Advanced Catalytic Synthesis of Tamibarotene for Commercial Scale API Manufacturing
The pharmaceutical industry constantly seeks more efficient pathways for synthesizing critical oncology intermediates, and patent CN103408450A presents a groundbreaking advancement in the production of Tamibarotene, a potent Retinoic Acid Receptor Alpha (RARα) agonist used for treating acute promyelocytic leukemia. This patent discloses a novel method utilizing a highly active acenaphthoimidazole N-heterocyclic carbene allyl palladium chloride compound as a catalyst to drive an aminocarbonylation coupling reaction. Unlike traditional multi-step syntheses that rely on harsh Friedel-Crafts conditions and expensive acyl chlorides, this innovative approach enables the direct one-step construction of the key amide bond using carbon monoxide as a carbonyl source. For a reliable pharmaceutical intermediates supplier, adopting this technology translates to a drastic simplification of the supply chain, offering a robust alternative for cost reduction in API manufacturing while maintaining the high purity standards demanded by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Tamibarotene has been plagued by inefficient routes that involve four to five distinct reaction steps, often necessitating complex protection and deprotection strategies that severely impact atom economy. As illustrated in prior art such as Route One, the process typically begins with 2,5-dimethyl-2,5-hexanediol, undergoing chlorination and Friedel-Crafts cyclization to form the tetramethylnaphthalene core, followed by nitration, reduction, and finally acylation.
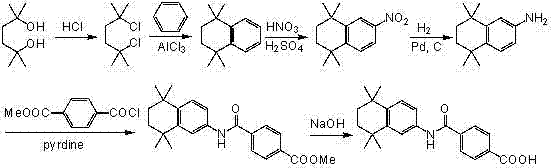
These conventional pathways frequently require the use of hazardous reagents like phosphorus pentachloride and concentrated hydrochloric acid, generating significant amounts of acidic waste and byproducts that complicate environmental compliance. Furthermore, the reliance on difficult-to-synthesize starting materials, such as methyl 4-chlorocarbonylbenzoate, not only inflates raw material costs but also introduces supply chain vulnerabilities due to the limited availability of these specialized precursors. The cumulative effect of these inefficiencies is a process that is operationally complex, environmentally burdensome, and economically suboptimal for large-scale commercial production.
The Novel Approach
In stark contrast, the methodology described in CN103408450A revolutionizes the synthesis by condensing the entire process into just two high-yielding steps, bypassing the need for tedious functional group manipulations. The core innovation lies in the direct aminocarbonylation of 5,6,7,8-tetrahydro-5,5,8,8-tetramethyl-2-naphthylamine with ethyl 4-iodobenzoate under a carbon monoxide atmosphere, catalyzed by the novel palladium complex. This strategy effectively replaces the problematic acyl chloride coupling with a cleaner, more atom-economical carbonylation reaction that utilizes inexpensive and readily available aryl iodides. By eliminating the need for protection groups and avoiding the generation of stoichiometric amounts of inorganic salts associated with traditional coupling agents, this novel approach offers a streamlined pathway that is inherently safer and more sustainable for industrial application.
Mechanistic Insights into Pd-NHC Catalyzed Aminocarbonylation
The success of this synthetic route hinges on the exceptional performance of the acenaphthoimidazole N-heterocyclic carbene (NHC) allyl palladium chloride catalyst, which facilitates the oxidative addition of the aryl iodide and the subsequent insertion of carbon monoxide with remarkable efficiency. The bulky acenaphthene backbone of the NHC ligand provides substantial steric protection around the palladium center, stabilizing the active catalytic species and preventing the formation of inactive palladium black aggregates that often plague carbonylation reactions. This structural integrity allows the catalyst to operate effectively at relatively low loadings, typically between 0.5 mol% and 1.0 mol%, while maintaining high turnover numbers throughout the reaction cycle.
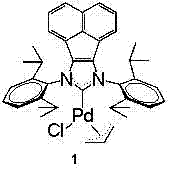
From an impurity control perspective, the high selectivity of this catalytic system is paramount for producing high-purity pharmaceutical intermediates suitable for oncology applications. The mechanism proceeds through a well-defined cycle of oxidative addition, CO migratory insertion, and nucleophilic attack by the amine, followed by reductive elimination to release the amide product. This precise control minimizes side reactions such as homocoupling of the aryl iodide or incomplete carbonylation, which are common pitfalls in less optimized systems. Consequently, the crude reaction mixture contains significantly fewer organic impurities, reducing the burden on downstream purification processes and ensuring that the final API meets stringent quality specifications without extensive recrystallization or chromatography.
How to Synthesize Tamibarotene Efficiently
The implementation of this catalytic route involves a straightforward two-step sequence that begins with the preparation of the specialized palladium catalyst followed by the key coupling and hydrolysis steps. The process is designed to be operationally simple, utilizing standard laboratory equipment and avoiding extreme conditions such as cryogenic temperatures or high-pressure reactors, which facilitates easy technology transfer to manufacturing sites. Detailed standardized synthesis steps for the preparation of the catalyst and the subsequent conversion to Tamibarotene are provided in the guide below.
- Prepare the novel acenaphthoimidazole N-heterocyclic carbene allyl palladium chloride catalyst by reacting the benzimidazole salt with allyl palladium chloride dimer and potassium tert-butoxide in THF.
- Perform the key aminocarbonylation coupling reaction between 5,6,7,8-tetrahydro-5,5,8,8-tetramethyl-2-naphthylamine and ethyl 4-iodobenzoate under CO atmosphere using the prepared catalyst.
- Hydrolyze the resulting ethyl ester intermediate using sodium hydroxide in methanol/water to obtain the final Tamibarotene acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers compelling strategic benefits that extend beyond mere technical feasibility. By fundamentally altering the chemical logic of the synthesis, the process removes several historical bottlenecks associated with raw material sourcing and waste management, thereby enhancing the overall resilience of the supply chain. The shift away from proprietary or difficult-to-source acyl chlorides towards commodity chemicals like ethyl 4-iodobenzoate ensures a more stable and competitive pricing structure for the final intermediate.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents such as phosphorus pentachloride and methyl 4-chlorocarbonylbenzoate directly lowers the bill of materials, while the reduced number of synthetic steps decreases labor, energy, and solvent consumption significantly. Furthermore, the high catalytic efficiency means that precious metal usage is minimized, and the avoidance of complex protection/deprotection sequences reduces the total volume of waste generated, leading to substantial cost savings in waste disposal and environmental compliance.
- Enhanced Supply Chain Reliability: Sourcing risks are mitigated by relying on widely available starting materials like 5,6,7,8-tetrahydro-5,5,8,8-tetramethyl-2-naphthylamine and ethyl 4-iodobenzoate, which are produced by multiple global suppliers. This diversification of the supply base prevents single-source dependencies and ensures consistent availability of critical inputs, thereby reducing lead times for high-purity pharmaceutical intermediates and safeguarding against market volatility.
- Scalability and Environmental Compliance: The process is inherently scalable due to its simplicity and the use of robust reaction conditions that do not require specialized high-pressure equipment for carbon monoxide insertion. The replacement of corrosive acids and chlorinating agents with a cleaner catalytic cycle aligns with modern green chemistry principles, simplifying the permitting process for new manufacturing lines and reducing the environmental footprint of the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel catalytic technology for Tamibarotene production. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the operational advantages.
Q: What are the primary advantages of this Pd-NHC catalyzed route over traditional Friedel-Crafts methods?
A: The novel route significantly shortens the synthesis to just two steps, eliminates the need for hazardous reagents like phosphorus pentachloride and concentrated hydrochloric acid, and utilizes cheaper, more accessible starting materials like ethyl 4-iodobenzoate instead of difficult-to-synthesize chloroformates.
Q: How does the new catalyst improve impurity profiles for API manufacturing?
A: The high catalytic activity and selectivity of the acenaphthoimidazole NHC-Pd complex minimize side reactions and byproduct formation, resulting in a cleaner crude product that simplifies downstream purification and ensures stringent purity specifications required for oncology drugs.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the process avoids complex protection/deprotection strategies and low-temperature operations, utilizing standard atmospheric pressure CO insertion and simple hydrolysis, which makes it highly scalable and economically viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tamibarotene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Pd-NHC catalyzed route for the commercial production of Tamibarotene and other complex oncology intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of materials that meet stringent purity specifications. Our state-of-the-art facilities are equipped with rigorous QC labs capable of monitoring trace impurities and validating the efficacy of the catalytic process at every stage of development.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this innovative method can optimize your supply chain and accelerate your time to market.
