Industrial Scale Production of High-Purity Fmoc-Glu(OtBu)-OH via Copper Complexation
Introduction to Advanced Peptide Intermediate Manufacturing
The landscape of peptide synthesis is constantly evolving, driven by the demand for higher purity intermediates that can withstand the rigors of solid-phase peptide synthesis (SPSS). A pivotal development in this sector is detailed in patent CN110746323B, which outlines a robust industrial production method for Fmoc-Glu(OtBu)-OH, a critical building block for numerous bioactive peptides and pharmaceutical agents. This specific derivative, characterized by its orthogonally protected side chain, is essential for constructing complex molecular architectures where selective deprotection strategies are paramount. The traditional challenges associated with glutamic acid derivatives often revolve around the difficulty in distinguishing between the alpha and gamma carboxyl groups during protection steps, leading to difficult-to-separate isomers and compromised yields. The innovative approach presented in this patent addresses these historical bottlenecks by introducing a copper-mediated purification strategy that fundamentally alters the impurity profile of the final product.
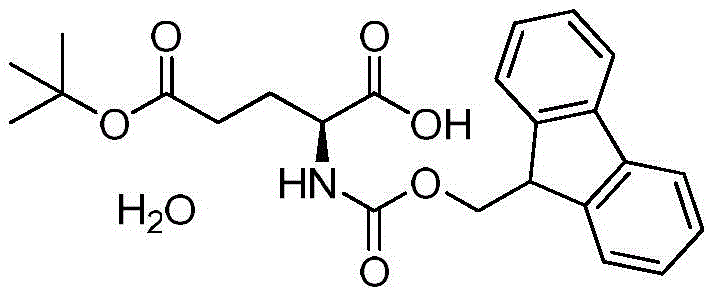
By leveraging the unique coordination chemistry of copper ions with amino acids, the process achieves a level of selectivity and purity that is difficult to match with conventional organic extraction methods alone. The resulting material boasts a purity exceeding 99.5% with single impurities controlled below 0.1%, a specification that is increasingly becoming the standard for GMP-grade active pharmaceutical ingredient (API) manufacturing. For R&D directors and procurement specialists alike, understanding the mechanistic underpinnings of this synthesis is crucial for evaluating supply chain reliability and cost-efficiency. This report delves deep into the technical nuances of this copper-complex route, highlighting how it serves as a reliable pharmaceutical intermediates supplier solution for high-end drug synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Fmoc-Glu(OtBu)-OH has been plagued by issues related to regioselectivity and purification complexity. Conventional routes often involve the direct esterification of glutamic acid followed by N-protection, a sequence that frequently generates significant amounts of the undesired alpha-tert-butyl ester isomer alongside the target gamma-ester. Separating these structural analogs typically requires extensive column chromatography or multiple recrystallization cycles, which drastically reduces overall yield and increases solvent consumption. Furthermore, the removal of trace metal catalysts or acidic residues from earlier steps often necessitates additional washing procedures that can lead to product loss or degradation of the base-sensitive Fmoc group. These inefficiencies not only inflate the cost of goods sold (COGS) but also introduce variability in the quality of the intermediate, posing risks for downstream peptide coupling reactions where stoichiometry is critical.
The Novel Approach
The methodology described in the patent introduces a paradigm shift by utilizing a copper complex intermediate, [Glu(OtBu)]2Cu, as a strategic purification handle. Instead of struggling to purify the free amino acid ester directly, the process converts the crude H-Glu(OtBu)-OH into a stable copper chelate. This transformation is advantageous because the copper complex exhibits distinct solubility properties compared to organic impurities and unreacted starting materials. The patent highlights that this intermediate can be effectively purified through simple water pulping, a technique that is far more scalable and cost-effective than chromatographic separation. Once the copper complex is purified to homogeneity, it serves as a pristine substrate for the subsequent Fmoc protection step. This sequence ensures that the final Fmoc-Glu(OtBu)-OH is derived from a highly pure precursor, inherently limiting the formation of downstream impurities.
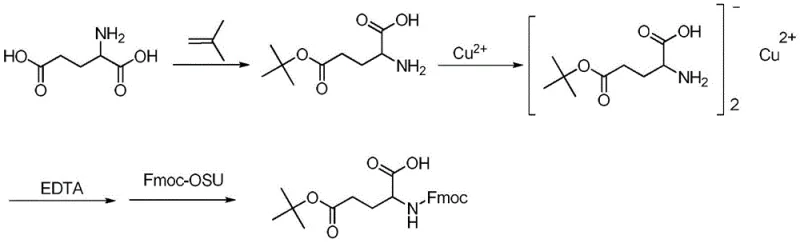
Mechanistic Insights into Copper-Mediated Purification and Protection
The core innovation of this process lies in the exploitation of the coordination geometry of copper(II) ions with amino acids. In the second step of the synthesis, the mono-tert-butyl ester of glutamic acid reacts with copper salts to form a bis-chelate complex. Mechanistically, the copper ion coordinates with both the free alpha-carboxylate and the alpha-amino group, forming a stable five-membered ring structure. This coordination not only protects the amine functionality temporarily but also locks the molecule into a specific conformation that facilitates crystallization or precipitation from aqueous media. The stability of this complex allows for vigorous washing with water to remove soluble organic byproducts, such as di-esters or unreacted glutamic acid, which do not form similar insoluble complexes under the specified pH conditions. This step acts as a powerful 'chemical filter' that significantly enriches the optical and chemical purity of the intermediate before the expensive Fmoc reagent is even introduced.
Following the purification of the copper complex, the introduction of the Fmoc group is achieved using Fmoc-OSU (N-(9-fluorenylmethoxycarbonyloxy)succinimide) under mildly alkaline conditions. The presence of the copper ion actually aids in directing the acylation to the nitrogen atom while the carboxylate remains coordinated. The final and perhaps most elegant step involves the use of EDTA (ethylenediaminetetraacetic acid) to dismantle the complex. EDTA has a significantly higher formation constant for copper than the amino acid does; therefore, upon addition, it sequesters the copper ion into a water-soluble EDTA-Cu complex, releasing the free Fmoc-Glu(OtBu)-OH into the organic phase. This demetallation strategy avoids the use of strong acids or sulfide scavengers that could generate foul-smelling waste or degrade the product, representing a cleaner and more environmentally benign approach to metal removal in fine chemical synthesis.
How to Synthesize Fmoc-Glu(OtBu)-OH Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for scaling this reaction from laboratory benchtop to industrial reactor vessels. The process begins with the acid-catalyzed reaction of L-glutamic acid with isobutene to install the tert-butyl ester, followed by the critical copper complexation and purification phase. The final stage involves the coupling with Fmoc-OSU and the subsequent EDTA workup to isolate the target molecule. Each step has been optimized to balance reaction kinetics with ease of isolation, ensuring that the process remains robust even when handling multi-kilogram batches. The detailed standardized synthetic steps for implementing this high-efficiency route are provided in the guide below, offering a practical reference for process chemists looking to adopt this technology.
- React L-glutamic acid with isobutene in the presence of an acid catalyst (e.g., sulfuric acid) to form the mono-tert-butyl ester intermediate H-Glu(OtBu)-OH.
- Complex the intermediate with a copper salt (e.g., CuSO4) to form [Glu(OtBu)]2Cu, followed by rigorous water pulping purification to remove impurities.
- React the purified copper complex with Fmoc-OSU under alkaline conditions, then use EDTA to remove the copper ion and isolate the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-complex mediated synthesis offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the purification workflow. By replacing complex chromatographic separations with aqueous pulping and crystallization, the process significantly reduces solvent usage and processing time. This reduction in unit operations translates directly into lower manufacturing costs and a smaller environmental footprint, aligning with modern green chemistry initiatives. Furthermore, the ability to consistently achieve purity levels above 99.5% minimizes the risk of batch rejection during quality control testing, thereby enhancing supply chain reliability and reducing the safety stock requirements for downstream peptide manufacturers.
- Cost Reduction in Manufacturing: The elimination of expensive chromatography resins and the reduction in solvent volume required for purification lead to substantial cost savings. Additionally, the use of commodity chemicals like copper sulfate and EDTA keeps raw material costs low compared to specialized protecting group reagents. The high yield of the copper complex formation (reported between 70-90%) ensures that the expensive Fmoc-OSU reagent is reacted with a high-quality substrate, minimizing waste of this costly coupling agent and optimizing the overall atom economy of the process.
- Enhanced Supply Chain Reliability: The robustness of the water-pulping purification step makes the process less sensitive to minor variations in reaction conditions, ensuring consistent batch-to-batch quality. This reliability is crucial for long-term supply contracts where specification adherence is mandatory. Moreover, the raw materials required, such as L-glutamic acid and isobutene, are widely available commodities, reducing the risk of supply disruptions associated with niche specialty chemicals. The simplified workflow also shortens the production cycle time, allowing for faster turnaround on orders and improved responsiveness to market demand fluctuations.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind, utilizing standard unit operations like filtration, centrifugation, and crystallization that are easily transferable to large-scale glass-lined reactors. The avoidance of heavy metal scavengers and the efficient recovery of copper through EDTA complexation simplify waste treatment protocols. The aqueous waste streams generated are primarily saline and organic-salt solutions that are easier to treat compared to the complex mixed-solvent wastes generated by chromatographic purifications, facilitating compliance with stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Fmoc-Glu(OtBu)-OH using this novel copper-complex method. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and quality parameters associated with this synthesis route. Understanding these details is vital for technical teams evaluating the feasibility of integrating this intermediate into their existing peptide synthesis workflows.
Q: How does the copper complexation step improve the purity of Fmoc-Glu(OtBu)-OH?
A: The formation of the [Glu(OtBu)]2Cu complex allows for effective purification through simple water pulping, removing unreacted starting materials and side products before the critical Fmoc protection step, ensuring final purity exceeds 99.5%.
Q: What are the advantages of using EDTA in the final deprotection step?
A: EDTA acts as a strong chelating agent that efficiently sequesters copper ions from the intermediate complex under mild alkaline conditions, facilitating a clean release of the free amino acid derivative without harsh acidic treatments that could degrade the Fmoc group.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process utilizes common industrial reagents like isobutene and copper sulfate, avoids chromatography by using crystallization and pulping for purification, and demonstrates robust control over impurities, making it highly scalable for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fmoc-Glu(OtBu)-OH Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity amino acid derivatives play in the successful development of next-generation therapeutics. Our technical team has extensively analyzed the copper-complex mediated synthesis route described in CN110746323B and possesses the expertise to implement this process at a commercial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the stringent purity specifications required for GMP applications are consistently met. Our rigorous QC labs are equipped to verify the low impurity profiles (<0.1% single impurity) and high optical purity necessary for demanding peptide coupling reactions, providing our partners with a secure and high-quality supply of this essential building block.
We invite pharmaceutical companies and contract research organizations to collaborate with us to optimize their supply chains for peptide intermediates. By leveraging our advanced manufacturing capabilities, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized production of Fmoc-Glu(OtBu)-OH can enhance the efficiency and cost-effectiveness of your drug development programs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
