Advanced Chemical Synthesis of Chiral Norvaline for High-Volume Pharmaceutical Production
Advanced Chemical Synthesis of Chiral Norvaline for High-Volume Pharmaceutical Production
The pharmaceutical industry constantly seeks robust, scalable pathways for critical amino acid intermediates, particularly those serving as building blocks for major antihypertensive agents. Patent CN101007772B discloses a highly efficient synthetic methodology for chiral norvaline, specifically targeting the production of L-norvaline and D-norvaline with superior economic and operational metrics. This technology represents a significant departure from traditional biotechnological approaches, leveraging a modified Strecker synthesis coupled with classical chiral resolution to achieve high purity and yield. For R&D directors and procurement strategists, understanding this chemical trajectory is vital, as it offers a reliable alternative to fermentation processes that are often plagued by low volumetric productivity and complex downstream processing requirements. The method utilizes readily available commodity chemicals, fundamentally altering the cost structure and supply security for this key pharmaceutical intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of chiral norvaline has been hindered by significant technological bottlenecks that impact both cost and availability. Prior art, such as the fermentation method described in patent JP7553587, suffers from inherently low productivity, with reported yields around 3.7g/L, which is drastically inferior to standard industrial amino acid fermentation benchmarks. This low titer necessitates massive fermentation volumes to achieve meaningful output, driving up capital expenditure and energy consumption for separation and purification. Furthermore, alternative chemical routes, like the one disclosed in Chinese patent application 200410084417.7, rely on acetone cyanohydrin as a starting material. Acetone cyanohydrin is not only more expensive than simple inorganic cyanides but also presents handling and stability challenges that complicate logistics and storage. These conventional limitations create a fragile supply chain vulnerable to raw material price volatility and production inefficiencies.
The Novel Approach
The methodology outlined in CN101007772B circumvents these historical constraints by adopting a streamlined chemical synthesis starting from n-butyraldehyde, sodium cyanide, and ammonium chloride. This route eliminates the need for complex biological media or expensive organic cyanohydrins, replacing them with bulk commodities that ensure consistent pricing and availability. The process flow is designed for operational simplicity, moving directly from the Strecker reaction to amidation and finally to chiral resolution without requiring exotic catalysts or extreme conditions. By shifting the paradigm from biological fermentation to controlled chemical synthesis, manufacturers can achieve significantly shorter production cycles and higher space-time yields. This approach not only reduces the direct cost of goods sold but also simplifies the regulatory landscape by avoiding the variability inherent in biological systems, thereby offering a more predictable manufacturing profile for global supply chains.
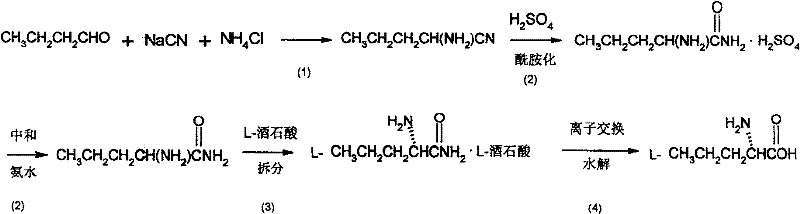
Mechanistic Insights into Strecker Synthesis and Chiral Resolution
The core of this innovative process lies in the precise execution of the Strecker reaction followed by a sophisticated resolution strategy. Initially, n-butyraldehyde undergoes nucleophilic addition with cyanide ions in the presence of ammonium chloride to form aminovaleronitrile. This reaction is conducted in a mixed solvent system of water and lower alcohols (methanol or ethanol) at temperatures between 50°C and 80°C. The choice of solvent ratio and temperature is critical to maximizing the conversion rate while minimizing side reactions such as aldol condensation of the aldehyde. Following the formation of the nitrile, the process employs a controlled acid hydrolysis using concentrated sulfuric acid. This step converts the nitrile group into an amide functionality, yielding racemic aminovaleramide. The use of sulfuric acid allows for a clean transformation that can be easily quenched and neutralized, setting the stage for the subsequent chiral separation without introducing difficult-to-remove impurities.
Chirality is established through the formation of diastereomeric salts using optically pure tartaric acid. When the racemic aminovaleramide interacts with L-tartaric acid (or D-tartaric acid), it forms two distinct salts: the L-aminopentanamide-L-tartrate and the D-aminopentanamide-L-tartrate. These diastereomers possess different physical properties, most notably solubility in methanol-water mixtures. By carefully controlling the crystallization conditions, including temperature (0°C to 15°C) and solvent ratios, the less soluble salt precipitates selectively, effectively enriching the desired enantiomer. The final step involves hydrolyzing the resolved amide salt using a cation exchange resin at elevated temperatures (100°C to 150°C). This resin-mediated hydrolysis is particularly advantageous as it facilitates the recovery of the valuable tartaric acid resolving agent, which can be recycled, further enhancing the economic viability and environmental profile of the overall process.
How to Synthesize Chiral Norvaline Efficiently
The synthesis of chiral norvaline via this patented route requires strict adherence to reaction parameters to ensure optimal optical purity and yield. The process integrates standard chemical engineering unit operations that are well-understood in fine chemical manufacturing, making it highly transferable to commercial scale. Operators must focus on the precise stoichiometry of the Strecker reactants and the careful control of pH during the neutralization phases to prevent racemization or degradation. The following guide outlines the critical stages of this synthesis, emphasizing the key control points that define the quality of the final API intermediate. For detailed standard operating procedures and specific batch records, please refer to the technical documentation provided below.
- Perform Strecker synthesis by reacting n-butyraldehyde with sodium cyanide and ammonium chloride in a water-alcohol solvent system at 50-80°C to form aminovaleronitrile.
- Hydrolyze the nitrile group using concentrated sulfuric acid followed by neutralization to obtain racemic aminovaleramide.
- Resolve the racemic mixture using L-tartaric or D-tartaric acid to form diastereomeric salts, followed by recrystallization and resin-catalyzed hydrolysis to yield the final chiral amino acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers profound strategic benefits that extend beyond simple unit cost reductions. The shift from fermentation or complex organic precursors to a commodity-based chemical synthesis fundamentally de-risks the supply chain. By relying on n-butyraldehyde and sodium cyanide, manufacturers tap into a global market of bulk chemicals with stable pricing and multiple sourcing options, insulating the production of chiral norvaline from the supply shocks often associated with specialized biochemical reagents. This raw material flexibility ensures business continuity and allows for more accurate long-term budgeting, which is crucial for managing the margins of high-volume pharmaceutical products like perindopril.
- Cost Reduction in Manufacturing: The elimination of expensive starting materials like acetone cyanohydrin and the avoidance of costly fermentation media significantly lowers the variable cost of production. Furthermore, the ability to recover and recycle the chiral resolving agent (tartaric acid) through the ion exchange process creates a closed-loop system that minimizes waste and reduces the consumption of high-value auxiliaries. This structural cost advantage allows suppliers to offer more competitive pricing without compromising on quality, providing a direct boost to the profitability of downstream drug manufacturing.
- Enhanced Supply Chain Reliability: Chemical synthesis offers a level of predictability that biological processes cannot match. Fermentation is susceptible to contamination, strain degeneration, and seasonal variations in raw material quality, all of which can lead to batch failures and delayed shipments. In contrast, this chemical route operates under defined physical parameters, ensuring consistent batch-to-batch performance. This reliability translates into shorter lead times and the ability to respond rapidly to fluctuations in market demand, securing the supply of this critical intermediate for global pharmaceutical clients.
- Scalability and Environmental Compliance: The process is designed for straightforward scale-up, utilizing standard reactors and separation equipment found in most multipurpose chemical plants. The simplified workflow reduces the number of processing steps, which in turn lowers energy consumption and solvent usage. Additionally, the efficient recovery of tartaric acid and the use of aqueous workups where possible contribute to a reduced environmental footprint. This alignment with green chemistry principles facilitates easier regulatory approval and supports the sustainability goals of modern pharmaceutical supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of chiral norvaline using this specific patented methodology. These insights are derived directly from the experimental data and process descriptions found in the source intellectual property, providing a factual basis for evaluating the technology's feasibility. Understanding these details is essential for technical teams assessing the integration of this intermediate into their existing manufacturing portfolios.
Q: What are the primary advantages of this chemical synthesis over fermentation for Norvaline?
A: Unlike fermentation methods which suffer from low yields (approx. 3.7g/L) and long cycles, this chemical route utilizes abundant raw materials like n-butyraldehyde and sodium cyanide, enabling significantly higher throughput and reduced production timelines suitable for industrial scale.
Q: How is chirality controlled in this synthesis process?
A: Chirality is introduced through classical resolution. The racemic aminovaleramide intermediate reacts with optically pure tartaric acid (L or D) to form diastereomeric salts. These salts exhibit different solubility properties, allowing the desired enantiomer to be isolated via selective crystallization and subsequent hydrolysis.
Q: Is this process scalable for commercial API intermediate production?
A: Yes, the process relies on standard unit operations such as liquid-liquid extraction, crystallization, and ion exchange chromatography. The use of commodity chemicals eliminates the biological variability associated with fermentation, ensuring consistent batch-to-batch quality essential for GMP manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Norvaline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful commercialization of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory synthesis to industrial manufacturing is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of chiral norvaline meets the exacting standards required for API synthesis. Our infrastructure is designed to support the complex chemical transformations described in patent CN101007772B, delivering consistency and reliability to our global partners.
We invite pharmaceutical companies and contract manufacturing organizations to collaborate with us to optimize their supply chains for perindopril and other related therapeutics. By leveraging our expertise in chiral resolution and amino acid synthesis, we can help you achieve significant operational efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals.
