Advanced One-Pot Synthesis of Chiral Cyclopropyl Amino Acids for Scalable Pharmaceutical Manufacturing
Advanced One-Pot Synthesis of Chiral Cyclopropyl Amino Acids for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways that balance structural complexity with operational simplicity, particularly for chiral building blocks used in enzyme inhibitors and biological probes. Patent CN107445894B, published in late 2020, introduces a significant methodological advancement in the preparation of chiral cyclopropyl amino acids, a critical scaffold in modern medicinal chemistry. This technology leverages the unique properties of cesium carbonate to drive a cyclization reaction under remarkably mild conditions, specifically at room temperature, thereby eliminating the energy-intensive heating steps often required in traditional heterocyclic synthesis. For R&D directors and process chemists, this represents a pivotal shift towards greener, more efficient manufacturing protocols that maintain high stereochemical integrity while drastically reducing the operational burden associated with multi-step sequences. The ability to generate these complex architectures directly from readily available sulfonamide and chlorocyclopropane precursors underscores a strategic evolution in how we approach the construction of nitrogen-containing heterocycles for drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyclopropyl amino acid derivatives has been plagued by procedural inefficiencies that hinder large-scale adoption and increase the cost of goods sold. Traditional routes often rely on the condensation of aminocyclopropyl carboxylic acids with complex precursors like 1,2,3,4-tetrahydroquinoline-2-carboxylic acid, a process characterized by an excessive number of reaction steps and the generation of numerous unstable intermediates. These convoluted pathways not only depress overall reaction yields due to cumulative losses at each stage but also create significant challenges in impurity profiling and purification, as separating structurally similar byproducts becomes increasingly difficult with each additional transformation. Furthermore, the reliance on harsh reaction conditions or expensive transition metal catalysts in older methodologies often necessitates rigorous downstream processing to remove trace metals, adding both time and cost to the manufacturing timeline. For procurement managers, these inefficiencies translate into higher raw material costs and longer lead times, while supply chain heads face increased risks associated with the availability of specialized reagents and the management of hazardous waste streams generated by complex synthetic sequences.
The Novel Approach
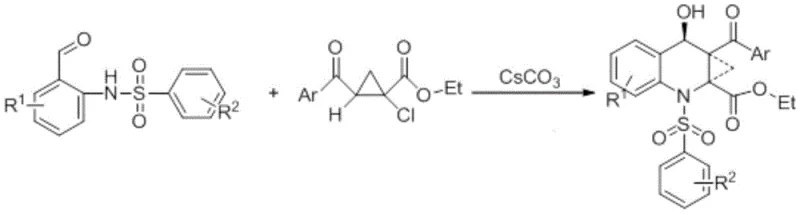
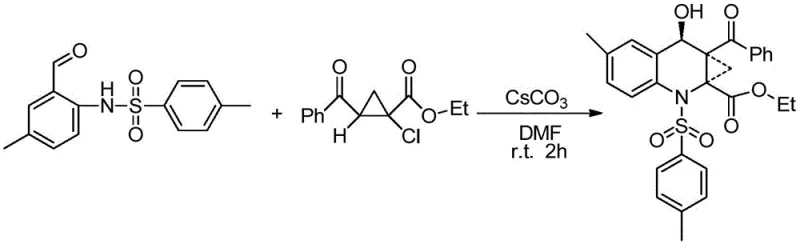
In stark contrast to these legacy methods, the technology disclosed in CN107445894B offers a streamlined, one-pot solution that fundamentally simplifies the production landscape for these valuable intermediates. By utilizing N-(2-formyl-4-tolyl)-4-toluenesulfonamide and ethyl-2-benzoyl-1-chlorocyclopropane carboxylic acid as starting materials, the process achieves the desired cyclization in a single operational step with high atom economy. The use of cesium carbonate as a promoter in N,N-dimethylformamide (DMF) allows the reaction to proceed efficiently at room temperature, removing the need for thermal activation and significantly lowering energy consumption. This approach not only accelerates the reaction kinetics, typically completing within 1 to 2 hours, but also ensures a cleaner reaction profile that facilitates easier isolation of the target product. The simplicity of the workup procedure, involving standard aqueous extraction and column chromatography, makes this method highly attractive for industrial scale-up, offering a reliable pharmaceutical intermediate supplier pathway that minimizes both environmental impact and operational complexity.
Mechanistic Insights into Cesium Carbonate Catalyzed Cyclization
The efficacy of this synthetic route is deeply rooted in the unique physicochemical properties of cesium carbonate, which acts as more than just a simple base in this transformation. The cesium ion possesses a softer Lewis acidity compared to smaller alkali metal cations like potassium or sodium, which enhances its solubility in polar aprotic organic solvents such as DMF, DMSO, and ethers. This superior solubility ensures a homogeneous reaction environment where the carbonate anion is highly available to deprotonate the sulfonamide nitrogen, generating a nucleophilic species capable of attacking the electrophilic centers on the chlorocyclopropane derivative. The mechanism likely involves an initial nucleophilic substitution followed by an intramolecular aldol-type condensation or cyclization to form the fused quinoline-cyclopropane core. The mild basicity of cesium carbonate is sufficient to drive this cascade without promoting unwanted side reactions such as ring-opening of the strained cyclopropane moiety, which is a common pitfall when using stronger, less selective bases. This delicate balance of reactivity is crucial for maintaining the structural integrity of the chiral center and ensuring high product purity.
From an impurity control perspective, the choice of solvent and base plays a pivotal role in minimizing the formation of byproducts that could complicate downstream purification. Experimental data within the patent demonstrates that DMF is the superior solvent choice, yielding significantly higher conversion rates compared to acetonitrile, THF, or DMSO, likely due to its ability to stabilize the transition states involved in the cyclization. The reaction's tolerance for ambient temperature further reduces the risk of thermal degradation or polymerization of sensitive intermediates, which are common issues in heated processes. Additionally, the straightforward extraction protocol using water and dichloromethane effectively removes inorganic salts and polar impurities, leaving the organic phase enriched with the target molecule. This inherent selectivity reduces the burden on the final purification step, allowing for high-purity isolation via standard silica gel chromatography without the need for preparative HPLC, thus enhancing the overall feasibility of cost reduction in API manufacturing.
How to Synthesize Chiral Cyclopropyl Amino Acid Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters identified in the patent to ensure reproducibility and maximum yield. The process begins with the precise weighing of the sulfonamide aldehyde and the chlorocyclopropane ester, which are then dissolved in anhydrous DMF to create a homogeneous solution. The addition of cesium carbonate must be controlled to manage any exotherm, although the reaction is generally mild at room temperature. Monitoring the reaction progress via thin-layer chromatography (TLC) on F-254 plates is essential to determine the exact endpoint, preventing over-reaction or decomposition. Once the starting materials are consumed, the workup involves a liquid-liquid extraction that separates the organic product from the inorganic cesium salts, followed by drying and concentration. The detailed standardized synthesis steps for this procedure are outlined below to guide technical teams in replicating this high-efficiency protocol.
- Mix N-(2-formyl-4-tolyl)-4-toluenesulfonamide, ethyl-2-benzoyl-1-chlorocyclopropane carboxylic acid, and cesium carbonate in DMF solvent at a molar ratio of 1: 1:2.
- Stir the reaction mixture at room temperature for 1 to 2 hours until TLC analysis indicates completion.
- Extract the product with water and dichloromethane, dry the organic layer, and purify the crude residue via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this cesium carbonate-mediated synthesis offers tangible benefits that extend beyond mere chemical elegance, directly impacting the bottom line through operational efficiencies. The elimination of multiple synthetic steps and the avoidance of expensive transition metal catalysts significantly reduce the raw material bill of materials, while the simplified workup procedure lowers labor costs and solvent consumption. Because the reaction proceeds at room temperature, there is no requirement for specialized heating equipment or the energy expenditure associated with maintaining elevated temperatures over long periods, contributing to substantial cost savings in utility usage. Furthermore, the high selectivity of the reaction minimizes the generation of hazardous waste, aligning with increasingly stringent environmental regulations and reducing the costs associated with waste disposal and compliance reporting. These factors combine to create a more resilient and cost-effective supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the drastic simplification of the synthetic route, which eliminates the need for isolating and purifying multiple intermediates that characterize conventional methods. By consolidating the synthesis into a single pot, manufacturers save significantly on solvent volumes, filtration media, and reactor occupancy time, all of which are major cost centers in fine chemical production. The use of cesium carbonate, while potentially more expensive per kilogram than simple bases like potassium carbonate, is justified by the dramatic increase in yield and purity, which reduces the overall amount of starting material required to produce a kilogram of final product. Additionally, the absence of heavy metal catalysts removes the necessity for costly scavenging resins or complex metal removal protocols, further streamlining the production budget and enhancing the economic viability of the process.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials, such as substituted sulfonamides and chlorocyclopropane derivatives, ensures a robust supply chain that is less susceptible to disruptions caused by the scarcity of exotic reagents. The mild reaction conditions also mean that the process can be executed in a wider range of manufacturing facilities without requiring specialized high-pressure or high-temperature infrastructure, increasing the pool of potential contract manufacturing organizations (CMOs) capable of producing the material. This flexibility allows for better risk mitigation and diversification of supply sources, ensuring continuous availability of the intermediate for downstream drug substance manufacturing. Moreover, the short reaction time of 1 to 2 hours enables faster batch turnover, allowing suppliers to respond more agilely to fluctuations in demand and reducing the lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or ton scale is facilitated by the homogeneous nature of the reaction mixture and the lack of aggressive exotherms, making it inherently safer and easier to control in large reactors. The use of DMF, while requiring careful handling, is a well-understood solvent in the industry with established recovery and recycling protocols, minimizing the environmental footprint of the process. The high atom economy and reduced waste generation align with green chemistry principles, helping companies meet their sustainability goals and regulatory obligations regarding solvent emissions and hazardous waste disposal. This environmental compatibility not only reduces compliance costs but also enhances the corporate social responsibility profile of the manufacturing operation, making it a more attractive partner for global pharmaceutical clients who prioritize sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method, providing clarity for stakeholders evaluating its adoption. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for process development teams. Understanding these nuances is critical for assessing the feasibility of integrating this technology into existing manufacturing workflows and for optimizing the production of chiral cyclopropyl amino acid derivatives.
Q: Why is cesium carbonate preferred over other bases for this cyclization?
A: Cesium carbonate exhibits superior solubility in organic solvents like DMF compared to potassium carbonate or sodium acetate, acting as a more effective inorganic base due to the softer Lewis acidity of the cesium ion, which facilitates the reaction at room temperature.
Q: What are the optimal reaction conditions for maximum yield?
A: The patent data indicates that using N,N-dimethylformamide (DMF) as the solvent at room temperature with a reaction time of 1-2 hours yields the best results, achieving up to 82% isolated yield compared to other solvents like THF or acetonitrile.
Q: How does this method improve upon traditional synthesis routes?
A: Unlike conventional methods involving cumbersome multi-step sequences with tetrahydroquinoline derivatives, this novel approach achieves the target structure in a single pot with fewer intermediates, significantly simplifying post-treatment and reducing environmental waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Cyclopropyl Amino Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies that drive innovation in drug development while maintaining commercial viability. Our team of expert chemists has extensively evaluated the methodology described in CN107445894B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this efficient process to life. We are committed to delivering high-quality intermediates that meet stringent purity specifications, leveraging our rigorous QC labs to ensure every batch conforms to the highest standards of identity and potency. Our state-of-the-art facilities are equipped to handle the specific solvent systems and purification techniques required for this chemistry, ensuring a seamless transition from laboratory scale to full-scale manufacturing.
We invite pharmaceutical partners to collaborate with us to leverage this cutting-edge synthesis for their pipeline projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this streamlined route can optimize your supply chain economics. We encourage you to contact us today to discuss your project needs,索取 specific COA data for our reference standards, and review our comprehensive route feasibility assessments. Let us be your trusted partner in navigating the complexities of chiral intermediate synthesis, ensuring your projects move forward with speed, efficiency, and confidence.
