Advanced Synthesis of Substituted Tetrahydroquinoline-4-one Hydrochloride for Industrial Scale-up
Introduction to Patent CN102382050A Technology
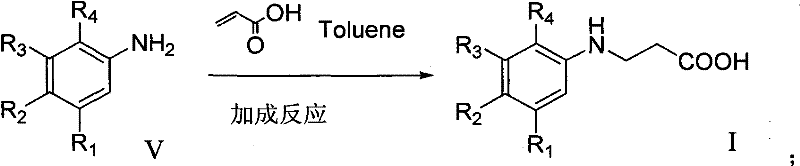
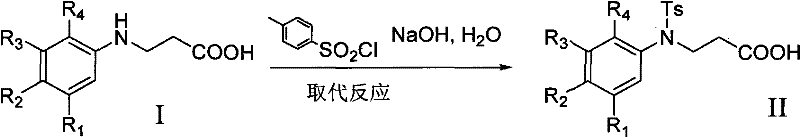
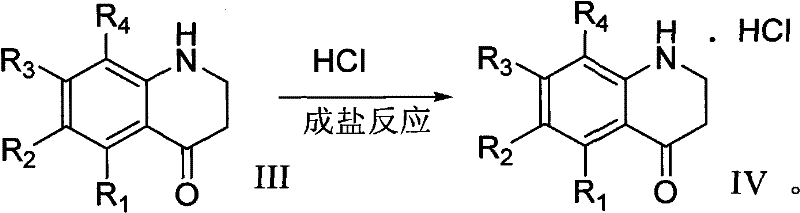
The pharmaceutical and agrochemical industries constantly demand efficient, scalable routes for complex heterocyclic scaffolds, and the technology disclosed in patent CN102382050A represents a significant advancement in the preparation of substituted 1,2,3,4-tetrahydroquinoline-4-one hydrochloride derivatives. This specific class of compounds serves as a critical building block for numerous bioactive molecules, yet traditional synthetic pathways have often been plagued by low yields and difficult purification challenges that hinder commercial viability. The invention outlines a robust four-step sequence that begins with the addition reaction of aniline or substituted aniline with acrylic acid, followed by a strategic sulfonyl protection, cyclization using polyphosphoric acid, and final salt formation. By integrating a protection-deprotection strategy, this methodology effectively bypasses the formation of intractable impurities that typically necessitate costly chromatographic separation, thereby offering a streamlined pathway for the reliable pharmaceutical intermediate supplier seeking to optimize their manufacturing portfolio.
Furthermore, the versatility of this synthetic approach allows for the introduction of various substituents such as chlorine, bromine, methyl, ethyl, and methoxy groups at the 5, 6, 7, and 8 positions of the quinoline ring system, providing substantial flexibility for medicinal chemistry campaigns. The process is characterized by its use of common organic solvents like toluene and standard reagents, ensuring that the barrier to entry for adoption is low while the output quality remains exceptionally high. For R&D directors evaluating new process technologies, the emphasis on moderate reaction conditions and high product content makes this patent a compelling candidate for technology transfer, particularly when aiming for cost reduction in pharmaceutical intermediate manufacturing where purity specifications are stringent and regulatory compliance is paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tetrahydroquinoline-4-one derivatives has relied heavily on direct cyclization strategies where the amino group is not protected prior to the ring-closing step. While conceptually straightforward, these conventional methods suffer from severe drawbacks when translated to an industrial setting, primarily due to the lack of selectivity which leads to complex reaction mixtures and significant byproduct formation. The resulting crude products often possess very poor purity profiles, rendering them unsuitable for subsequent downstream reactions without extensive purification efforts. In many documented cases, the only viable method to achieve acceptable purity levels involves column chromatography, a technique that is notoriously difficult to scale, consumes vast amounts of solvent, and introduces significant bottlenecks in production throughput. Consequently, existing synthetic technologies for unsubstituted or simply substituted variants are often deemed unsuitable for amplification and mass production, creating a supply chain vulnerability for manufacturers who rely on these key intermediates for their final active pharmaceutical ingredients.
The Novel Approach
In stark contrast to the limitations of direct cyclization, the novel approach detailed in the patent data introduces a pivotal sulfonyl protection step that fundamentally alters the reaction landscape to favor high-yield, high-purity outcomes. By converting the intermediate amine into a sulfonamide derivative using p-toluenesulfonyl chloride, the nucleophilicity of the nitrogen is modulated, preventing unwanted side reactions during the subsequent acidic cyclization phase. This strategic modification allows the cyclization to proceed cleanly under the influence of polyphosphoric acid, after which the protecting group is efficiently removed under mild basic conditions. The result is a crude product of such high quality that it bypasses the need for column chromatography entirely, relying instead on simple extraction and crystallization techniques. This shift from complex purification to straightforward workup procedures represents a paradigm shift in process chemistry, enabling the commercial scale-up of complex pharmaceutical intermediates with drastically reduced operational complexity and waste generation.
Mechanistic Insights into Ts-Protection and PPA Cyclization
The core chemical innovation of this process lies in the precise orchestration of functional group transformations, beginning with the Michael addition of the aniline nitrogen to the beta-carbon of acrylic acid. This initial step, conducted in toluene at temperatures ranging from 25°C to 135°C, generates the beta-amino propionic acid derivative which serves as the precursor for the cyclization scaffold. Following this, the introduction of the tosyl group via reaction with p-toluenesulfonyl chloride in the presence of an alkaline catalyst such as sodium hydroxide creates a robust sulfonamide linkage. This protection is crucial because it stabilizes the nitrogen atom against protonation or oxidation during the harsh acidic conditions of the next step, ensuring that the electrophilic aromatic substitution occurs exclusively at the desired position on the benzene ring to form the six-membered lactam ring.
The cyclization mechanism itself is driven by polyphosphoric acid (PPA), which acts as both a solvent and a potent dehydrating agent to facilitate the intramolecular Friedel-Crafts acylation. The carbonyl carbon of the propionic acid side chain becomes highly electrophilic under these conditions, attacking the ortho-position of the aniline ring to close the heterocycle. Once the ring is formed, the sulfonamide protecting group is cleaved using sodium carbonate, regenerating the secondary amine within the tetrahydroquinoline structure. Finally, the free base is converted into its hydrochloride salt through reaction with hydrochloric acid in methanol, a step that not only stabilizes the molecule but also enhances its crystallinity and handling properties for storage and transport. This mechanistic clarity ensures that impurity profiles are well-understood and controllable, a key requirement for regulatory filings.

How to Synthesize Substituted Tetrahydroquinoline-4-one Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control, particularly during the exothermic sulfonylation and the high-temperature cyclization steps. The patent provides specific molar ratios, such as using 1.2 equivalents of acrylic acid relative to aniline and optimizing the PPA ratio to ensure complete conversion without excessive degradation. Operators should note that the intermediate compounds, specifically the sulfonamide derivative, can often be used directly in the next step without isolation, further streamlining the workflow and reducing material loss. The detailed standardized synthesis steps below outline the precise conditions for each transformation, ensuring reproducibility and consistency across different batch sizes.
- Conduct an addition reaction between substituted aniline and acrylic acid in toluene at 65°C to form the amino-propionic acid intermediate.
- Perform sulfonyl substitution using p-toluenesulfonyl chloride and sodium hydroxide to protect the amine group, yielding the sulfonamide derivative.
- Execute cyclization using polyphosphoric acid (PPA) followed by deprotection with sodium carbonate to form the tetrahydroquinoline-4-one core.
- Complete the process by reacting the free base with hydrochloric acid in methanol and ether to precipitate the final hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of adopting this patented methodology are profound, primarily driven by the elimination of expensive and time-consuming purification unit operations. By removing the dependency on column chromatography, the process drastically reduces solvent consumption and waste disposal costs, which are often the hidden drivers of high manufacturing expenses in fine chemical production. Furthermore, the use of commodity chemicals like toluene, acrylic acid, and p-toluenesulfonyl chloride ensures that raw material sourcing is stable and cost-effective, mitigating the risk of supply disruptions associated with exotic or specialized reagents. This stability allows for more accurate forecasting and inventory management, essential components of a resilient supply chain strategy in the volatile pharmaceutical market.
- Cost Reduction in Manufacturing: The most significant cost driver in traditional synthesis is the purification stage, which this method effectively neutralizes by designing a route that yields high-purity intermediates through crystallization and extraction alone. Eliminating the need for silica gel and large volumes of elution solvents translates directly into lower variable costs per kilogram of product. Additionally, the ability to telescope steps or minimize isolation points reduces labor hours and equipment occupancy time, enhancing the overall asset utilization of the manufacturing facility. These efficiencies compound to offer substantial cost savings without compromising the quality of the final API intermediate.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials means that the supply chain is less susceptible to the bottlenecks that often plague specialty chemical markets. Aniline derivatives and acrylic acid are produced on a massive global scale, ensuring consistent availability and competitive pricing. Moreover, the robustness of the reaction conditions, which tolerate a range of temperatures and concentrations, reduces the likelihood of batch failures due to minor process deviations. This reliability ensures that delivery schedules can be met consistently, reducing lead time for high-purity pharmaceutical intermediates and strengthening the partnership between suppliers and their downstream clients.
- Scalability and Environmental Compliance: From an environmental perspective, the reduction in solvent usage and the avoidance of chromatographic waste streams align perfectly with modern green chemistry principles and regulatory expectations. The process generates less hazardous waste, simplifying the permitting process for manufacturing sites and reducing the environmental footprint of production. Scalability is inherently built into the design, as the exotherms are manageable and the workup procedures involve standard unit operations like filtration and phase separation that are easily replicated from pilot plant to multi-ton commercial reactors. This makes the technology ideal for the commercial scale-up of complex polymer additives or pharmaceutical intermediates where volume and consistency are key.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and experimental data. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this route into their existing production lines. The answers provided reflect the specific advantages and operational parameters defined in the intellectual property, ensuring accuracy and relevance for decision-makers.
Q: Why is the tosyl protection step critical in this synthesis route?
A: The tosyl protection step prevents side reactions during the cyclization phase and significantly improves product purity, eliminating the need for complex column chromatography purification which is required in conventional direct cyclization methods.
Q: What are the primary advantages of this method for large-scale manufacturing?
A: This method utilizes cheap and readily available raw materials, operates under moderate reaction conditions, and features simple post-treatment processes such as filtration and extraction, making it highly suitable for industrial amplification.
Q: Can this synthesis accommodate various substituents on the aniline ring?
A: Yes, the process is robust and adaptable for anilines substituted with hydrogen, chlorine, bromine, methyl, ethyl, or methoxy groups at the 5, 6, 7, or 8 positions, allowing for the production of a diverse range of derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydroquinoline-4-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN102382050A for securing a competitive edge in the global marketplace. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless and efficient. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of tetrahydroquinoline derivative meets the exacting standards required by top-tier pharmaceutical and agrochemical companies worldwide.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this protected cyclization strategy. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive innovation and profitability in your supply chain.
