Advancing Pharmaceutical Intermediate Synthesis via Mild Nickel Catalysis for Commercial Scale-Up
Introduction to Advanced Nickel-Catalyzed Synthetic Methodologies
The landscape of organic synthesis for complex heterocyclic scaffolds is undergoing a significant transformation, driven by the urgent need for greener, more efficient, and scalable methodologies. A pivotal development in this domain is documented in Chinese Patent CN110467556B, which discloses a novel nucleophilic reaction method utilizing nickel catalysis to couple imine ions with acetophenones. This technology represents a substantial leap forward for the production of 3-substituted isoindolinones, a privileged structural motif frequently encountered in bioactive alkaloids and pharmaceutical candidates. By shifting away from traditional, harsh acidic conditions to a mild divalent nickel catalytic system, this invention addresses critical pain points regarding reaction severity, operational complexity, and overall yield efficiency. For R&D directors and process chemists seeking robust routes for high-purity pharmaceutical intermediates, this patent offers a compelling alternative that balances mechanistic elegance with practical manufacturability.
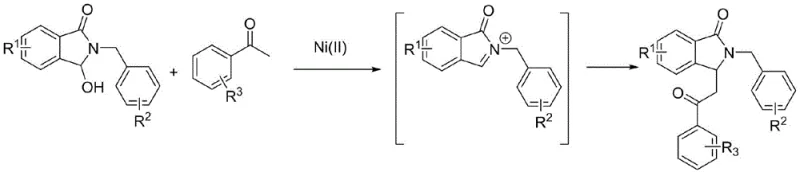
The core innovation lies in the ability of Ni(II) compounds to facilitate the in-situ generation of reactive iminium cations from 3-hydroxy-2-phenethyl-isoindolin-1-one precursors. Unlike previous iterations that relied on strong protonic acids which often led to substrate decomposition or required cryogenic controls, this nickel-mediated approach operates effectively at moderate temperatures ranging from 80°C to 120°C. The versatility of the system is further underscored by its compatibility with a wide array of organic solvents, including 1,2-dichloroethane, DMF, and toluene, allowing process engineers to optimize solubility and heat transfer parameters specifically for their reactor configurations. This flexibility is paramount when transitioning from bench-scale discovery to pilot plant operations, ensuring that the chemistry remains robust regardless of the specific equipment constraints.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbon-carbon bonds adjacent to nitrogen atoms in isoindolinone systems has been fraught with challenges, primarily due to the reliance on aggressive catalytic systems. Early methodologies, such as those pioneered by Professor Tsuda for Erythrina alkaloid synthesis, necessitated the use of various strong protonic acid catalysts to drive the intramolecular nucleophilic reaction of N-imine ions. While effective in specific contexts, these acidic conditions are inherently corrosive, posing significant risks to reactor integrity and requiring specialized materials of construction that drive up capital expenditure. Furthermore, the harsh acidic environment often compromises the stability of sensitive functional groups, leading to side reactions, polymerization, or degradation of the starting materials, which ultimately depresses the overall yield and complicates the downstream purification process.
Additionally, traditional acid-catalyzed pathways frequently suffer from poor atom economy and difficult workup procedures. The neutralization of large quantities of strong acid generates substantial amounts of salt waste, creating an environmental burden that conflicts with modern green chemistry principles. For procurement managers focused on cost reduction in API manufacturing, the disposal costs associated with acidic effluent and the loss of valuable starting material due to decomposition are non-trivial factors. Moreover, the operational window for these reactions is often narrow, requiring precise temperature control and anhydrous conditions that are difficult to maintain consistently in large-scale batch reactors, thereby introducing variability in product quality and batch-to-batch reproducibility.
The Novel Approach
In stark contrast, the nickel-catalyzed methodology described in the patent data introduces a paradigm shift towards milder, more controllable reaction conditions. By employing divalent nickel salts such as nickel perchlorate, nickel chloride, or nickel bromide, the reaction proceeds smoothly at temperatures between 80°C and 120°C, eliminating the need for corrosive strong acids. This transition not only enhances the safety profile of the operation but also significantly broadens the scope of compatible substrates. The mild Lewis acidic nature of the Ni(II) center is sufficient to activate the hydroxyl leaving group and generate the electrophilic iminium species without inducing the widespread degradation observed in Brønsted acid catalysis. Consequently, reported yields for this novel approach consistently reach impressive levels, typically ranging from 85% to 95%, demonstrating superior efficiency over legacy methods.
From a process engineering perspective, the simplicity of the novel approach cannot be overstated. The reaction mixture can be processed using standard extraction techniques followed by silica gel column chromatography, avoiding the complex neutralization and filtration steps required for acid removal. This streamlined workflow reduces the total processing time and minimizes the exposure of personnel to hazardous chemicals. For supply chain heads concerned with reducing lead time for high-purity pharmaceutical intermediates, the ability to achieve high conversion rates with a simple workup translates directly into faster throughput and more reliable delivery schedules. The robustness of the nickel catalyst across different solvent systems further ensures that the process can be adapted to existing manufacturing infrastructure without requiring extensive retrofitting or new equipment procurement.
Mechanistic Insights into Ni(II)-Catalyzed Iminium Ion Generation
The mechanistic underpinning of this transformation involves a sophisticated interplay between the nickel catalyst and the substrate to generate a highly reactive electrophile under mild conditions. The process initiates with the coordination of the Ni(II) species to the hydroxyl group of the 3-hydroxy-2-phenethyl-isoindolin-1-one substrate. This coordination enhances the leaving group ability of the hydroxyl moiety, facilitating its departure and the subsequent formation of a stabilized iminium cation intermediate. This in-situ generation of the iminium ion is the critical rate-determining step, and the nickel catalyst plays a pivotal role in lowering the activation energy barrier for this elimination process. Unlike protonation, which relies on the concentration of free protons in solution, the metal-mediated pathway offers a more controlled and localized activation, preventing the bulk solution from becoming overly acidic and thus preserving the integrity of acid-sensitive functionalities on the aromatic rings.
Once the iminium cation is generated, it acts as a potent electrophile, susceptible to nucleophilic attack by the enol or enolate form of the acetophenone derivative. The nickel center may also assist in organizing the transition state, potentially coordinating with the carbonyl oxygen of the acetophenone to increase its nucleophilicity or to orient the reactants for optimal orbital overlap. This dual activation mode ensures high regioselectivity and stereoselectivity in the formation of the new carbon-carbon bond at the C3 position of the isoindolinone ring. The resulting adduct then undergoes proton transfer and tautomerization to yield the final 3-substituted isoindolinone product. Understanding this mechanism is crucial for R&D teams aiming to further optimize the reaction, as it highlights the importance of catalyst loading and the electronic nature of the substituents on both the isoindolinone and acetophenone partners.
Furthermore, the tolerance of the catalytic system towards various substituents (R1, R2, R3) such as halogens, nitro groups, and alkoxy groups suggests that the electronic properties of the aromatic rings do not drastically inhibit the catalytic cycle. Electron-withdrawing groups on the acetophenone, for instance, might typically reduce nucleophilicity, yet the reaction proceeds efficiently, indicating the high electrophilicity of the nickel-generated iminium species. This mechanistic resilience allows for the synthesis of a diverse library of analogues, which is invaluable for medicinal chemistry campaigns exploring structure-activity relationships. The ability to predictably manipulate the electronic environment of the reactants without compromising the catalytic turnover provides a powerful tool for designing complex molecular architectures with precision.
How to Synthesize 3-Substituted Isoindolinones Efficiently
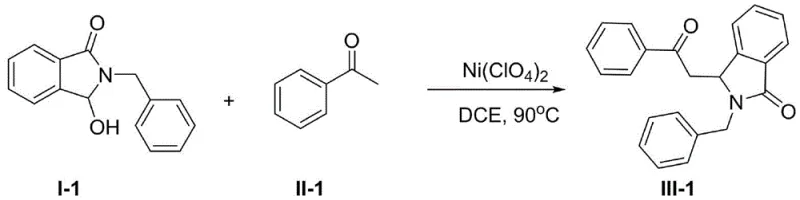
Implementing this nickel-catalyzed protocol in a laboratory or pilot setting requires careful attention to reagent stoichiometry and thermal management to maximize yield and purity. The general procedure involves dissolving the 3-hydroxy-2-phenethyl-isoindolin-1-one and the acetophenone derivative in a dry organic solvent, followed by the addition of the nickel catalyst. The mixture is then heated to the specified temperature range and monitored until completion. The beauty of this method lies in its operational simplicity; it does not require inert atmosphere gloveboxes for setup, although maintaining anhydrous conditions is beneficial for consistent results. The following guide outlines the standardized steps derived from the patent examples to ensure reproducible outcomes.
- Dissolve 3-hydroxy-2-phenethyl-isoindolin-1-one and acetophenone derivatives in an organic solvent such as 1,2-dichloroethane or DMF.
- Add a Ni(II) compound catalyst, such as Ni(ClO4)2 or NiCl2, and stir the mixture at temperatures between 80°C and 120°C for 4 to 8 hours.
- Perform aqueous workup by extracting with dichloromethane, washing with brine, drying over sodium sulfate, and purifying the residue via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this nickel-catalyzed technology offers tangible benefits that extend beyond mere chemical yield. The shift from expensive or hazardous acid catalysts to readily available nickel salts represents a direct opportunity for cost reduction in pharmaceutical intermediate manufacturing. Nickel compounds such as nickel chloride or nickel nitrate are commodity chemicals with stable pricing and abundant global supply, mitigating the risk of raw material shortages that often plague specialized organocatalysts or precious metal complexes. Furthermore, the mild reaction conditions translate to lower energy consumption, as the process does not require extreme heating or cryogenic cooling, thereby reducing the utility costs associated with large-scale production runs.
- Cost Reduction in Manufacturing: The elimination of strong acid catalysts removes the need for costly corrosion-resistant reactor linings and extensive neutralization waste treatment protocols. By utilizing base metal catalysis instead of precious metals like palladium or rhodium, the direct material cost per kilogram of product is significantly lowered. Additionally, the high yields reported (85-95%) minimize the loss of valuable starting materials, ensuring that the input costs are efficiently converted into saleable product. This economic efficiency is compounded by the simplified purification process, which reduces the volume of solvents and silica gel required for chromatography, further driving down the variable costs of production.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures high batch-to-batch consistency, a critical factor for maintaining reliable supply lines to downstream API manufacturers. The use of common solvents like 1,2-dichloroethane, DMF, or toluene means that solvent sourcing is not a bottleneck, as these are standard inventory items for most chemical facilities. Moreover, the stability of the nickel catalyst allows for flexible scheduling; the reaction is not overly sensitive to minor fluctuations in temperature or mixing rates, reducing the likelihood of batch failures that could disrupt delivery timelines. This reliability fosters stronger partnerships with clients who depend on just-in-time delivery models for their own production schedules.
- Scalability and Environmental Compliance: Scaling this process from grams to tons is facilitated by the homogeneous nature of the catalytic system and the absence of gas evolution or exothermic spikes that are difficult to manage in large vessels. The reduced generation of acidic waste aligns with increasingly stringent environmental regulations, simplifying the permitting process for new production lines. The ability to recycle solvents and the potential for catalyst recovery further enhance the sustainability profile of the manufacturing process. For companies aiming to meet corporate sustainability goals, adopting this greener synthetic route demonstrates a commitment to environmental stewardship while maintaining commercial viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed nucleophilic reaction method. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity for technical teams evaluating this technology for integration into their existing workflows. Understanding these nuances is essential for making informed decisions about process adoption and resource allocation.
Q: What are the primary advantages of using Ni(II) catalysts over traditional protonic acids for isoindolinone synthesis?
A: The Ni(II) catalytic system operates under significantly milder conditions (80-120°C) compared to the harsh acidic environments required by traditional methods, resulting in higher yields (85-95%) and simplified purification processes without the need for extreme pH control.
Q: Is this nucleophilic reaction method compatible with diverse functional groups on the acetophenone substrate?
A: Yes, the method demonstrates excellent functional group tolerance, successfully accommodating electron-withdrawing groups like nitro and halogens, as well as electron-donating groups like methoxy, making it highly versatile for generating diverse pharmaceutical libraries.
Q: How does this process impact the scalability of producing 3-substituted isoindolinones?
A: The use of commercially available nickel salts and common organic solvents, combined with a straightforward workup procedure involving standard extraction and column chromatography, facilitates easy scale-up from laboratory grams to multi-kilogram commercial production runs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Substituted Isoindolinones Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the nickel-mediated synthesis described in CN110467556B. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate such innovative patent technologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready supply is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this cost-effective and scalable synthetic route for your specific pharmaceutical projects. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this nickel-catalyzed method can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data for our isoindolinone intermediates and to discuss detailed route feasibility assessments for your target molecules. Let us be your strategic partner in navigating the complexities of modern chemical manufacturing.
