Advanced Synthesis of Thieno[3,2-c]pyridine Intermediates for Parkinson’s Drug Development
Advanced Synthesis of Thieno[3,2-c]pyridine Intermediates for Parkinson’s Drug Development
The rapid advancement of neurodegenerative disease therapeutics has placed immense pressure on the supply chain for complex heterocyclic intermediates. Specifically, the development of novel targeting molecules such as PXT002331 (Foliglurax) has highlighted the critical need for efficient synthetic routes to thieno[3,2-c]pyridine derivatives. Patent CN113387959B, published in May 2023, introduces a groundbreaking methodology that addresses the longstanding bottlenecks in producing methyl thieno[3,2-c]pyridine-6-carboxylate. This patent details a robust four-step synthesis starting from readily available 2-thiophenecarboxaldehyde and glycine, achieving a total yield of approximately 49.2%. For R&D directors and procurement strategists, this represents a pivotal shift from low-yielding, expensive legacy processes to a streamlined, cost-effective manufacturing protocol. The structural complexity of the target molecule, often associated with potent antiparkinsonian activity, demands a synthesis that balances chemical precision with economic viability.
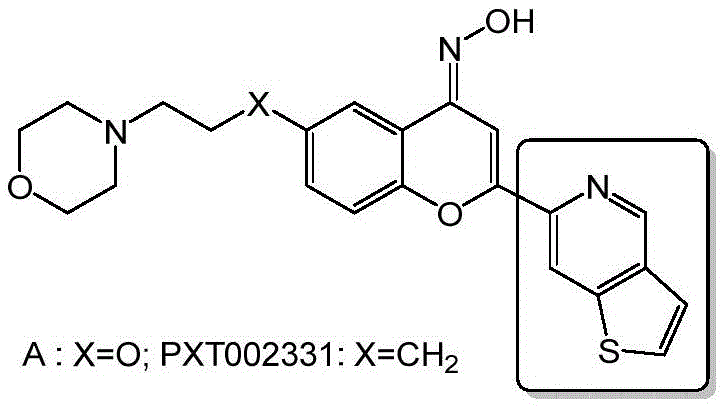
The significance of this patent extends beyond mere academic interest; it provides a tangible solution for the reliable thieno[3,2-c]pyridine supplier seeking to secure long-term contracts with major pharmaceutical developers. By leveraging aldol condensation followed by a Pictet-Spengler cyclization and final aromatization, the inventors have circumvented the regioselectivity issues that plagued earlier attempts. This technical breakthrough ensures that the supply of high-purity pharmaceutical intermediates can be maintained without the volatility associated with scarce starting materials. As we delve deeper into the mechanistic nuances and commercial implications, it becomes clear that this methodology sets a new standard for the commercial scale-up of complex heterocyclic compounds in the neurology sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations described in CN113387959B, the synthesis of thieno[3,2-c]pyridine-6-carboxylic acid methyl ester was fraught with inefficiencies that made industrial adoption nearly impossible. The most prominent prior art, documented in WO2004/39815, relied heavily on a Wittig-Horner reaction using 3-thiophenedicarboxaldehyde as the key starting material. This approach suffered from severe regioselectivity problems, where the reaction pathway favored undesired isomers, resulting in a dismal yield of the designed product of less than 10%. Furthermore, 3-thiophenedicarboxaldehyde is not only expensive but also difficult to source in bulk quantities, creating a fragile supply chain vulnerable to disruptions. For a procurement manager, relying on such a process意味着 accepting high costs and unpredictable lead times, which are untenable for the mass production of life-saving medications. The inability to satisfy mass production requirements meant that many promising drug candidates remained stuck in preclinical phases due to the lack of sufficient material for toxicology and efficacy studies.
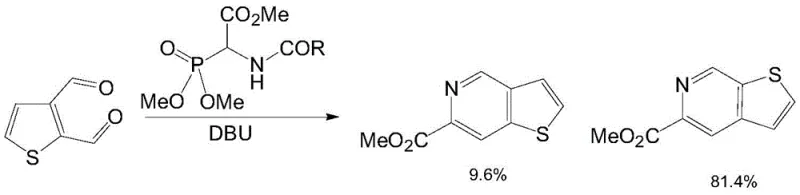
The Novel Approach
In stark contrast, the novel approach outlined in the patent utilizes a convergent strategy that begins with the aldol condensation of 2-thiophenecarboxaldehyde and glycine. This choice of starting materials is strategic; both are commodity chemicals available at a fraction of the cost of the dicarboxaldehyde precursor used in the old method. The new route effectively avoids the adverse factors of the prior art by shifting the bond-forming events to more controllable reactions, such as the formation of a beta-hydroxy-gamma-amino acid intermediate. This intermediate is then seamlessly converted into the cyclic core through a series of well-defined transformations including esterification and cyclization. The result is a total yield of nearly 50%, which is a five-fold improvement over the previous benchmark. This dramatic increase in efficiency translates directly into cost reduction in pharmaceutical intermediate manufacturing, allowing producers to offer competitive pricing while maintaining healthy margins. The robustness of this new pathway ensures that the synthesis of thieno[3,2-c]pyridine-6-carboxylic acid methyl ester can be efficiently and conveniently obtained, meeting the rigorous demands of modern drug development pipelines.
Mechanistic Insights into the Aldol-Pictet-Spengler Cascade
The core of this synthetic innovation lies in the elegant orchestration of four distinct chemical transformations that build the fused thienopyridine ring system with high fidelity. The process initiates with an aldol condensation between 2-thiophenecarboxaldehyde and glycine in an aqueous sodium hydroxide solution. This step generates a beta-hydroxy-gamma-aminothiophene butyrate intermediate, establishing the carbon backbone necessary for the subsequent ring closure. Following isolation, this amino acid undergoes esterification using thionyl chloride in methanol to form the methyl ester hydrochloride salt, which activates the amine for the crucial cyclization step. The third stage involves a Pictet-Spengler reaction where the amine condenses with formaldehyde under acidic conditions. This electrophilic substitution occurs specifically at the beta-position of the thiophene ring, closing the piperidine ring to form thieno[3,2-c]piperidine-7-hydroxy-6-carboxylic acid methyl ester. Finally, the tetrahydro-intermediate is subjected to dehydration and aromatic ring isomerization catalyzed by p-toluenesulfonic acid in dioxane at 100°C. This final aromatization step is critical, as it converts the saturated piperidine ring into the fully aromatic pyridine system, delivering the target molecule with the required electronic properties for biological activity.
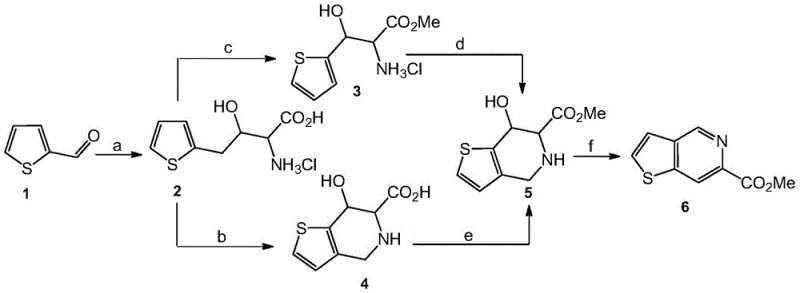
From an impurity control perspective, this mechanism offers significant advantages by minimizing the formation of hard-to-remove side products. The use of crude intermediates in subsequent steps, as permitted by the robustness of the reactions, reduces the accumulation of impurities that often arise from excessive purification procedures. For instance, the beta-hydroxy-gamma-aminothiophene butyrate is used directly after vacuum drying without further chromatography, yet the final product still achieves high purity after a single silica gel column separation at the end. This indicates that the reaction selectivity is inherently high, preventing the generation of complex impurity profiles that would otherwise require extensive downstream processing. Understanding these mechanistic details allows R&D teams to optimize reaction parameters such as temperature and stoichiometry to further enhance yield and purity, ensuring that the final API intermediate meets the stringent specifications required for clinical trials.
How to Synthesize Thieno[3,2-c]pyridine-6-carboxylic Acid Methyl Ester Efficiently
The practical implementation of this synthesis requires careful attention to reaction conditions, particularly during the esterification and cyclization stages where moisture and pH control are paramount. The patent provides a detailed operational framework that guides chemists from the initial condensation to the final isolation of the pale yellow solid product. By adhering to the specified molar ratios and temperature profiles, manufacturers can replicate the reported 49.2% total yield with high consistency. The following guide outlines the standardized procedure derived from the patent examples, serving as a foundational protocol for process development teams aiming to scale this technology.
- Perform aldol condensation between 2-thiophenecarboxaldehyde and glycine in sodium hydroxide solution to form beta-hydroxy-gamma-aminothiophene butyrate.
- Convert the amino acid intermediate into its methyl ester hydrochloride salt using thionyl chloride in methanol.
- Execute a Pictet-Spengler cyclization with formaldehyde under acidic conditions to form the tetrahydro-thienopyridine core.
- Finalize the synthesis via dehydration and aromatic ring isomerization catalyzed by p-toluenesulfonic acid to yield the target pyridine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the bottom line and logistical continuity, the shift to this new synthetic route offers profound strategic benefits that extend far beyond simple yield improvements. The elimination of expensive and scarce starting materials like 3-thiophenedicarboxaldehyde fundamentally alters the cost structure of the supply chain, replacing it with abundant commodity chemicals. This transition mitigates the risk of supply shortages and price volatility, ensuring a steady flow of materials essential for continuous manufacturing operations. Furthermore, the simplified purification requirements reduce the consumption of solvents and stationary phases, contributing to both economic and environmental sustainability goals.
- Cost Reduction in Manufacturing: The most immediate impact is seen in the raw material costs, as the new route utilizes 2-thiophenecarboxaldehyde and glycine, which are significantly cheaper and more accessible than the precursors required for the Wittig-Horner method. Additionally, the ability to use crude intermediates without intermediate purification steps drastically reduces labor, solvent, and waste disposal costs associated with multiple isolation procedures. The avoidance of transition metal catalysts in the final aromatization step further lowers expenses by removing the need for expensive metal scavengers and rigorous heavy metal testing, streamlining the overall production budget.
- Enhanced Supply Chain Reliability: By decoupling production from niche starting materials, manufacturers can secure long-term supply agreements with multiple vendors for the basic building blocks, thereby reducing lead time for high-purity pharmaceutical intermediates. The robustness of the reaction conditions, which tolerate standard industrial equipment and do not require exotic reagents, ensures that production can be easily transferred between different facilities or regions without loss of quality. This flexibility is crucial for maintaining supply continuity in the face of global logistical challenges or regional disruptions, providing a safety net for pharmaceutical partners.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the use of standard unit operations like filtration, extraction, and distillation which are easily adapted from kilogram to tonne scales. The reduction in the number of purification steps and the use of common organic solvents simplify waste management, making it easier to comply with increasingly strict environmental regulations regarding hazardous waste discharge. This alignment with green chemistry principles not only reduces regulatory risk but also enhances the corporate social responsibility profile of the manufacturing entity, appealing to ethically conscious investors and partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this critical intermediate. These answers are derived directly from the technical disclosures in patent CN113387959B and are intended to clarify the feasibility and advantages of adopting this new manufacturing route for your specific project needs.
Q: What is the primary advantage of this new synthesis route over the prior art?
A: The new route utilizes inexpensive 2-thiophenecarboxaldehyde and glycine, achieving a total yield of nearly 50%, which is significantly higher than the less than 10% yield of the previous Wittig-Horner method using expensive starting materials.
Q: Does this process require expensive transition metal catalysts?
A: No, the process relies on organic acids like p-toluenesulfonic acid for the final aromatization step, eliminating the need for costly transition metal catalysts and simplifying purification.
Q: Is the intermediate suitable for large-scale manufacturing?
A: Yes, the method uses common reagents like formaldehyde and thionyl chloride, and the intermediates can often be used crude without purification, making it highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thieno[3,2-c]pyridine-6-carboxylic Acid Methyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful development of next-generation Parkinson's therapies hinges on the availability of high-quality, reliably sourced intermediates. Our team of expert process chemists has thoroughly analyzed the methodology presented in CN113387959B and is fully prepared to implement this advanced synthesis at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your clinical and commercial supply needs are met with precision and timeliness. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of thieno[3,2-c]pyridine derivative we deliver meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this new route can optimize your budget. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that accelerate your path to market while securing a sustainable and cost-effective supply chain for your critical neurodegenerative disease therapeutics.
