Scalable Kinetic Resolution Technology for High-Purity Aromatic Chiral Secondary Alcohols
Scalable Kinetic Resolution Technology for High-Purity Aromatic Chiral Secondary Alcohols
The pharmaceutical and fine chemical industries are constantly seeking robust, cost-effective methodologies for the synthesis of chiral building blocks, particularly aromatic chiral secondary alcohols which serve as critical intermediates in drug discovery and development. A significant breakthrough in this domain is documented in patent CN101314559B, which outlines a novel preparation method that circumvents the limitations of traditional asymmetric synthesis. This technology leverages a kinetic resolution strategy utilizing inexpensive chiral amino acid derivatives, specifically tert-butoxycarbonyl-protected proline (Boc-Pro), coupled with efficient activation agents. By shifting away from precious metal catalysts towards organocatalytic esterification, this process offers a compelling value proposition for manufacturers aiming to reduce production costs while maintaining rigorous purity standards required for regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active aromatic secondary alcohols has relied heavily on three primary strategies, each carrying significant operational and economic burdens for large-scale manufacturing. The first approach involves the use of chiral ligands to modify metal hydride reagents, such as BINAL-H, which requires complex ligand synthesis and strictly anhydrous conditions that are difficult to maintain industrially. The second method employs transition metal complexes, typically based on ruthenium or rhodium, for asymmetric hydrogenation; while effective, these metals are prohibitively expensive, subject to volatile market pricing, and pose severe challenges regarding residual metal removal in final API products. The third conventional route utilizes boron-oxazolidine catalyst systems (CBS reduction), which, although highly selective, often necessitates cryogenic temperatures and specialized handling of borane reagents. Collectively, these traditional methods suffer from high raw material costs, complex waste streams containing heavy metals, and operational inflexibility that hinders rapid scale-up.
The Novel Approach
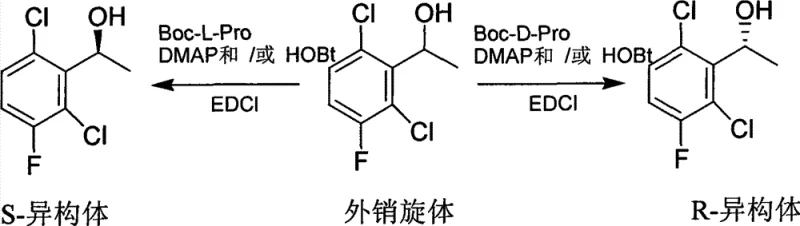
In stark contrast, the methodology described in CN101314559B introduces a streamlined kinetic resolution pathway that transforms these economic and technical challenges into opportunities for efficiency. This novel approach exploits the differential reaction rates of enantiomers when exposed to a chiral acylating agent. By reacting a racemic mixture of aromatic secondary alcohols with Boc-L-Proline or Boc-D-Proline in the presence of EDCI (1-ethyl-3-(3-dimethylaminopropyl)carbodiimide), the process selectively esterifies one enantiomer while leaving the other largely untouched. This allows for the isolation of the desired unreacted alcohol with exceptionally high optical purity. The reaction proceeds under mild thermal conditions ranging from 0°C to 40°C in common aprotic solvents like dichloromethane or THF, eliminating the need for cryogenic cooling or high-pressure hydrogenation equipment. Furthermore, the reliance on amino acid derivatives ensures that the reagent cost is a fraction of that associated with noble metal catalysts.
Mechanistic Insights into Boc-Proline Mediated Kinetic Resolution
The core of this technological advancement lies in the precise stereochemical control exerted during the esterification process. The mechanism initiates with the activation of the carboxylic acid group of the Boc-protected proline by EDCI, forming an O-acylisourea intermediate. In the presence of nucleophilic catalysts like DMAP (4-dimethylaminopyridine) or HOBt (1-hydroxybenzotriazole), this intermediate is converted into a more stable and reactive active ester species. The chiral environment provided by the proline backbone creates a steric differentiation between the two enantiomers of the racemic alcohol substrate. One enantiomer fits sterically and electronically into the transition state much more favorably, leading to rapid acylation and formation of the corresponding ester. Conversely, the mismatched enantiomer reacts significantly slower, remaining in the solution as the free alcohol. This kinetic differentiation is the driving force that allows for the separation of enantiomers simply by isolating the unreacted alcohol fraction after the reaction reaches partial conversion.
Impurity control in this system is inherently robust due to the nature of the reagents and the reaction pathway. Unlike metal-catalyzed reductions which can generate over-reduced byproducts or isomerization impurities, this esterification is highly chemoselective for the hydroxyl group. The use of Boc-protecting groups on the proline prevents unwanted side reactions at the amine nitrogen, ensuring that the chiral auxiliary remains intact throughout the process. Additionally, the byproducts generated, primarily urea derivatives from the EDCI and the esterified alcohol, are generally polar and easily separable from the target non-polar aromatic alcohol via standard silica gel chromatography or recrystallization. The patent data indicates that this mechanistic precision consistently yields products with an enantiomeric excess (ee) exceeding 99%, demonstrating the high fidelity of the chiral induction even on a preparative scale.
How to Synthesize Aromatic Chiral Secondary Alcohol Efficiently
Implementing this kinetic resolution protocol requires careful attention to stoichiometry and reaction monitoring to maximize the yield of the desired enantiomer. The process is designed to be operationally simple, utilizing standard laboratory glassware and readily available solvents, making it highly transferable from pilot plant to full commercial production. The key to success lies in optimizing the molar ratio of the chiral resolving agent to the racemic substrate, typically maintaining the Boc-Proline at 0.5 to 0.7 equivalents to ensure theoretical maximum yield of the unreacted enantiomer while driving the resolution to completion. Detailed standardized synthetic steps for specific substrates, including exact reagent quantities and workup procedures, are outlined in the technical guide below.
- Dissolve the racemic aromatic secondary alcohol and the chiral resolving agent (Boc-L-Pro or Boc-D-Pro) in an aprotic organic solvent such as dichloromethane or THF at temperatures between 0°C and 40°C.
- Add the coupling agent EDCI along with catalytic amounts of DMAP and/or HOBt to activate the carboxylic acid for esterification.
- Monitor the reaction progress via chiral HPLC until completion, then purify the unreacted chiral alcohol using column chromatography or recrystallization to achieve high enantiomeric excess.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this kinetic resolution technology represents a strategic shift towards more resilient and cost-efficient manufacturing models. By replacing scarce and expensive transition metals with abundant amino acid derivatives, companies can decouple their production costs from the volatile commodities market associated with precious metals like Ruthenium and Rhodium. This substitution not only lowers the direct bill of materials but also simplifies the supply chain by reducing dependency on specialized catalyst suppliers who often have long lead times and strict allocation policies. Furthermore, the elimination of heavy metals removes the need for expensive and time-consuming metal scavenging steps downstream, streamlining the purification workflow and reducing overall processing time.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic reduction in reagent costs. Traditional asymmetric hydrogenation requires catalysts that can cost thousands of dollars per gram, whereas Boc-Proline is a commodity chemical available at a fraction of that price. Additionally, the reaction does not require specialized high-pressure reactors or cryogenic cooling systems, allowing facilities to utilize existing standard reactor infrastructure. The simplified workup procedure, which avoids complex metal removal protocols, further reduces labor and consumable costs associated with purification, leading to substantial overall cost savings per kilogram of produced intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is significantly improved because all key reagents, including EDCI, DMAP, and Boc-Proline, are produced on a massive global scale for the peptide synthesis industry. This ensures a stable, multi-vendor supply base that mitigates the risk of single-source bottlenecks. The robustness of the reaction conditions, which tolerate a range of temperatures from 0°C to 40°C, also means that production is less susceptible to disruptions caused by utility failures or equipment limitations. This flexibility allows manufacturing sites to maintain consistent output schedules even under varying operational constraints, ensuring reliable delivery to downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this organocatalytic route offers a greener alternative to metal-heavy processes. The absence of toxic heavy metals simplifies waste treatment and disposal, reducing the environmental footprint and regulatory burden associated with hazardous waste management. The solvents used, such as dichloromethane or ethyl acetate, are standard industrial solvents with well-established recovery and recycling protocols. The high atom economy of the resolution, combined with the ability to potentially recycle the esterified byproduct back to the racemate, supports sustainable manufacturing practices. This alignment with green chemistry principles facilitates easier regulatory approval and enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this kinetic resolution technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the process suitability for your specific project needs. Understanding these nuances is critical for R&D teams planning tech transfers and for procurement teams assessing vendor capabilities.
Q: Does this kinetic resolution method leave heavy metal residues?
A: No, unlike traditional asymmetric reductions using Ruthenium or Rhodium catalysts, this method utilizes organocatalysts (Boc-Proline) and carbodiimides, ensuring the final product is free from toxic heavy metal contaminants.
Q: What is the typical enantiomeric excess (ee) achieved with this process?
A: According to the patent embodiments, the process consistently achieves an enantiomeric excess (ee%) greater than 99% after purification, making it suitable for stringent pharmaceutical applications.
Q: Can this method be scaled for industrial production?
A: Yes, the method operates under mild conditions (0-40°C) using commercially available reagents and standard solvents, avoiding cryogenic temperatures or high-pressure equipment, which facilitates easy scale-up to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Chiral Secondary Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity chiral intermediates in the development of next-generation therapeutics. Our technical team has extensively analyzed the kinetic resolution pathways described in CN101314559B and possesses the expertise to optimize these reactions for diverse substrate scopes beyond the specific examples listed in the patent. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee enantiomeric excess levels that meet or exceed global pharmacopeial standards.
We invite potential partners to engage with our technical procurement team to discuss how this cost-effective technology can be integrated into your supply chain. By leveraging our manufacturing capabilities, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity targets. We encourage you to contact us directly to request specific COA data for our catalog of chiral alcohols or to initiate a dialogue regarding route feasibility assessments for your proprietary compounds. Let us collaborate to drive down costs and accelerate your time to market with reliable, high-quality chemical solutions.
