Advanced Synthesis of Cabazitaxel Precursors: Technical Upgrades for Commercial Manufacturing
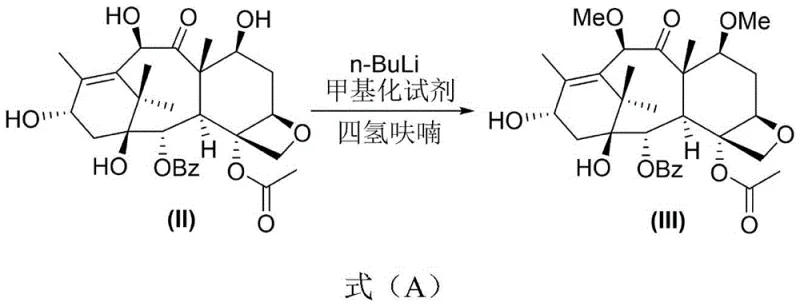
The landscape of oncology drug manufacturing is constantly evolving, driven by the need for more efficient synthesis routes for complex taxane derivatives. Patent CN110746382B introduces a significant breakthrough in the preparation of 7,10-dimethoxy-10-DAB, a critical precursor for Cabazitaxel, which is widely used in treating hormone-refractory metastatic prostate cancer. This innovation addresses long-standing challenges in the semi-synthetic production of taxanes, specifically focusing on improving yield and simplifying purification processes that have historically bottlenecked production capacity. By leveraging a selective methylation strategy using n-BuLi, the disclosed method offers a robust pathway that enhances the overall feasibility of producing high-purity pharmaceutical intermediates. For R&D directors and procurement specialists, understanding this technological shift is vital for securing a reliable pharmaceutical intermediates supplier capable of meeting stringent quality and volume demands in the global market.
Cabazitaxel represents a next-generation taxane with superior activity against drug-resistant tumor cells, largely due to its reduced affinity for P-glycoprotein. However, the commercial viability of such potent agents depends heavily on the efficiency of their synthetic routes. The traditional methods for obtaining the 7,10-dimethoxy-10-DAB core often suffer from low yields and the generation of difficult-to-remove by-products, necessitating costly and time-consuming column chromatography. This new approach fundamentally reengineers the synthesis logic, proposing a direct methylation of 10-DAB that bypasses unnecessary protection groups. Such improvements are not merely academic; they translate directly into cost reduction in API intermediate manufacturing and ensure a more stable supply chain for downstream drug developers seeking to mitigate production risks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 7,10-dimethoxy-10-DAB has been plagued by inefficiencies that hinder large-scale commercialization. Conventional literature and prior art often describe routes that involve multiple steps of hydroxyl protection and deprotection, which drastically reduce atom economy and increase the generation of chemical waste. These multi-step sequences not only extend the production timeline but also introduce numerous opportunities for yield loss at each stage, compounding the overall cost of goods. Furthermore, the post-treatment methods traditionally employed rely heavily on column chromatography for purification, a technique that is notoriously difficult to scale up and poses significant environmental and operational burdens in an industrial setting. The accumulation of these factors results in a process that is economically unsustainable for high-volume production, creating supply vulnerabilities for manufacturers of complex polymer additives or pharmaceutical intermediates who depend on consistent raw material availability.
The Novel Approach
In stark contrast, the methodology outlined in CN110746382B streamlines the synthesis by utilizing a direct selective methylation strategy that eliminates the need for cumbersome protection groups. By employing excess n-BuLi and a specific methylating agent in tetrahydrofuran, the reaction achieves high selectivity for the 7 and 10 positions, directly converting 10-DAB into the desired dimethoxy derivative. This reduction in synthetic steps inherently lowers the consumption of reagents and solvents, contributing to substantial cost savings and a smaller environmental footprint. Moreover, the purification process is revolutionized by replacing chromatography with a simple recrystallization step using mixed solvents, which is far more amenable to industrial scale-up. This novel approach ensures that the commercial scale-up of complex taxane derivatives becomes a viable reality, offering a competitive edge to suppliers who can adopt this technology to enhance their production capabilities.
Mechanistic Insights into n-BuLi-Catalyzed Selective Methylation
The core of this technological advancement lies in the precise control of the methylation reaction using n-BuLi as a strong base to deprotonate the hydroxyl groups at the 7 and 10 positions of the 10-DAB scaffold. The mechanism involves the formation of lithium alkoxides which then react with the methylating agent, such as methyl triflate or methyl iodide, to install the methoxy groups with high regioselectivity. Maintaining the reaction temperature between -78°C and -35°C is critical to suppress side reactions and ensure that the lithiation occurs exclusively at the desired sites without affecting other sensitive functional groups on the taxane core. This level of control is essential for maintaining the structural integrity of the molecule, which is paramount for the biological activity of the final drug substance. Understanding this mechanistic nuance allows R&D teams to optimize reaction parameters further, ensuring that the process remains robust even when transitioning from laboratory benchtop to pilot plant scales.
Impurity control is another critical aspect where this method excels, primarily due to the simplicity of the workup and purification stages. In traditional syntheses, side products often co-elute with the desired compound, requiring rigorous chromatographic separation that can degrade the product or leave residual solvents. The new method's reliance on recrystallization from specific solvent systems, such as ethyl acetate and petroleum ether, leverages solubility differences to effectively exclude impurities. This results in a final product with high purity specifications, reducing the burden on quality control labs and minimizing the risk of batch rejection. For procurement managers, this implies a lower risk profile associated with the supply of high-purity pharmaceutical intermediates, as the process is inherently designed to minimize variability and ensure consistent quality across different production batches.
How to Synthesize 7,10-dimethoxy-10-DAB Efficiently
The synthesis protocol described in the patent provides a clear roadmap for producing this valuable intermediate with high efficiency and reproducibility. The process begins with the careful preparation of the reaction mixture under inert atmosphere, followed by the controlled addition of reagents to manage exotherms and ensure safety. Detailed standard operating procedures for this synthesis are essential for maintaining consistency, particularly regarding the stoichiometry of n-BuLi and the methylating agent. The subsequent purification via recrystallization is straightforward yet requires precise temperature management to maximize recovery yields. For technical teams looking to implement this route, adhering to the specified molar ratios and solvent compositions is key to unlocking the full potential of this method.
- Perform selective methylation of 10-DAB using n-BuLi and a methylating agent in THF at low temperatures (-78°C to -35°C) to generate 7,10-dimethoxy-10-DAB.
- Purify the crude methylation product via recrystallization in a mixed solvent system of esters and ethers to ensure high purity without column chromatography.
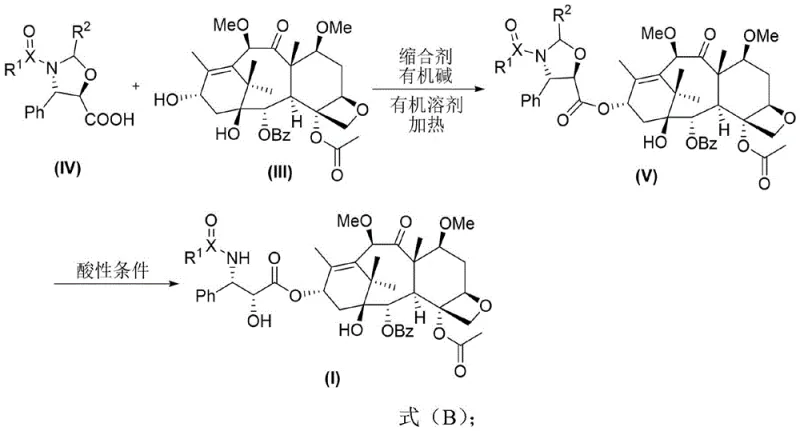
- Conduct condensation with oxazoline carboxylic acid derivatives using DCC and DMAP, followed by acidic hydrolysis to yield the final cabazitaxel precursor derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers profound benefits for supply chain stability and cost management. The elimination of column chromatography and the reduction in reaction steps directly translate to shorter manufacturing cycles, which is crucial for reducing lead time for high-purity pharmaceutical intermediates. This efficiency gain allows suppliers to respond more rapidly to market demands, ensuring that downstream drug manufacturers do not face delays in their own production schedules. Furthermore, the use of readily available raw materials and common solvents mitigates the risk of supply disruptions caused by specialized reagent shortages. These factors combined create a more resilient supply chain that can withstand external pressures and maintain continuous operation.
- Cost Reduction in Manufacturing: The streamlined process significantly lowers the cost of goods by reducing the consumption of expensive reagents and solvents associated with multi-step protection strategies. By avoiding chromatography, the method also cuts down on silica gel usage and solvent waste disposal costs, which are significant overheads in fine chemical manufacturing. The higher yield obtained through selective methylation means more product is generated per unit of starting material, further enhancing the economic viability of the process. These cumulative savings allow for more competitive pricing structures without compromising on the quality of the final intermediate.
- Enhanced Supply Chain Reliability: The simplicity of the reaction conditions and the use of standard industrial solvents make this process highly robust and less prone to operational failures. This reliability ensures a consistent output of 7,10-dimethoxy-10-DAB, which is critical for maintaining the continuity of Cabazitaxel production lines. Suppliers adopting this method can offer greater assurance of delivery, reducing the inventory buffers that buyers often need to hold. This stability is invaluable for strategic planning and long-term contracting in the volatile pharmaceutical market.
- Scalability and Environmental Compliance: The process is inherently designed for scale, with purification steps that are easily adapted to large-scale crystallizers and filters. This scalability ensures that production can be ramped up quickly to meet surging demand without the need for complex equipment modifications. Additionally, the reduction in waste generation aligns with increasingly stringent environmental regulations, making it a sustainable choice for modern chemical manufacturing. This compliance reduces regulatory risks and enhances the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. They are derived from the specific advantages and procedural details outlined in the patent documentation to provide clarity for potential partners. Understanding these aspects is crucial for evaluating the feasibility of integrating this technology into existing production frameworks.
Q: How does the new methylation method improve upon conventional 7,10-dimethoxy-10-DAB synthesis?
A: The patented method utilizes excess n-BuLi for selective methylation, eliminating the need for multi-step protection and deprotection sequences. This significantly reduces reaction steps and avoids the extensive column chromatography typically required, leading to higher atom economy and simplified post-treatment.
Q: What are the critical reaction conditions for ensuring high yield in this process?
A: Maintaining strict temperature control between -78°C and -35°C during the n-BuLi addition is crucial to prevent side reactions. Additionally, the molar ratio of 10-DAB to n-BuLi to methylating agent should be optimized around 1:3.5:4 to ensure complete conversion while minimizing waste.
Q: Is this synthesis route scalable for industrial production of taxane intermediates?
A: Yes, the process is designed with industrial scalability in mind. The use of common solvents like THF and toluene, combined with a recrystallization purification step instead of chromatography, makes it highly suitable for large-scale manufacturing of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7,10-dimethoxy-10-DAB Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthesis technologies like the one described in CN110746382B for the production of critical oncology intermediates. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which guarantee that every batch of 7,10-dimethoxy-10-DAB meets the highest international standards. We are dedicated to supporting our partners in bringing life-saving medications to market faster and more efficiently through our technical expertise and manufacturing capabilities.
We invite you to collaborate with us to leverage these technological advancements for your specific supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project requirements, demonstrating how this optimized route can benefit your bottom line. Please contact us to request specific COA data and route feasibility assessments, and let us help you secure a stable and cost-effective supply of high-quality pharmaceutical intermediates for your future projects.
