Scalable Copper-Catalyzed Synthesis of Thioindoline Derivatives for High-Purity Pharmaceutical Applications
Scalable Copper-Catalyzed Synthesis of Thioindoline Derivatives for High-Purity Pharmaceutical Applications
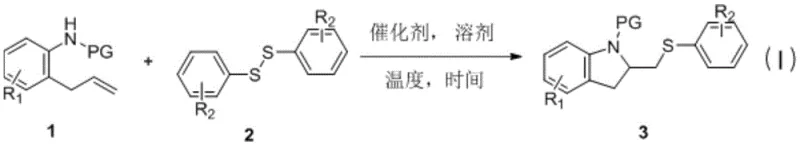
The landscape of organic synthesis for heterocyclic compounds is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally benign processes. A significant breakthrough in this domain is documented in patent CN108484472B, which details a novel synthetic method for thioindoline compounds. These structures serve as critical scaffolds in the development of bioactive molecules, including various natural products and pharmaceutical agents with potent medicinal value. The patent introduces a streamlined one-pot methodology that utilizes 2-allyl phenyl aniline derivatives and disulfides as primary raw materials, mediated by a cupric salt catalyst. This innovation addresses long-standing challenges in the field, offering a robust alternative to legacy methods that often suffer from苛刻 reaction conditions and poor atom economy. For global procurement teams and R&D directors seeking reliable sources of complex intermediates, this technology represents a pivotal shift towards more sustainable and scalable manufacturing paradigms.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the advancements detailed in the referenced patent, the synthesis of thioindoline derivatives was fraught with significant technical and operational hurdles. The existing state-of-the-art primarily relied on iodine-catalyzed reactions involving allyl phenyl aniline derivatives and sodium mercaptide. This conventional approach was not only limited in its selectivity and universality but also imposed severe constraints on process engineering. Most critically, the reaction necessitated cryogenic conditions, typically requiring temperatures as low as -20°C to proceed with any degree of control. Such extreme thermal requirements translate directly into substantial energy consumption and increased capital expenditure for specialized cooling infrastructure. Furthermore, the yields obtained through these traditional pathways were often suboptimal, leading to inefficient material usage and complicating downstream purification processes. The reliance on specific mercaptide reagents also introduced handling complexities due to their odor and stability issues, further detracting from the overall viability of the process for large-scale industrial application.
The Novel Approach
In stark contrast to the limitations of prior art, the method disclosed in patent CN108484472B offers a transformative solution by leveraging a copper-catalyzed radical cyclization strategy. This novel approach eliminates the need for cryogenic conditions, operating efficiently at elevated temperatures around 100°C, which significantly simplifies reactor design and energy management. By utilizing readily available disulfides instead of unstable mercaptides, the process enhances safety and ease of handling while expanding the scope of accessible chemical space. The use of common cupric salts, such as copper acetate, as catalysts provides a cost-effective alternative to precious metal systems or stoichiometric oxidants. This methodology enables the direct construction of the thioindoline core through a tandem addition-cyclization sequence, achieving high reaction efficiency in a single pot. The versatility of this system allows for the accommodation of various substituents on both the aniline and disulfide components, thereby facilitating the rapid generation of diverse libraries of thioindoline derivatives for drug discovery and material science applications.
Mechanistic Insights into Copper-Catalyzed Radical Cyclization
Understanding the underlying mechanistic pathway is crucial for R&D directors aiming to optimize reaction parameters and ensure consistent product quality. The proposed mechanism for this transformation involves a sophisticated interplay of radical and ionic species initiated by the copper catalyst. Initially, the disulfide bond undergoes homolytic cleavage or activation to generate a sulfur-centered free radical. This highly reactive species then adds across the terminal double bond of the 2-allyl phenyl aniline derivative, forming a new carbon-sulfur bond and generating a carbon-centered radical intermediate at the benzylic position. Subsequently, this carbon radical undergoes a single electron transfer (SET) process, likely mediated by the copper species, to generate a transient carbocation. This electrophilic center is then intercepted by the neighboring nitrogen atom, which acts as an internal nucleophile, triggering an intramolecular cyclization event that closes the five-membered indoline ring. The final deprotonation step restores aromaticity or neutrality, yielding the stable thioindoline product. This elegant cascade demonstrates the power of transition metal catalysis in orchestrating complex bond-forming events with high precision.
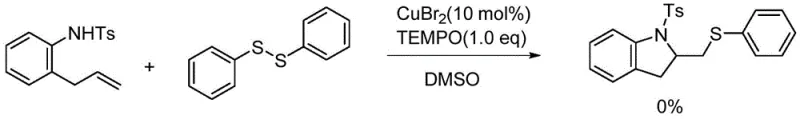
To validate this radical hypothesis, the inventors conducted control experiments using classical radical trapping agents. Specifically, the addition of TEMPO (2,2,6,6-tetramethylpiperidin-1-yl)oxyl to the reaction mixture resulted in the complete suppression of product formation. This observation serves as definitive proof that free radical intermediates are essential for the reaction to proceed. The inhibition of the cyclization in the presence of TEMPO confirms that the pathway does not operate via a purely ionic or concerted mechanism but relies heavily on the generation and propagation of radical species. This mechanistic understanding is vital for troubleshooting potential side reactions and optimizing the catalyst loading to balance radical generation with productive turnover. It also highlights the importance of excluding oxygen or other radical scavengers from the reaction environment to maintain high yields and reproducibility during scale-up operations.
How to Synthesize 2-[(Phenylthio)methyl]-1-tosylindoline Efficiently
The practical implementation of this synthetic route is designed for simplicity and robustness, making it highly attractive for process chemists looking to establish reliable supply chains. The standard protocol involves charging a reaction vessel with the 2-allyl phenyl aniline derivative and the chosen disulfide in a polar aprotic solvent such as DMF. A catalytic amount of copper acetate is then introduced to initiate the cycle. The mixture is heated to reflux conditions, typically around 100°C, and maintained for a period sufficient to drive the conversion to completion, usually approximately 24 hours. Workup procedures are straightforward, involving aqueous quenching and extraction, followed by standard purification techniques like column chromatography. This operational simplicity reduces the barrier to entry for manufacturing and minimizes the risk of operator error. For detailed procedural specifics regarding reagent grades, exact stoichiometry, and safety protocols, please refer to the standardized synthesis guide below.
- Combine 2-allyl phenyl aniline derivatives and disulfides in an organic solvent such as DMF.
- Add a cupric salt catalyst, preferably copper acetate, at a molar ratio of approximately 10%.
- Heat the reaction mixture to 100°C for 24 hours to facilitate radical cyclization and product formation.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this copper-catalyzed methodology offers profound benefits for procurement managers and supply chain heads focused on cost optimization and reliability. The shift away from cryogenic processing and expensive or hazardous reagents translates directly into reduced operational expenditures. By utilizing commodity chemicals like disulfides and copper salts, manufacturers can mitigate the risks associated with volatile raw material markets and ensure a steady supply of inputs. The robustness of the reaction conditions also implies a lower rate of batch failures, enhancing overall supply continuity. Furthermore, the environmental profile of the process is improved by avoiding the use of stoichiometric iodine and reducing energy consumption associated with cooling, aligning with increasingly stringent global sustainability mandates. These factors collectively contribute to a more resilient and cost-efficient supply chain for high-value thioindoline intermediates.
- Cost Reduction in Manufacturing: The elimination of cryogenic equipment and the use of inexpensive copper catalysts significantly lower the capital and operational costs associated with production. Unlike previous methods that required specialized low-temperature reactors and energy-intensive cooling systems, this process operates at standard heating conditions, drastically simplifying the infrastructure requirements. Additionally, the high atom economy and improved yields reduce the cost per kilogram of the final product by minimizing waste and maximizing raw material utilization. The avoidance of costly purification steps often necessitated by side products in older methods further contributes to substantial economic savings, making the final API or intermediate more competitive in the global market.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available disulfides and common copper salts ensures a secure and predictable supply of raw materials. Sodium mercaptides used in traditional methods can be unstable and difficult to source in bulk quantities, creating potential bottlenecks. In contrast, disulfides are shelf-stable industrial commodities with well-established supply networks. This stability reduces the risk of production delays caused by raw material shortages or degradation during storage. Moreover, the tolerance of the reaction to various functional groups allows for flexibility in sourcing different substituted starting materials, providing procurement teams with multiple options to hedge against supply disruptions for specific precursors.
- Scalability and Environmental Compliance: The one-pot nature of the reaction and the absence of sensitive reagents make this process inherently scalable from laboratory benchtop to multi-ton industrial reactors. The simplicity of the workup procedure facilitates easier handling of large volumes without complex separation technologies. From an environmental standpoint, the use of copper, a less toxic metal compared to heavy alternatives, and the reduction in energy consumption align with green chemistry principles. This compliance with environmental standards reduces the regulatory burden and waste disposal costs, ensuring long-term viability of the manufacturing process in regions with strict ecological regulations. The ability to scale without compromising safety or efficiency is a key driver for meeting the growing demand for thioindoline-based therapeutics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of thioindoline compounds using this patented technology. These insights are derived directly from the experimental data and mechanistic studies presented in the patent documentation, providing clarity on the process capabilities and limitations. Understanding these aspects is essential for stakeholders evaluating the feasibility of integrating this technology into their existing production portfolios or sourcing strategies. The answers reflect the consensus of the technical team based on the rigorous validation performed during the patent filing process.
Q: What are the advantages of this copper-catalyzed method over traditional iodine catalysis?
A: Unlike traditional methods requiring harsh cryogenic conditions (-20°C) and iodine catalysts with limited selectivity, this novel approach operates at mild temperatures (100°C) using cheap copper salts, resulting in higher yields and broader substrate compatibility.
Q: Is the reaction mechanism radical-based?
A: Yes, mechanistic studies using TEMPO as a radical trapping agent confirmed that the reaction proceeds via a sulfur free radical addition pathway, followed by single electron transfer to form a carbocation intermediate.
Q: Is this process suitable for large-scale industrial production?
A: Absolutely. The process utilizes common industrial solvents like DMF, stable disulfide raw materials, and requires no special equipment, making it highly adaptable for commercial scale-up from kilogram to tonnage levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thioindoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced synthetic methodologies like the one described in patent CN108484472B for driving innovation in the pharmaceutical and agrochemical sectors. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering high-purity thioindoline intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to adapt and optimize such copper-catalyzed processes allows us to offer cost-effective solutions without compromising on quality or delivery timelines, positioning us as a preferred partner for complex molecule synthesis.
We invite global partners to collaborate with us to leverage this cutting-edge technology for your specific project needs. Whether you require custom synthesis services or bulk supply of thioindoline derivatives, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out to us to request specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance your supply chain efficiency and product competitiveness. Let us help you navigate the complexities of fine chemical manufacturing with confidence and precision.
