactors, while the green chemistry profile facilitates easier regulatory approval. This scalability supports the commercial scale-up of complex polymer additives or drug intermediates, ensuring that supply can meet growing market demand sustainably.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this green synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the method's applicability and advantages for potential partners.
Q: Why is copper sulfate preferred over rhodium catalysts for this synthesis?
A: Copper sulfate is significantly cheaper and more abundant than rhodium salts, drastically reducing raw material costs. Additionally, it eliminates the need for rigorous heavy metal removal steps often required with precious metal catalysts in pharmaceutical applications.
Q: What are the environmental benefits of using water as a solvent?
A: Using water eliminates the need for volatile organic compounds (VOCs), reducing fire hazards and toxic waste generation. This aligns with green chemistry principles and simplifies wastewater treatment processes compared to traditional organic solvent systems.
Q: Does this method support diverse substrate functional groups?
A: Yes, the method demonstrates broad substrate universality, successfully tolerating electron-withdrawing groups like nitro and trifluoromethyl, as well as electron-donating groups like methoxy and halogens, ensuring versatility for complex intermediate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Alpha-Keto Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting innovative synthetic technologies like the one described in CN111116368B to enhance our service offerings. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the highest industry standards, providing you with a reliable aryl α-keto ester supplier you can trust for critical projects.
We invite you to collaborate with us to leverage this green chemistry breakthrough for your next development program. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this copper-catalyzed route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of these vital chemical intermediates.
